3
Staging and assessment of hepatobiliary malignancies
Colorectal liver metastases
Transabdominal ultrasound
Ultrasound has a diagnostic sensitivity of 36–61% for detecting lesions measuring 1–2 cm, even when performed by experienced radiologists.1 It is very useful in guiding fine-needle aspiration (FNA) to confirm unresectability by cytopathology. However, FNA has a risk of seeding metastases in up to10% of patients and a risk of a false-negative result, which does not justify its use in candidates for potentially curative therapy.2
A meta-analysis of the performance of ultrasound for CRLMs found a pooled sensitivity of 63% (95% confidence interval (CI) 56–70%; five studies) with a specificity of 97.6% (95% CI 95.6–99.5%; four studies).3
Computed tomography and magnetic resonance imaging
Nowadays, CT is the most commonly used imaging modality for staging and follow-up of these patients. Current multidetector scanners allow for multiplanar reformatting with the same resolution as the original axial images. This may improve detection and characterisation of small lesions.3 Multiplanar reformatting also enables the demonstration of hepatic arterial, venous and portal anatomy, which can be useful in preoperative planning.4 CT allows for accurate volumetric assessment of tumour size, liver volume to be resected and future liver remnant volume.5
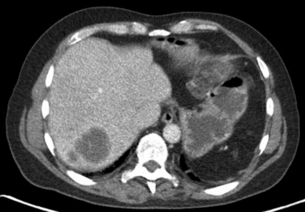
A multiphase contrast-enhanced CT should be performed to stage liver metastases. The study should include an unenhanced, arterial, portovenous and delayed phase. CRLMs may show an enhancing rim on arterial phase imaging and are hypodense on portovenous phase imaging (Fig. 3.1). A slice thickness of 2–4 mm is recommended for axial viewing.
A meta-analysis on the performance of CT found a pooled sensitivity of 74.8% (95% CI 71.2–78.3%; 12 studies) and specificity of 95.6% (95% CI 93.4–97.8%; seven studies).3
The standard MRI protocol includes T2-weighted, unenhanced T1-weighted and dynamic contrast-enhanced T1-weighted sequences. T2-weighted images are used for the detection and characterisation of lesions (haemangioma, cyst).6,7
A meta-analysis on the performance of MRI found a pooled sensitivity of 81.1% (95% CI 76.0–86.1%; five studies) and specificity of 97.2% (95% CI 94.5–99.9%; two studies).3 On per lesion analysis, MRI appeared to be the modality showing higher sensitivities across individual studies compared to CT. Pooled data showed comparable results, with MRI having a combined sensitivity of 88% and accuracy of 87% compared to CT with sensitivity of 74% and accuracy of 78%. Therefore, most centres choose the imaging technique of their preference.
Subgroup analyses of these studies showed that MRI has better sensitivity at picking up the smaller lesions < 1 cm compared to CT and also positron emission tomography (PET)-CT.8 The majority of lesions missed by CT and PET-CT were micrometastases of < 1 cm. This meta-analysis concludes that MRI is preferred as a first-line modality for evaluating the liver. It has high overall sensitivity and specificity estimates and can accurately depict lesions smaller than 10 mm. Secondly, it provides excellent anatomical details.
Positron emission tomography
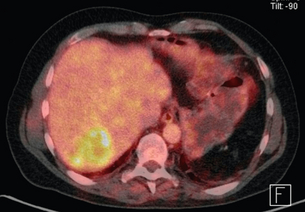
With the PET-CT combination it is possible to produce fusion images with high-resolution anatomical localisation of CT together with the functional data of FDG-PET. This combination of scanning characteristics is becoming more widely available (Fig. 3.2).
Kinkel et al.9 performed a meta-analysis and concluded that, at equivalent specificity, PET-CT is more sensitive than ultrasound (US), CT and MRI for the detection of hepatic metastases from gastrointestinal cancers.
Mainenti et al.11 found that PET-CT showed a trend to perform better than the other modalities. The pooled estimates of a meta-analysis for PET-CT was 93.8% (95% CI 90.0–97.7; six studies) and specificity per patient was 98.7% (95% CI 97.2–100%; six studies).3
Diagnostic laparoscopy and laparoscopic ultrasound
Several centres favour the selective use of laparoscopy. Jarnagin et al.13 have described a clinical risk score (CRS) that predicted survival after hepatic resection and was also suitable in identifying high-risk patients most likely to benefit from laparoscopy. In a study of 103 patients, occult unresectable disease was found in 12% of patients with a low score versus 42% of patients with a high score.13,17 Similarly, in a series of 200 patients, a detection rate of only 6% was reported in patients with the lowest CRS, whereas this was 75% for the highest CRS scores.14 Another study demonstrated a yield of 50% in a selected group of patients, while in the group of 49 patients selected for direct surgical exploration, 46 patients (94%) were eventually resected.15
Staging and assesment of resectability
The significance of resection margins of less than 10 mm is not clear.19 The results of this meta-analysis demonstrate that a negative margin of 1 cm or more confers a survival advantage when compared with subcentimeter negative margins. The management of synchronous liver metastases and extrahepatic abnormalities (particularly the hilar lymph nodes) is also unclear. There is a huge variation in techniques for resection (anatomical vs. non-anatomical, vena cava reconstruction, etc.). Although promising, clear results on the effectiveness of combination treatments with (neo)adjuvant chemotherapy are still lacking.
Modern multidisciplinary consensus defines resectable CRLM simply as tumours that can be resected completely, leaving an adequate liver remnant.21 Before surgical exploration, most surgeons would require that there was no radiographic evidence of involvement of the hepatic artery, major bile ducts, main portal vein, or coeliac/para-aortic lymph nodes, and an adequate predicted functional hepatic remnant.22
In the presence of a limited number of lesions in the lung, liver resection is generally performed first. After radical resection of liver metastases, resection of the lung can follow. There is no reason to refrain from liver resection in a patient with advanced age who has good general (cardiopulmonary) fitness. Risk scoring systems (such as the clinical risk score23 and others24–26) to predict which patients with CRLMs are most likely to benefit from resection are of uncertain clinical utility, particularly since most patients undergo different neoadjuvant chemotherapy regimens. Some studies have shown that the risk scoring system needs refinement27,28 and that the outcome in patients treated by neoadjuvant chemotherapy was not predicted by the traditional clinical scoring system, but rather by response to chemotherapy as evaluated by CT and PET-CT.29
Hepatocellular carcinoma
Transabdominal ultrasound
Transabdominal ultrasound is recommended by the European Associasion for the Study of the Liver (EASL), the American Association for the Study of Liver Disease (AASLC) and the Asian Pacific Association for the Study of Liver Disease (APASL) as a surveillance tool for patients at high risk of developing HCC. Meta-regression analysis has demonstrated a significantly higher sensitivity for early HCC with US every 6 months than with annual surveillance.30
Computed tomography and magnetic resonance imaging
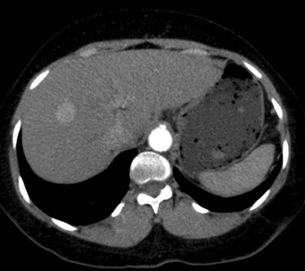
Multiphase contrast-enhanced CT or MRI should be performed when evaluating HCC. The hallmark of HCC on CT and MRI is strong arterial enhancement (Fig. 3.3) and washout of contrast agent in the delayed phase (Fig. 3.4).31 This feature in particular enables differentiation from intrahepatic cholangiocarcinoma, which shows delayed enhancement.32
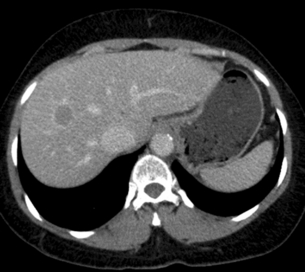
Figure 3.4 CT image of same patient with HCC in the liver showing venous washout of the lesion with enhancing rim.
HCC arising in a non-cirrhotic liver is often large, due to its long asymptomatic course and late presentation.33 This contrasts with the multifocal tumours more commonly found in patients with chronic liver disease, who are often in surveillance programmes. Large HCCs may demonstrate a number of characteristic appearances on CT, which make differentiation from other causes of focal liver masses relatively straightforward. These features include mosaic appearance, with fibrous septa separating areas of variable attenuation which represent internal regions of haemorrhage, necrosis, fatty degeneration and fibrosis. The fibrous capsule has a low attenuation on unenhanced images and enhances on the portal venous phase.
A number of benign lesions, including haemangiomas, focal confluent fibrosis, peliosis, benign regenerative nodules and transient hepatic attenuation difference, can mimic small HCC lesions on CT.34
On MRI, HCCs can have a variable appearance on unenhanced T1-weighted images and typically show an increased signal on T2-weighted images.35 Following gadolinium administration, HCC demonstrates characteristic early enhancement on arterial phase imaging36 and washout on delayed images, resulting in a hypo-intense lesion compared to the surrounding parenchyma.37
Diagnostic laparoscopy and laparoscopic ultrasound
Laparoscopic evaluation can avoid exploratory laparotomy in 45–63% of patients with unresectable disease.38,39 The procedure has been shown to be accurate in assessing the presence of advanced intrahepatic disease and the quality of the liver remnant, but it was less sensitive in determining the extent of local invasion, especially in large (> 10 cm) tumours. Another study clearly reported the value of LUS in detecting small HCCs; 134 new nodules were visualised with this technique in 64 of 186 (34%) patients in whom 28 nodules (in 23 patients) were histologically diagnosed as HCC.40 Of these 23 patients, 18 were diagnosed as having a solitary HCC before laparoscopy. Similarly, new lesions of histologically proven HCC were found in 22% of patients.41 Even when preoperative staging includes a CT, LUS was superior in detecting additional tumours.42 Ultrasound confirmed all 201 tumours seen on CT and detected 21 additional tumours (9.5%) in 11 patients (20%).
Staging and assesment of resectability
Tumour staging to select a treatment regimen is complicated by the fact that many of the staging systems (including the TNM staging system of the American Joint Committee on Cancer (AJCC) – see Table 3.1) are based on surgical findings, while surgery is applicable to only about 5% of these patients. Others recommend the Barcelona Clinic Liver Cancer (BCLC) staging system.43 The Barcelona algorithm does not address the value of resection for some subgroups of patients with HCC who may potentially benefit from this approach, including some patients defined as being ‘early stage’ (with a single tumour size > 2 cm or multiple nodules) and ‘intermediate stage’ (patients who have multinodular tumours but a good performance status). The Milano/Mazzaferro criteria for liver transplantation for HCC (i.e. solitary tumour ≤ 5 cm or up to three tumours all ≤ 3 cm) are widely accepted.44 However, according to the Barcelona algorithm, only selected patients with three nodules ≤ 3 cm (those without ‘associated diseases’) should also undergo liver transplantation.43 The American Hepato-Pancreato-Biliary Association (AHPBA)/American Joint Commission on Cancer (AJCC) Consensus Conference on HCC in 2002 concluded that no single staging system could be used to accurately stage patients across the spectrum of HCCs.44,45
Table 3.1
TNM staging for hepatocellular cancer


Note: cTNM is the clinical classification, pTNM is the pathological classification.
*The fibrosis score as defined by Ishak is recommended because of its prognostic value in overall survival. This scoring system uses a 0–6 scale.
†Data from AJCC sixth edition.
Modified from the AJCC Cancer Staging Manual, seventh edition (2010), published by Springer, New York.
The ideal patient for resection has a solitary HCC confined to the liver that shows no radiographic evidence of invasion of the hepatic vasculature, no evidence of portal hypertension and well-preserved hepatic function. According to the current TNM/UICC staging system for HCC (Table 3.1), most consider stage IIIB, IIIC, IVA or IVB disease to be incurable by resection. However, hepatic resection for stage IIIB, IIIC and IVA disease may be considered in some centres of excellence as clinical benefits, and long-term survival can be achieved in a selected minority of patients.
Pancreatic and periampullary carcinoma
Most pancreatic tumours are located in the head of the pancreas (60–65%), while 20% are present in the body and 10% in the tail region. Unfortunately only a minority (5–20%) of pancreatic tumours are resectable. Tumours in the pancreatic head often present earlier due to compression of the adjacent common bile duct causing obstructive jaundice. Therefore these tumours are often smaller at time of presentation and more often resectable. These smaller tumours (< 2 cm) without liver metastases have better 5-year survival.46 Hence, by accurately detecting and staging smaller tumours, imaging has the potential to influence survival.
Transabdominal ultrasound
This is usually the first screening examination of the abdomen in patients with obstructive jaundice. It is a useful diagnostic modality with a reasonable sensitivity (> 90%) for detecting bile duct obstruction, determining the level of the obstruction (e.g. intrahepatic, proximal or distal) and identifying the presence of gallstone disease.47 Transabdominal ultrasound is cost-effective, widely available but highly operator dependent and often limited by the inability to adequately visualise the pancreas due to bowel gas interference. The goal of ultrasound is therefore to primarily establish a differential diagnosis among the various causes of obstructive jaundice and in identifying liver metastases. Ultrasound is highly sensitive in detecting gallbladder stones (> 90%),48 but this sensitivity drops to 50–75% for the detection of bile duct stones.49 On ultrasound, a pancreatic tumour appears as a hypoechoic (poorly reflective) mass. A tell-tale sign suggestive of malignant obstruction is the combined presence of a dilated common bile duct and pancreatic duct (double duct sign).
Additionally, transabdominal ultrasound has a high sensitivity for detecting liver metastases and ascites, and an FNA can be performed. Sensitivity for liver lesions depends on the size of the lesion and is > 90% for lesions larger than 2 cm, 60% for lesions of 1–2 cm and 20% for lesions < 1 cm in diameter.1
Computed tomography and magnetic resonance imaging
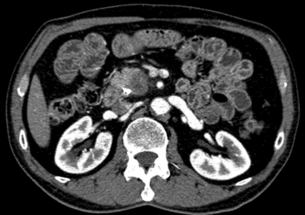
The arterial phase optimally demonstrates the arterial vessels (superior mesenteric artery, coeliac artery and hepatic artery) and their relation to the pancreatic tumour (Fig. 3.5). The portal venous phase demonstrates the portal and superior mesenteric veins and the junction (the confluence) at the pancreatic neck (Fig. 3.6). The parenchymal phase is between these two phases and demonstrates pancreatic adenocarinoma as a hypovascular tumour compared to the rest of the parenchyma.
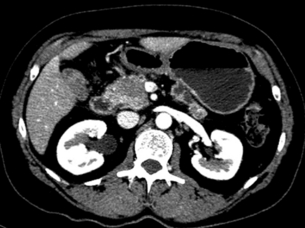
Figure 3.6 CT image of patient with pancreatic head mass with portal vein contact of approximately 90 degrees.
Tumours in the head of the pancreas usually cause dilatation of the common bile duct (CBD). The double duct sign appears when both the CBD and the pancreatic duct are dilated. Smooth dilatation is more suggestive of malignancy, whereas irregular or beaded dilatation is more suggestive of pancreatitis. Tumours that extend beyond the contours of the pancreas with infiltration of the peripancreatic fat are seen as blurring of the normal dark peripancreatic fat. The sensitivity and specificity of CT for detection of a pancreatic head mass were 91% and 85%, respectively, versus 84% and 82% for MRI in a pooled meta-analysis.50 The meta-analysis found that CT was significantly better than MRI and US.
CT has been reported to have a positive predictive value of 100%, negative predictive value of 56% and overall accuracy of 70% for unresectable pancreatic carcinoma.51
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree