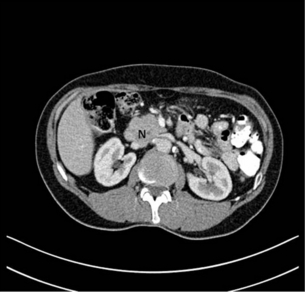
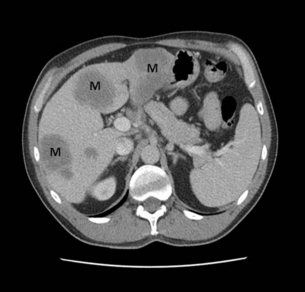
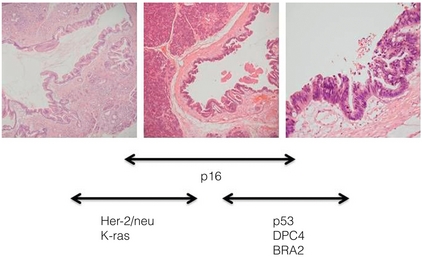
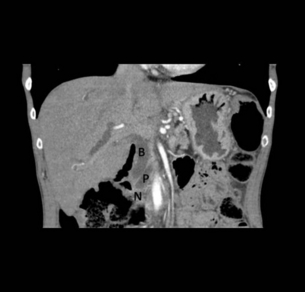
15 Adenocarcinoma of the pancreas accounts for 3% of new cancer cases per annum,1 yet it is the fourth leading cause of cancer-related death in Western countries. The insidious nature of the disease and its vagueness of presentation contribute to late diagnosis. Eighty per cent of patients have unresectable tumours at initial diagnosis. The overall survival at 5 years still remains at 6%, unchanged over the last four decades.1 However, in recent years improvements in preoperative imaging and staging modalities, coupled with advancements in adjuvant therapies with the use of immunomodulators and monoclonal antibodies, have resulted in some degrees of optimism. Progress has been made as the molecular basis of the disease is better understood. With such poor survival rates and recent high-profile media attention, a renewed interest in tackling this elusive cancer has arisen. An estimated 44 000 new cases of adenocarcinoma of the pancreas occurred in the USA in 2011, and almost 38 000 died in the same year.2 In the UK, 8085 people were diagnosed with pancreatic cancer and 8020 people died during 2009.3 The incidence of pancreatic cancer varies with age, sex and ethnicity. In 2008, the standardised incidence rate of pancreatic cancer was 3.9 per 100 000 population, while the standardised mortality rate was slightly lower at 3.7 per 100 000 population. Pancreatic cancer is the eleventh commonest cancer in males and eighth commonest cancer in females.4 The peak incidence for the disease occurs between the seventh and eighth decades of life, and is rare under the age of 30. In the American population, African and Hawaiian ethnicities confer a higher incidence than Caucasian, whereas Asian and Hispanic ethnic groups have a lower risk of developing the disease. The incidence of pancreatic cancer is rising, particularly in Europe, although this observation is subject to reporting bias related to improved diagnostics. However, pancreatic neoplasm must be detected at an early stage to enable the potential for curative treatment. Tobacco smoking is by far the leading preventable cause of pancreatic cancer, with an estimated 2.5-fold increase in risk when compared to non-smokers.5 In 1986, the International Agency for Research on Cancer (IARC) classified smoking as a proven carcinogen with respect to cancer of the pancreas. Observational studies suggest that a dose-dependent relationship exists, necessitating long-term exposure.5 However, smokers who have quit for more than 10 years no longer experience an increased risk.5 While the chemical cause is unclear, it is hypothesised that N-nitroso compounds in tobacco are carried to the pancreas in the blood. The time in which cigarette smoking exerts its negative influence is also subject to debate; however, observational studies seem to point towards the latter stages of carcinogenesis, particularly in the 15 years preceding development. Excessive body weight appears to increase the risk of pancreatic cancer. It has been shown that obesity has a positive association, with a relative risk of 1.72.6 Diets high in saturated fat have a suggested contributing role in carcinogenesis,6 although data are limited. Caffeine and meat preservatives have been suggested to have a negative association, but in recent years this has become more debated due to the studies having methodological flaws, and more recent studies demonstrating the opposite. Vitamin C, vitamin D and high-fibre diets have suggested protective associations with pancreatic neoplasm.7–9 Workers exposed to ionising radiation, insecticides, aluminium, nickel, acrylamide and halogenated hydrocarbons are reported to have an increased risk of developing pancreatic adenocarcinoma. There is evidence of increased risk in people exposed to chlorinated hydrocarbon solvents (metal degreasing workers and dry cleaners), and those working in the paint and varnish industry and the textiles industry.10 Chronic pancreatitis is a progressive inflammatory process with associated irreversible histological changes. It is highly linked to excessive alcohol consumption, with up to an 18-fold increase in risk of pancreatic cancer compared to the general population. Quantifying the risk is still difficult due to confounding factors such as smoking, alcohol and diet.11 Diabetes has a positive association for pancreatic cancer. Meta-analysis has shown that type 2 diabetes increases the risk of pancreatic cancer by 82%.11 The high prevalence of diabetes in society excludes hyperglycaemia as a screening tool for pancreatic cancer. Pancreatic carcinogenesis has an established genetic predisposition. Familial conditions such as Peutz–Jeghers syndrome, germ-line mutation in the STK11/LKB11 gene,13 BRCA2 expression14 and familial atypical multiple mole melanoma (p16/CDKN2A germ-line mutation) may predispose to pancreatic cancer.14 Links with hereditary non-polyposis colorectal cancer (Lynch syndrome), BRCA1 and von Hippel–Lindau have been suggested but not confirmed, as conferring increased risk.15 With the speed of developing technology, matched with reduced genetic diagnostic expense, one can foresee the potential for the discovery of further genes relating to familial pancreatic cancer. Histologically distinct precursor lesions have been attributed to pancreatic carcinogenesis. Preneoplastic lesions are usually asymptomatic and can be incidentally discovered at the time of resection. They appear to follow a multi-step progression to invasive carcinoma.15 These precursor lesions include pancreatic intraepithelial neoplasia (PanIN), intraductal papillary mucinous neoplasm (IPMN) and mucinous cystic neoplasm (MCN).16 Because of their small size (usually < 5 mm), they are difficult to detect, making them elusive to computerised tomography (CT) or magnetic resonance imaging (MRI).15 Pan-INs are the most frequent preneoplastic lesions, observed in approximately 82% of pancreas with neoplasm.17 They are subclassified into PanIN-1, PanIN-2 and PanIN-3 depending upon the degree of cytological and architectural atypia.17 Each of these precursor lesions harbours a unique repertoire of clinicopathological and genetic characteristics that has an impact on the natural history and prognosis of these lesions. Workers in Johns Hopkins University proposed pancreatic intraepithelial neoplasms (PanIN; 1A → 1B → 2 → 3) as the precursor lesions to invasive carcinoma.18 The model is analogous to that of ductal carcinoma in situ (DCIS) of the breast or adenomatous polyps in colorectal cancer. The lesions display atypical mucinous epithelium replacing the physiological cuboidal epithelium. The evidence for PanIN being a true premalignant state is largely circumstantial. These lesions were first described adjacent to resected adenocarcinoma. The more atypical PanIN-2 and -3 were seen exclusively in neoplastic pancreas. These lesions also display similar genetic aberrations to the frankly invasive samples. In particular, the percentage of p16 and K-ras mutations increases with the more atypical PanIN. These data have heralded development of a tumour genesis model involving sequential progression from PanIN-1a to invasive adenocarcinoma.19 Classically, evolution from precursor lesions to pancreatic neoplasm (ductal adenocarcinoma) involves diverse molecular changes (Fig. 15.1). Recent studies indentified at least 119 independent loci that may potentially play a role in tumour progression, including K-ras, TP53, p16/CDKN2A, MYC and AKT2.20 The K-ras gene product mediates signal transduction in a number of growth factor receptors. K-ras single point mutation is observed in 90–95% of pancreatic ductal adenocarcinoma, representing the most common mutation in this disease.21 K-ras is currently the focus of multiple ongoing studies to see if it can be utilised as a diagnostic tool.13 Altered epidermal growth factor receptor expression (EGFR) causing overexpression is thought to be an early event in pancreatic carcinogenesis.22 Figure 15.1 Diagrammatic representation of the multi-step progression to invasive carcinoma from low-grade neoplasm on the left to high-grade on the right. Images courtesy of Dr Paul Crotty. Inactivation of numerous tumour suppressor genes, including p16/CDKN2A and TP53, plays a pivotal role in the development of pancreatic cancer. Loss of tumour suppression is noted in 70–95% of pancreatic neoplasm.17 Other targets including transforming growth factor-β (TGF-β) receptor genes, BRCA2, HER-2/NEU, DPC4, MKK4 and EBER-1 are currently under investigation. The discussion about these genes is outside the scope of this chapter. However, the best chance for cure in the treatment of pancreatic cancer lies with detecting these non-invasive lesions before progression to invasive carcinoma. The majority of patients present with vague and non-specific symptoms (Box 15.2). As a result, the disease is commonly widespread at diagnosis, and approximately 80% of patients present with unresectable disease. Tumours in the body and tail of the pancreas usually present late. Pain is the most consistent symptom. Painless jaundice is seen in 13% of patients while 34% present with only pain and 46% present with both pain and jaundice. Weight loss and anorexia are observed in 7% of patients. Rarely, tumour invasion into stomach or duodenum can present as haematemesis and malaena. Patients may also present with late-onset diabetes mellitus and acute pancreatitis.23 Limited series have examined the screening of asymptomatic cohorts, with little evidence to support the introduction of population screening for pancreatic cancer.14 However, there may be a place for targeted screening of high-risk groups in the near future. There still remains no ideal tumour marker for pancreatic carcinoma. Carbohydrate antigen 19-9 (CA 19-9; 0–37 U/mL) exists in tissue as an epitope of sialylated Lewis a-type blood group antigen, and is the most widely utilised tumour marker. It was based on a monoclonal antibody to colorectal cancer cell lines. CA 19-9 is elevated in only approximately 50% of cases.24 Among symptomatic patients, CA 19-9 has a sensitivity of 81–85% and specificity of 81–90%.24 However, the positive predictive value remains low among the asymptomatic population, making it a very poor screening test. Falsely elevated CA 19-9 is documented in other neoplasms including gastric, colorectal, cholangiocarcinoma and urothelial malignancies, as well as benign conditions such as pancreatitis, hepatitis, thyroiditis and biliary obstruction. In addition patients expressing Lewis blood group antigens (a and b) may have elevated levels.25 CA 19-9 may be used to assess recurrence of disease, with a level higher than 500 U/mL signifying advanced disease.25 A level exceeding 243 U/mL for patients undergoing primary chemoradiotherapy for locoregionally advanced disease also indicates poorer median survival (7.1 vs. 12.3 months).26 Several other tumour markers are currently being investigated including carcinoembryonic antigen (CEA), K-ras, p53, CA242, CA50, SPAN-1, DU-PAN2, CAM-17.1 and a number of mucins (MUC1, MUC3, MUC4 and MUC5AC). They are proposed as having application in pancreatic neoplasms, although none of these markers are sensitive enough to be recommended for clinical use. CA242 shows promise as an independent prognostic factor.27 Transabdominal (TA) ultrasound (US) is the initial investigation in the jaundiced patient. It is noted for its superior sensitivity for determining cholelithiasis over CT. Common bile duct dilatation (> 7 mm; > 10 mm in post-cholecystectomy patients) is an indirect sign, together with pancreatic duct dilatation (> 2 mm). The primary pancreatic lesion is often visible together with liver metastases and ascites if present. For lesions > 3 cm TAUS has approximately 95% sensitivity; however, this is considerably lower for smaller lesions.28 The main criticism of TAUS is machine quality difference and operator experience; thus it is user dependent.29 The role of colour Doppler US has been suggested to examine portal vein or superior mesenteric involvement. Ultrasound remains a useful imaging modality for the initial screening of the jaundiced patient, but further radiological modalities are necessary to examine the pancreas and assess resectability. CT remains the most common staging modality. Conventional CT has been replaced by more sensitive dynamic CT with thinner slice/cuts (1–3 mm) with multidetector and 3D reconstruction. The sensitivity is approximately 90% for lesions greater than 2 cm, decreasing to approximately 60% for smaller lesions.30,31 CT allows for assessment of the primary lesion, its relationship to the remainder of the pancreas and peripancreatic vasculature, and determination of resectability (Figs 15.2 and 15.3). Direct evidence of a tumour is often seen as a hypodense mass, with other subtle signs such as pancreatic atrophy, deformity of the glandular contour or dilatation of the common bile and pancreatic ducts (Fig. 15.4). Metastatic lesions can be detected, and portal vein or superior mesenteric artery involvement can be determined. Figure 15.4 CT scan of coronal view demonstrating biliary duct (B), pancreatic duct (P), obstruction by pancreatic neoplasm (N) denoting the double duct sign. MRI is mainly used as an adjuvant to other imaging modalities for planning treatment options. The combination of T1/T2-weighted imaging with magnetic resonance cholangiopancreatography (MRCP) is useful to visualise the primary tumour and its relationship to the biliary and pancreatic ducts, as well as peripancreatic vasculature. MRI/MRCP is considered to be equivalent to CT for assessing primary disease.33,34 Positron emission tomography (PET) shows accumulation of [18F]2-fluoro-2-deoxy-D-glucose (FDG) by tumour cells, and has the advantage of combining metabolic activity and imaging characteristics. PET-CT scanners are able to detect small pancreatic neoplasms up to 7 mm in diameter, and to diagnose metastatic disease in about 40%.35 PET is becoming a more common method of measuring tumour response to treatment and may help predict prognosis. However, FDG-PET is not accurate in pancreatic disease due to its reliance on normal glucose haemostasis. The combination of PET-CT has a sensitivity of 92%, and is superior to either modality alone.36,37
Pancreatic adenocarcinoma
Introduction
Epidemiology
Risk factors (see Box 15.1)
Diet and alcohol
Occupation
Past medical history
Hereditary pancreatic cancer
Precursor lesions
Presentation
Investigation
Markers
Diagnosis
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Pancreatic adenocarcinoma