Fig. 10.1
Patient positioning with both arms tucked to the side. The surgeon stands to the left and the assistant to the right of the patient
Surgical Technique
Exploration and Exposure
We recommend diagnostic laparoscopy to evaluate for clinically occult metastatic disease. This may be performed via open technique Hasson port placed in-line with the planned incision. The peritoneal cavity is then systematically inspected for ascites, peritoneal or liver nodules, or other evidence of metastatic disease. If any suspicious lesions are identified, an additional 5-mm port is placed to facilitate laparoscopic biopsy. Any suspicious lesions are sent for frozen section examination before proceeding.
Once absence of metastatic disease is confirmed, a laparotomy incision is made. We prefer a midline vertical incision from the xiphoid process to just above or just below the umbilicus, depending on the length of the patient’s torso. A horizontal Chevron-type incision is also feasible. The two have been shown to be equivalent in terms of postoperative pain and postoperative hernia formation. The midline incision preserves the integrity of the rectus muscles and has been shown to result in decreased wound infections [6]. The pre-peritoneal fatty tissue is excised to facilitate exposure and the falciform ligament is divided between clamps and ties or with an energy-sealing device. The liver and the celiac trunk are palpated for liver masses or abnormal celiac lymph nodes, respectively. A fixed retractor is then assembled. We prefer the Thompson retractor (Thompson Surgical Instruments, Traverse City, MI).
Dissection of the Inferior Border of Pancreas
The stomach is grasped and elevated by the assistant, whereas the transverse colon is grasped and placed inferiorly by the surgeon. The lesser sac is entered by dividing the greater omentum between the gastroepiploic vessels and the transverse colon. Entry into the lesser sac is confirmed by visualization of the posterior wall of the stomach. This opening should be extended along the greater curvature of the stomach, following the gastroepiploic vessels, but should be stopped prior to reaching the short gastric vessels. The opening is then similarly extended toward the distal stomach and the origin of the gastroepiploic vessels. It is important for the assistant to continue holding the stomach straight up in the air to expose this area. The avascular attachments between the posterior wall of the stomach and the pancreas are divided with electrocautery. The right gastroepiploic vein is identified and is carefully followed down to the superior mesenteric vein (SMV). The right gastroepiploic vein often shares a common trunk with the right colic vein, known as the gastrocolic trunk or Henle’s trunk (Fig. 10.2). The trunk may include the middle colic vein as a trifurcation. Careful dissection should be performed along the left side of the venous trunk until the SMV is reached. The right gastroepiploic vein can be ligated and divided at this point to prevent tearing or this step can be performed later once final determination of resectability has been made.
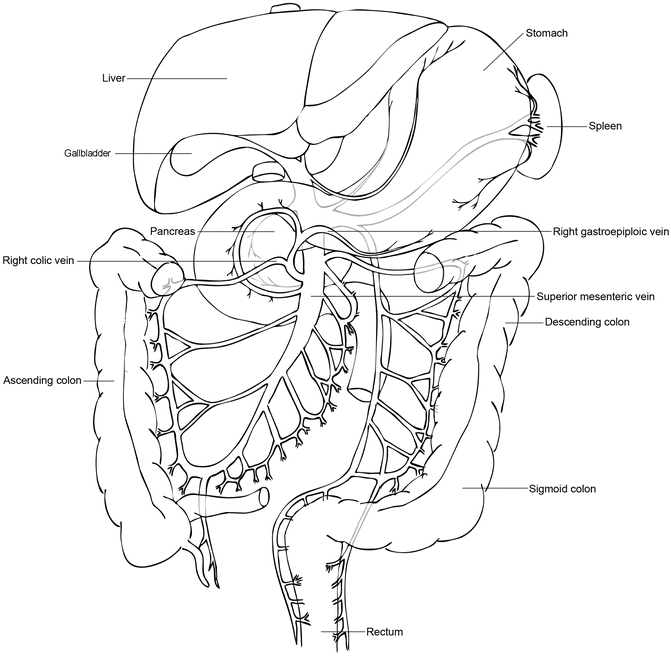

Fig. 10.2
Illustration of the venous confluence of the right gastroepiploic and right colic veins. These veins drain into the superior mesenteric vein as a common trunk, which may also include the middle colic vein. These veins are easily torn with excessive traction and are a common site of bleeding in pancreaticoduodenectomy
Once the SMV is identified, it is critical to determine that there is an adequate length of vein uninvolved by tumor to proceed safely with resection. This typically requires a minimum of 2–3 cm of infra-pancreatic SMV to allow for placement of a vascular clamp to permit vascular reconstruction. It is acceptable to begin a tunnel between the anterior surface of the SMV and the neck of the pancreas, however significant dissection at this stage is strongly discouraged as vascular control of inflow and outflow has not been obtained and exposure is severely limited at this stage of the operation. Furthermore, in most cases, high quality preoperative imaging should clearly define the status of the plane behind the pancreatic neck. If an attempt to dissect this plane is made at this stage, any resistance during dissection should lead to this maneuver being immediately aborted. If small-volume bleeding is encountered, it is best controlled by applying pressure for some time, as there is no exposure to control it directly. If there is high-volume bleeding that will not stop with pressure, vascular control of the portal vein above and SMV below must be obtained while holding direct pressure. If necessary, the neck of the pancreas should be divided to expose the underlying source of bleeding. Next, we typically prefer to identify the superior mesenteric artery (SMA) just posteromedial to the SMV. This will help define the right lateral border of the SMA and later facilitate the uncinate dissection.
Mobilization of the Hepatic Flexure
The transverse colon is grasped and elevated. Any attachments to the gallbladder are divided. The hepatic flexure attachments are divided with an energy sealant device exposing Gerota’s fascia of the right kidney and the proximal duodenum. There is a discrete, avascular tissue plane between the colon mesentery and Gerota’s fascia. This plane should be widely opened so that the colon can be packed caudally, maximizing exposure of the duodenum.
Kocher Maneuver
The duodenum is gently rolled to the patient’s left side by the surgeon, placing the underlying retroperitoneal attachments on tension. These attachments within an avascular plane are then divided with electrocautery, staying close to the edge of the duodenum. Care should be taken to avoid injury to the retroperitoneal structures in this region, including the right gonadal vessels, the right renal vein, and the inferior vena cava. This dissection should be continued under the head of the pancreas until the left renal vein and ligament of Treitz are reached. Tumor adherence to the inferior vena cava or aorta or abnormal lymphadenopathy in this region signifies advanced unredectable disease.
Dissection of the Porta Hepatis
Hepatic Artery
Attention is then turned to the supra-pyloric region. The gastrohepatic ligament is opened and the stomach is again grasped and elevated. This maneuver tents up the right gastric artery making the location of this typically small vessel apparent. The artery and surrounding tissue is divided at the left lateral aspect of the porta hepatis with an energy sealant device.
Next, the hepatic artery and associated hepatic artery lymph node are identified along the superior border of the pancreas. The node may be difficult to appreciate in obese patients, when it is covered by adipose tissue that must be opened to identify the plane between the node and the pancreatic parenchyma. This fatty tissue should be opened carefully, layer-by-layer until the anatomy is apparent. The lymph node is then removed and should be sent for frozen section analysis, since the finding of metastatic disease will impact the decision to proceed.
Removal of the hepatic artery lymph node provides exposure of the gastroduodenal artery (GDA) and common hepatic artery (CHA). The CHA should be dissected circumferentially to fully visualize its course and the GDA takeoff. The GDA is dissected circumferentially, ideally for a distance of 1–2 cm. The GDA should be gently occluded with a vessel loop or small vascular clamp and the proper hepatic artery should then be palpated to ensure that it still has a strong pulse. If occlusion of the GDA diminishes the pulse in the proper hepatic artery, this indicates narrowing of the celiac axis or variant hepatic arterial anatomy.
Once proper hepatic artery flow is confirmed, the GDA is ligated and divided. We prefer to divide the vessel sharply between two right-angle clamps leaving an approximately 1-cm stump of GDA coming off the CHA to allow for embolization of the stump should post-pancreatectomy hemorrhage develop. The proximal end is secured by 3-0 or 4-0 Prolene sutures followed by 3-0 silk ties. This vessel may also be divided with a vascular stapler if preferred.
Portal Vein
Division of the GDA allows for exposure of the portal vein (PV), which lies posterior to the GDA (Fig. 10.3). The GDA stump is gently retracted upward and the anterior surface of the portal vein is exposed. The space between the anterior surface of the PV and the neck of the pancreas is bluntly dissected with the goal of communicating with the space inferior to the neck of the pancreas. This should be performed very carefully and should again be aborted if any resistance is met. A vessel loop may then be passed around the neck of the pancreas to facilitate relocation of the tunnel later in the procedure.
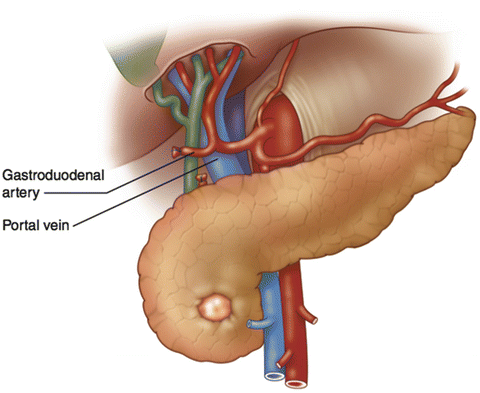

Fig. 10.3
Illustration of the anatomic position of the portal vein behind the gastroduodenal artery. Division of the gastroduodenal artery and elevation of the trunk provide exposure of the anterior surface of the portal vein above the pancreas
Common Bile Duct
Attention is then turned back to the porta hepatis and the gallbladder should be removed if the patient has not had prior cholecystectomy. It is not necessary to keep the gallbladder attached to the remainder of the specimen. The cystic duct is ligated and the gallbladder is passed off as a separate specimen to facilitate exposure. The common bile duct (CBD) is identified and is dissected circumferentially. This should be done by identifying the right lateral aspect of the PV and dissecting from left to right around the CBD. Performing this dissection from right to left can result in injury to the portal vein. There is often nodal and fatty tissue posterior to the bile duct and this area should be carefully palpated for the presence of a replaced or accessory right hepatic artery before it is inadvertently divided. Once the absence of a vessel is confirmed, this tissue can be divided with an energy sealant device, taking care to keep these lymph nodes with the specimen.
The CBD is then sharply divided. We recommend angling the scissors or knife so that the posterior wall of the CBD is left slightly longer than the anterior wall to facilitate creation of the hepaticojejunostomy during the reconstruction. There is often bleeding from the lateral longitudinal arteries of the bile duct. This can be controlled with electrocautery or with placement of a bulldog clamp on the CBD. The latter method also prevents spillage of bile during the remainder of the procedure. If a biliary stent was placed preoperatively, it is removed at this point and should be sent for culture. The culture results may be useful should the patient develop a surgical site infection postoperatively. At this point, a portion of the bile duct may be sent for frozen section analysis. The distal bile duct is typically oversewn with a silk suture to facilitate hemostasis.
Division of the Stomach
Attention is next turned to the stomach. We often perform a standard pancreaticoduodenectomy including a small portion of the antrum and pylorus with the specimen. Multiple randomized, controlled trials have confirmed no difference in outcomes with standard versus pylorus-preserving techniques. Any remaining gastrohepatic ligament attachments are cleared for about 3–4 cm proximal to the pylorus. A target area on the greater curvature side of the stomach is selected and is similarly cleared of any remaining omental attachments. The stomach is then divided with a linear stapler. After assuring hemostasis on the staple line, the proximal stomach can then be packed into the left upper quadrant.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








