Chapter 47 SURGERY FOR REFRACTORY URINARY INCONTINENCE: SPIRAL SLING
Treatment of stress urinary incontinence (SUI) caused by urethral hypermobility or intrinsic sphincter deficiency with urethral sling procedures may yield success rates of up to 80% to 90%, depending on the definition of success.1–5 In a minority of patients, however, there is persistence or worse incontinence after surgical therapy. Female patients with urethral incompetence and severe incontinence due to multiple failed surgeries, neurologic injury, or congenital anomalies represent a unique surgical challenge. Patients with neurologic conditions may have sacral arc lesions with paralysis of the skeletal musculature and an open urethra. Other patients who have failed multiple sling and anti-incontinence procedures may have severe symptoms of SUI and an open urethra with a low Valsalva leak point pressure (VLPP). These patients often have an incompetent, difficult to compress urethra, most likely resulting from a combination of urethral denervation and violation of the periurethral fascia, as well as their underlying risk factors for SUI.6 These patients have low chances of cure after repeat anti-incontinence surgery, and they are more likely to suffer from complications including retention.7 In this patient population, a routine sling procedure providing only posterior support will not typically yield an appropriate response. Management options include placement of a “tight” pubovaginal sling, with the intent of putting the patient in urinary retention, or constructing an artificial urinary sphincter (AUS). Many of these patients are often forced to undergo bladder neck closure with continent catheterizable augmentation.
This chapter describes a transvaginal sling procedure in adult women that encircles the urethra, providing circumferential coaptation. The spiral sling is a salvage procedure for a small, yet severely affected group of female patients with a totally incompetent urethra. The procedure was initially described in patients with congenital or neurologic diseases.8 It has more recently been described in patients with multiple failed surgeries for SUI.9 In our experience, it provides better outcomes and fewer complications than the AUS.
In addition, we discuss the AUS as a treatment option for the female patient with urinary incontinence. The AUS is an effective alternative to the urethral sling for the treatment of urinary incontinence caused by primary urethral sphincter insufficiency (type III SUI).10 The AUS is a synthetic device that includes an internal cuff made of silicone rubber with Dacron reinforcement and is designed to provide a uniform circumferential compression on the urethra. Continence or cuff inflation is maintained by continuous pressure in the cuff, which is modulated by the pressure balloon, which in turn is usually positioned in the prevesical space. The patient manipulates a labially placed pump to open the cuff before voiding. The American Medical System (Minnetonka, MN) AS-800 is the currently used model; it may be implanted via a transvaginal,10,11 transabdominal,12 or laparoscopic transperitoneal approach.13 In this chapter, we limit our discussion to the transvaginal approach.
THE SPIRAL SLING
Surgical Technique
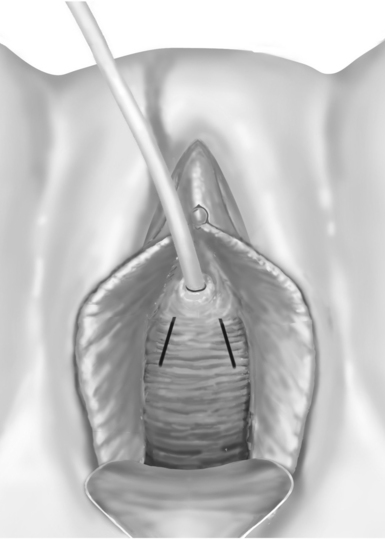
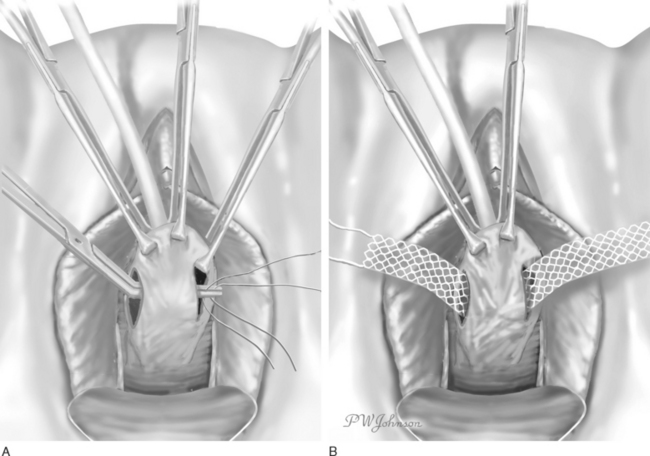
An Allis clamp is used to grasp the anterior vaginal wall just proximal to the meatus. Two parallel distal oblique incisions are made in the anterior vaginal wall (Fig. 47-1). One additional Allis clamp is placed at the distal portion of each of the oblique incisions to aid in the exposure. The dissection is carried out laterally, over the glistening periurethral fascia, toward the ipsilateral shoulder. The retropubic space is entered, and a complete urethrolysis is performed by detaching the urethropelvic ligaments from the arcus tendineous fascia pelvis and freeing all retropubic adhesions (Fig. 47-2). The urethral dissection is started in the midurethral area just proximal to the pubourethral ligaments and carried proximally to free the rest of the urethra and the bladder neck. The dissection is maintained close to the periosteum of the pubic bone to avoid injuries to the dorsal venous complex, bladder, and urethra. The dorsal aspect of the urethra is freed from the pubis. A suburethral tunnel is created in the anterior vaginal wall, 1.5 cm from the urethral meatus, with a fine, right angle clamp connecting the two oblique incisions (Fig. 47-3A).
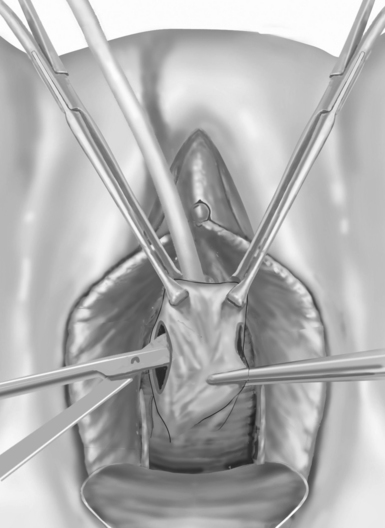
Figure 47-2 The retropubic space is entered at the level of the mid-urethra to distal urethra using curved scissors.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree