convenient, should not obviate the need to document the exact renal tract dysmorphology found in a particular patient, nor should it be assumed that there will be one underlying pathogenesis for CAKUT. Thus, the use of the term is analogous to using a disease group designation such as “glomerulonephritis” or “tubulopathy.” As medical and surgical therapies are refined, a new cohort of young babies with severe kidney malformations and renal failure, who might otherwise have perished, are surviving childhood (8,9); as yet, it is too early to know how they will fare through adulthood.
duct. In the female, apart from a few vestigial and inconstant structures, such as the epoophoron, the paroophoron, and Gartner duct, the whole mesonephros degenerates during the 3rd month of gestation.
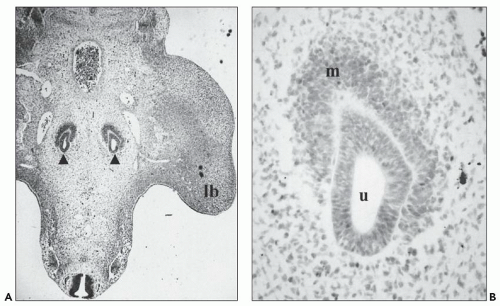
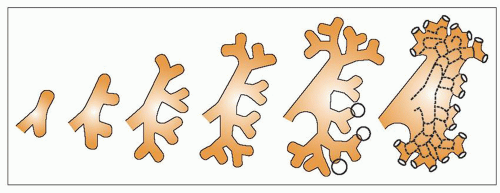
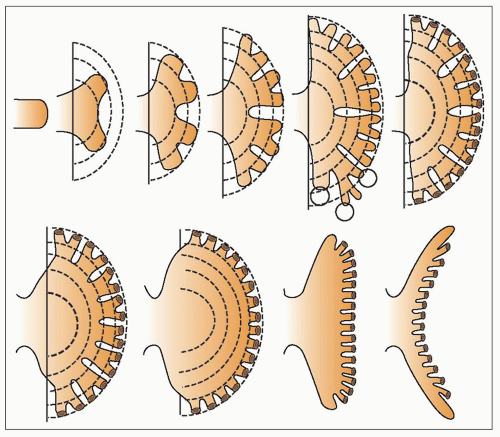
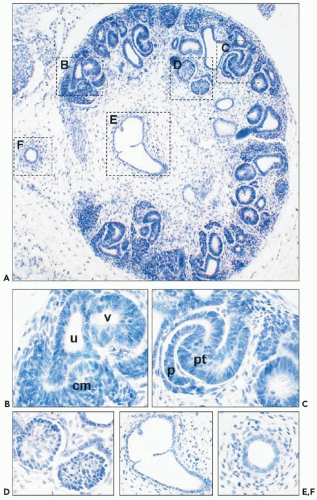
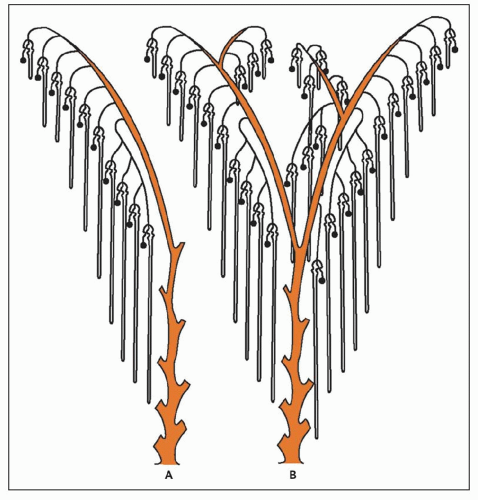
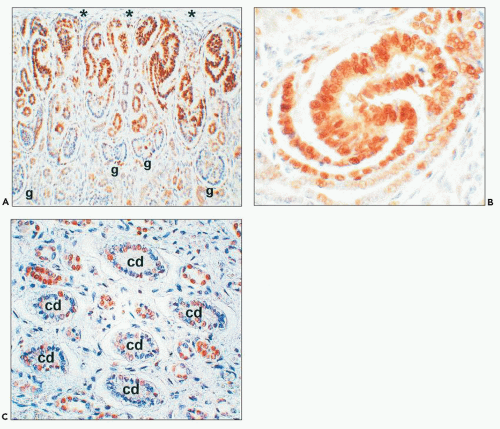
branching is so rapid at this stage, the interstitial portions of the branches are very short, and indeed, sometimes a number of branches appear to arise from a single stem. Distension of the whole system, which is ascribed to the onset of urine production as nephrons start to function, results in the coalescence of the first generations of branches to give the more familiar appearance of the pelvicaliceal system seen postnatally. This is completed by about the 10th to 12th week of gestation. When the minor calyces are formed, some 20 or so ampullae are related to each minor calyx, and it is from these that the papillary collecting ducts originate. Further branching of these ampullae is associated with nephron formation, and these branchings proceed more slowly with longer intervening periods of interstitial growth. From about the 11th week, the spherical expansion of the calyces is limited by the development of nephrons associated with their own and surrounding collecting duct systems that form the developing renal papillae. The minor calyces are indented by the papillae and change from spherical to a wineglass shape, with the cup-shaped portion around the developing papilla and a narrower stem-like infundibular portion connecting to the major calyx. This process is achieved by about the 14th week of gestation
and can be appreciated in histologic sections of the fetal kidneys at this gestational age.
arcades. Each ampulla ceases to divide and becomes capable of inducing the formation of a further three to six nephrons. With the induction of further nephrons, the connecting tubule of the older nephron shifts the position of its point of attachment away from the ampulla to the connecting tubule of the next-formed nephron, so they are joined together in a string or arcade of between four and seven nephrons. The innermost and first-formed member of each nephron arcade is a nephron formed during the first period of nephrogenesis and becomes a juxtamedullary nephron in the fully developed organ. The loops of Henle are longest in these nephrons, extending almost to the papillary tip.
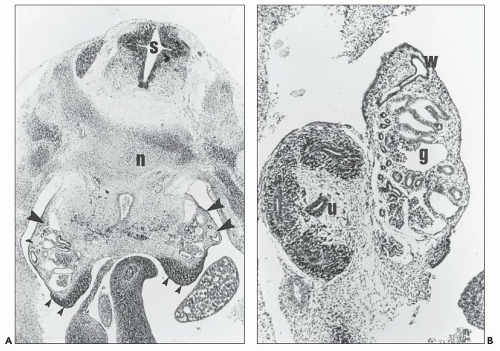
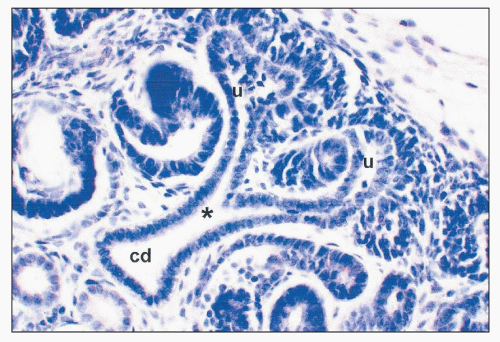 FIGURE 2.5 Metanephros from a 7-week human fetus. A branch point (asterisk) of the ureteric bud/collecting duct (cd) lineage leading to two ureteric bud ampullae (u). (hematoxylin, ×100.) |
producing the subcapsular so-called nephrogenic zone that can be seen in the fetal kidneys up to 36 weeks and is useful in dating gestational age.
that documents the expression patterns of many thousands of gene transcripts through mouse renal tract development (60).
(69), and the PAX family, which contain DNA-binding paired domains (70,71,72) (Fig. 2.9). The specific gene targets of certain of these transcription factors are being investigated. For example, WT1 down-regulates PAX2 (73); up-regulates amphiregulin, a growth factor that stimulates tubule formation in the metanephros (74); and modulates the expression of WNT4, a growth factor implicated in early nephron differentiation (67). Another transcription factor called hepatocyte nuclear factor 1β (HNF1β) promotes the expression of a battery of genes that maintain the differentiated state of renal epithelia; when HNF1β is mutated, renal tubules become cystic (36,65,75,76).
TABLE 2.1 Genetic bases of mouse kidney defects based on mutants | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree