Chapter 8 IMAGING OF THE FEMALE GENITOURINARY TRACT
REVIEW OF IMAGING MODALITIES
Projectional Radiography
In uroradiology, the role of projectional radiography, once an integral part of diagnosis, has been steadily diminishing as cross-sectional techniques have become more advanced (Fig. 8-1). X-ray–based images are now routinely captured on digital media instead of film, enabling rapid storage on a central server with multiple display options, including a picture archiving and retrieval system (PACS), or for distribution on the Internet. However, the principles of x-ray–based diagnosis have not changed. An image is formed when diagnostic-range x-ray energy interacts with the electron cloud of different human tissues (i.e., bone, soft tissue [water], fat, and air). The different electron densities of these tissues allow various degrees of x-ray beam penetration, resulting in differences in exposure (i.e., shades of gray) on the receptor film or digital media. Most naturally occurring electron-dense structures, such as calcified bone or dental enamel, attenuate x-ray energy strongly and by convention appear white on the conventional or digital receptor. Conversely, the least electron-dense substances, such as gas, allow almost 99% of x-ray energy to penetrate the tissue and reach the receptor, and by convention, these images are black. Between these two extremes are the shades of gray, which represent the summation of various three-dimensional (3D) electron densities on a two-dimensional (2D) receptor. Perception and interpretation of projectional images is highly operator dependent and requires a large degree of experience for proper interpretation.
A patterned approach to the abdominal radiograph is useful for instruction. On most radiographs, the lower poles of the kidneys are apparent with a laterally divergent axis that usually parallels the psoas muscles when visualized. Renal calculi are usually seen as ovoid or toothlike opacities overlying the kidneys or proximal ureters. They are more difficult to detect over the distal ureter because contrast is reduced by overlying bony pelvis. Gas projecting over the bladder is usually seen after recent Foley catheterization, but it may be caused by infections and fistulas. Bony abnormalities should not be overlooked because they can represent metastatic disease or suggest a neurogenic bladder, especially in patients with spinal abnormalities.1
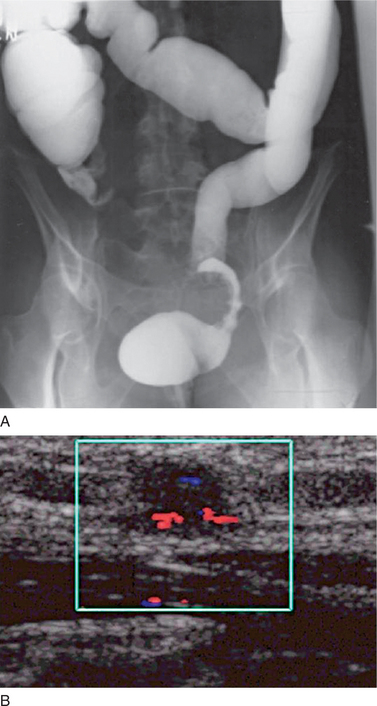
To address the problem of poor soft tissue attenuation differences and to image gross pathologic conditions, various radiographic contrast agents (usually iodinated agents with high electron density especially suited for x-ray interaction) were introduced into the field of uroradiology. The first introduction of retrograde urothelial injection agents in the 1920s was followed by the development of intravascular contrast agents, giving rise to intravenous urography, a mainstay of uroradiology for renal and ureteral evaluation for almost 50 years (Figs. 8-2 and 8-3).2 A large variety of catheter-injected uroradiologic studies were developed for niche applications, including urethrograms, cystograms, pyelograms, loopograms, nephrostograms, and suprapubic cystograms.
Fluoroscopy
Fluoroscopy is the dynamic counterpart of static projectional radiography because it displays real-time images of a particular process under investigation. During fluoroscopic studies, a low dose of continuous x-ray beams passes through the patient to an image intensifier to generate images on a high-resolution television monitor.3 The examiner can obtain a series of individual snapshots (i.e., frames per second) of the process, so-called spot films, or a real-time movie such as evaluation of dynamic processes for storage and review. However, fluoroscopic imaging is best performed with iodinated contrast material for genitourinary applications. Fluoroscopy is most useful for vascular applications, but it may be used for real-time evaluation of ureteral peristalsis; bladder filling and emptying, especially during voiding (i.e., voiding cystourethrography [VCUG]); and guiding a variety of genitourinary interventions, such as nephrostomy tube placement.
Cystography and Voiding Cystoureterography
VCUG is performed as an adjunct to a cystogram to determine the morphology of the bladder and urethra, especially if high-pressure vesicoureteral reflux and related complications are suspected. The bladder is filled, as in static cystography, followed by fluoroscopy and/or a video recording of the voiding process once the catheter is removed.1
Video Urodynamics
In video urodynamics (VUDs), multichannel urodynamic measurements are combined with concomitant fluoroscopic VCUG to determine whether morphologic findings with regard to bladder function and its outlet correlate with physiologic measurements. It is therefore possible to go beyond the level of a simple VCUG or cystometry by obtaining simultaneous measurements of abdominal and vesical pressures under fluoroscopic guidance. In addition to revealing a great deal about dynamics of the bladder and its outlet, VUDs has led to greater diagnostic and therapeutic efficacy. With modern advent of digital recording, the examiner can manipulate the data with various software packages with incorporation of other clinical information and statistical calculations to maximize the yield even further. Some of the most common indications for VUDs in women are various kinds of incontinence; bladder outlet obstruction, as can be seen in urethral obstruction; neuropathic bladder due to a variety of neurologic disorders; geriatric incontinence; urinary diversion; and unidiversion.4
The main problem with VUDs is that it often may not reproduce the presenting symptoms of the patient because it is nonphysiologic in nature and is performed in surroundings unfamiliar to the patient. The presence of severe grades of cystoceles, rectoceles, enteroceles, and uterine prolapse can lead to a rather unreliable interpretation of the examination results. In such clinical scenarios, it is especially important to repeat the study in a variety of positions to assess the severity of the pelvic floor pathology present in a given patient. Despite these limitations, VUDs is still highly recommended in patients with postoperative incontinence and in neuropathic and refractory cases.4
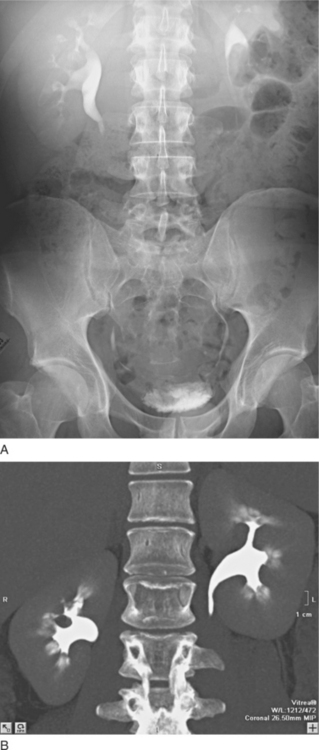
Intravenous Urography and Pyelography
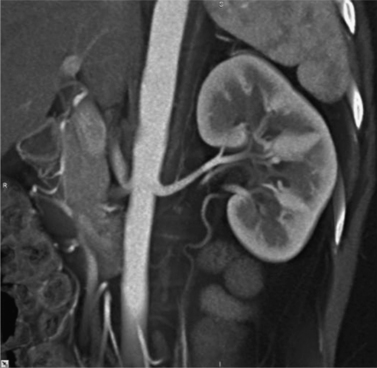
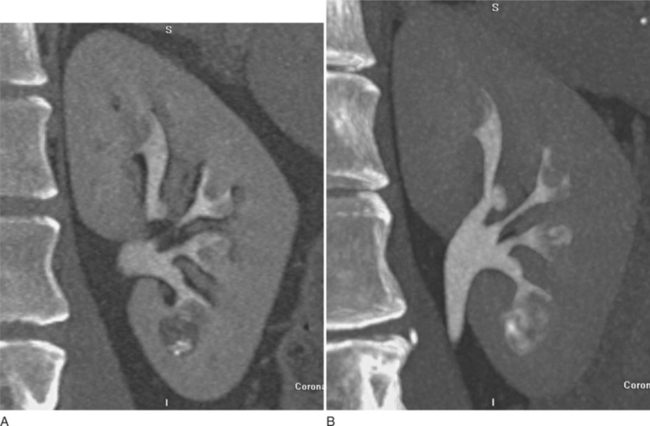
The traditional intravenous urogram (IVU), which is used to evaluate the kidneys, kidney function, and ureteral excretion and function, has undergone a radical transformation over the past 10 years, with increasing resolution and performance of CT, magnetic resonance imaging (MRI), and ultrasound (see Figs. 8-2 and 8-3). Evaluation of the kidneys is best performed with contrast-enhanced CT or MRI (Figs. 8-4 and 8-5). Evaluation of the ureters also may be performed with CT or MRI, although a fluoroscopic or radiographic examination, if performed properly, may be adequate, especially when detecting urothelial malignancies. Advances in competing technologies have limited or severely restricted the clinical application of the IVU. The intravenous urogram is no longer indicated for many upper tract indications, such as renal mass evaluation and evaluation of acute renal colic, and is best reserved for evaluation of hollow parts of the urinary system, such as follow-up of patients with transitional cell carcinoma.1
Retrograde Pyelography
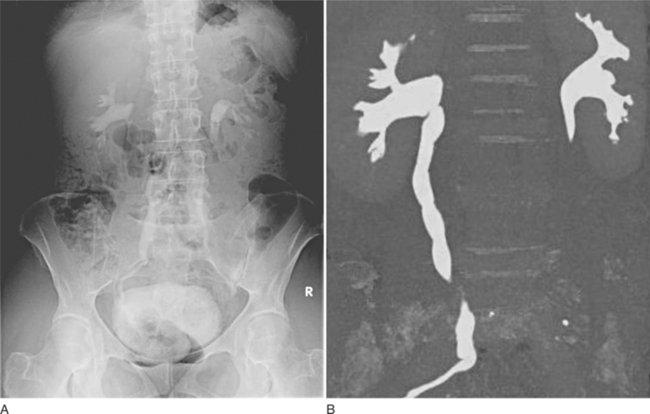
Retrograde pyelography allows morphologic evaluation of the collecting system and ureters. It involves retrograde injection of contrast material directly into the ureters after their cystoscopic cannulation to visualize the collecting system and the ureter without reliance on renal contrast excretion. It also is possible to biopsy suspected urothelial lesions in the course of the examination. Some of the most common indications for retrograde pyelography are further evaluation of congenital anomalies, urinary obstruction, and possible filling defects detected during other studies. However, because this is a rather invasive diagnostic study associated with several potential complications, it usually is not used as an initial diagnostic procedure.1
Ultrasonography
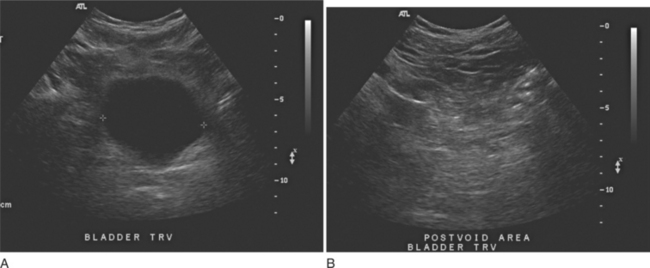
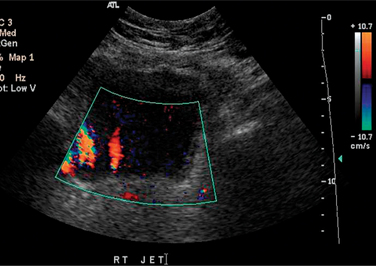
In sonography, also referred to as ultrasound or ultrasonography, sound waves with frequencies in the range of 2 to 20 MHz are generated by an array of piezoelectric crystals in a hand-held transducer. These tightly focused, high-frequency sound waves are propagated through human tissues and reflected back at tissue interfaces to the transducer’s crystals, subsequently producing a diagnostic medical image.3 The choice of imaging frequencies is determined by the size and composition of the tissue involved. Higher frequencies (>7 MHz) usually lead to improved spatial resolution at the expense of depth of imaging, whereas lower frequencies (2 to 5 MHz) enable imaging of deeper tissues with lower spatial resolution. The chief advantages of sonography are its lack of ionizing radiation, its real-time imaging capability, its 2D and 3D imaging capability, and its ability to depict flow direction and velocity in blood vessels and tissues.4 Disadvantages of sonography are its operator dependence and its inability to image through hollow viscera or bone. In urology, ultrasound is the initial test of choice for adult and pediatric renal and bladder imaging. A wide variety of pathologies, such as congenital anomalies, hydronephrosis, and vascular disorders, may be diagnosed. In bladder applications, it may be used to determine residual postvoid urine volume and to delineate the urethrovesical anatomy (Figs. 8-6 and 8-7).5
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree