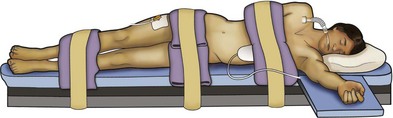
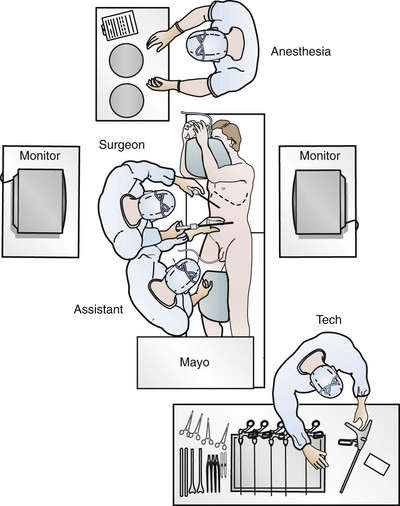
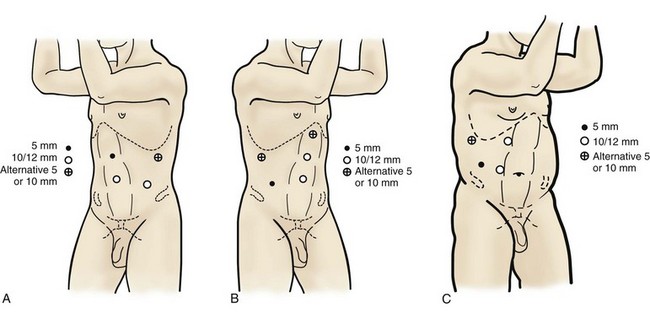
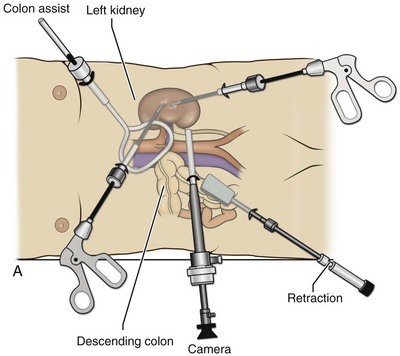
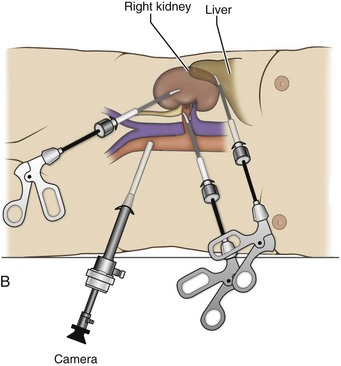
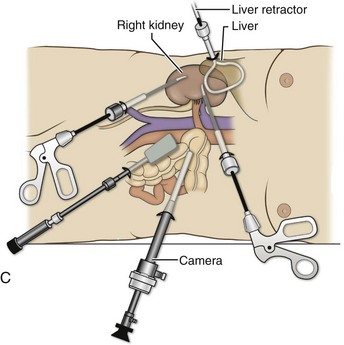
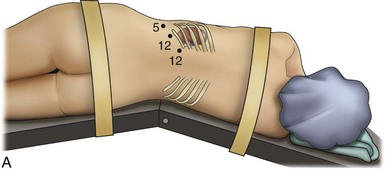
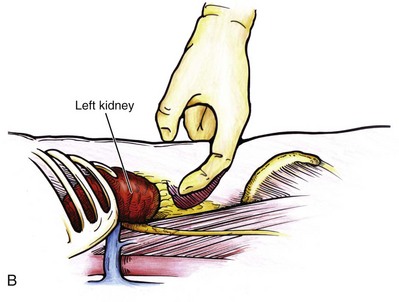
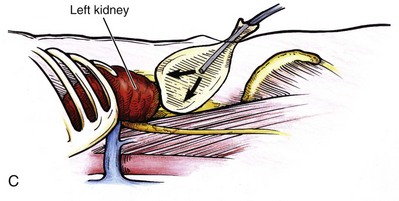
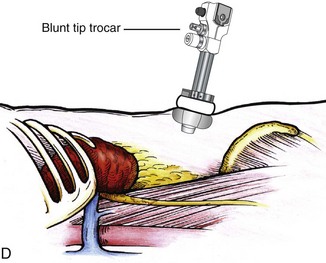
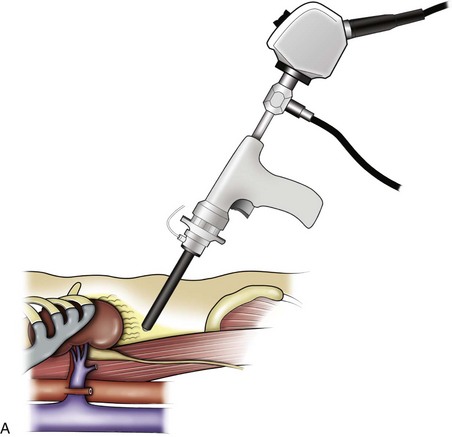
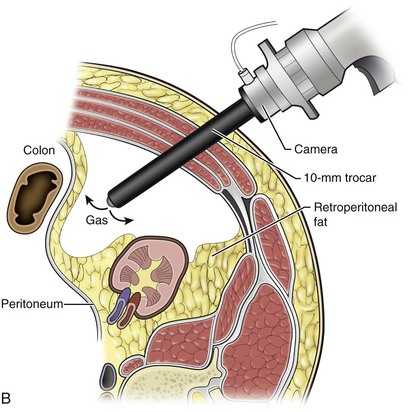
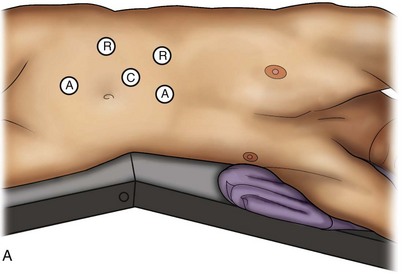
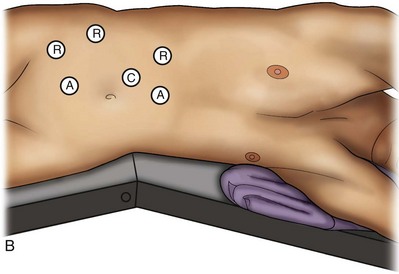
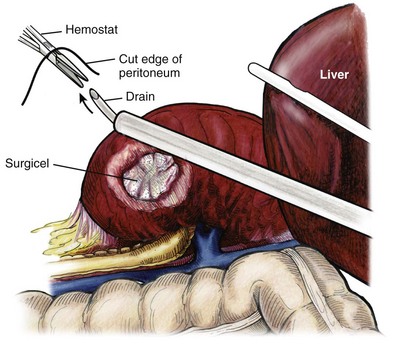
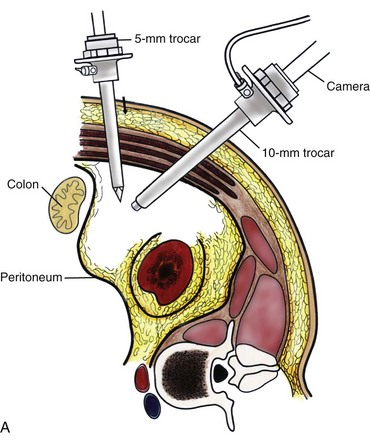
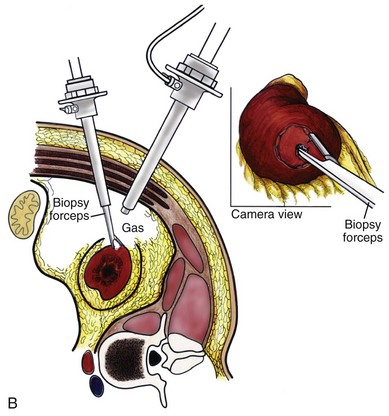
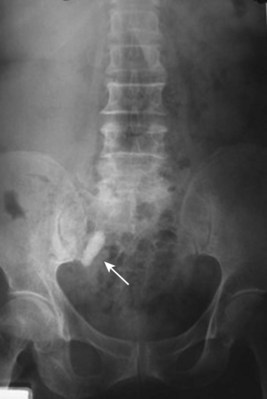
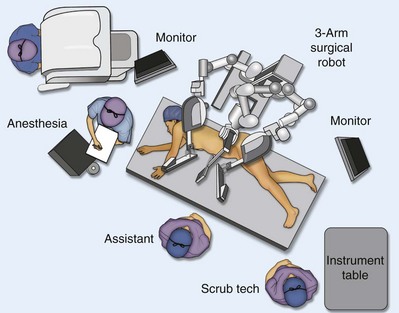
Louis R. Kavoussi, MD, MBA, Michael J. Schwartz, MD, Inderbir S. Gill, MD, MCh Surgical treatment is central to addressing many urologic conditions affecting the kidneys. Decades of experience have demonstrated the efficacy and durability of operative excision for malignancies, as well as reconstruction for obstruction. Unfortunately, traditional open surgical approaches, although extremely effective, are associated with significant postoperative discomfort and recuperation. Studies also demonstrate permanent body surface alterations with flank incisions resulting in significantly larger postoperative surface area and volume changes on the operated flank compared to the noninvolved flank. Indeed, patients report dissatisfaction with the body changes occurring in up to 60% of flank incisions (Chatterjee et al, 2004; Kobayashi et al, 2004). Minimally invasive surgical approaches were born out of the desire to address secondary issues related to surgery, including incisional pain, convalescence, and cosmesis. Initially applied to treat stone disease, advances in video technology and surgical tools have now been used to treat the most complex of renal pathologies, with less morbidity compared to standard operative approaches. Clayman and associates initiated this revolution in renal surgery in 1990 when they introduced the laparoscopic nephrectomy (Clayman et al, 1991). This was performed in an octogenarian with a kidney mass, and although the procedure took over 7 hours, its impact on postoperative recovery was immediately apparent. Subsequently, this approach has been applied to treat every aspect of operative renal disease. Multiple studies have demonstrated that laparoscopic renal surgery provides recuperative as well as cosmetic advantages in contrast to open surgery (Kerbl et al, 1994a; Dunn et al, 2000; Gill et al, 2003). With experience, this is now routinely accomplished without compromise to surgical outcomes. As such, the laparoscopic technique has evolved into the preferred alternative in treating surgical diseases of the kidney. This chapter will discuss indications, present techniques, review results, and outline potential complications of laparoscopy applied to the kidney. Basic laparoscopic principles are thoroughly discussed in Chapter 9. Patient selection and preparation for renal surgery is similar to that for comparable open surgery. A pertinent history and physical are necessary to identify potential issues that could arise during surgery. Prior abdominal, retroperitoneal, or renal surgery is not a contraindication to laparoscopic surgery; however, type and extent of prior abdominal surgery will drive trocar location, as well as choice of transperitoneal or extraperitoneal access (Chen et al, 1998; Cadeddu et al, 1999). In addition, degree of obesity may influence the type of access and location of trocars (Fugita et al, 2004; Hedican et al, 2004; Kapoor et al, 2004). Surgeon experience and available equipment will facilitate or deter use of the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, CA) in treating renal pathology. Informed consent is obtained with a detailed discussion of potential complications including the potential for open conversion. Coagulopathies should be corrected to minimize chances of intraoperative or postoperative bleeding. With the increased use of cardiac stenting, more patients are on chronic antiplatelet therapy. A conversation should occur with the patient’s cardiologist to plan cessation of therapy to minimize risk of perioperative coronary artery thrombosis. If necessary, complex laparoscopic renal procedures have been performed while patients continue aspirin therapy. Uremic patients with prolonged bleeding time may benefit from desmopresssin acetate (desamino-D-arginine vasopressin [DDAVP] 0.3 to 0.4 µg/kg) given intravenously an hour before surgery to improve platelet function (Mannucci et al, 1983). A side effect of this therapy, iatrogenic hyponatremia, has been reported (Pruthi et al, 2002). The majority of laparoscopic renal procedures require a general anesthetic, and a patient’s pulmonary and cardiac function must tolerate this anesthetic approach (Monk and Weldon, 1992). The pneumoperitoneum can impact upon patients with severe cardiopulmonary disease by compromising ventilation and venous return (Arthure, 1970; Hodgson et al, 1970; Nunn 1987; Lew et al, 1992). Patients with chronic pulmonary disease may not be able to compensate for the pneumoperitoneum-induced hypercarbia and may require working at lower pressures, use of helium as an insufflant, or open conversion (Monk and Weldon, 1992; Wolf et al, 1996; Makarov et al, 2007). The use of valveless trocar systems has also been associated with less carbon dioxide absorption (Herati et al, 2009). Obesity is not a contraindication for laparoscopic surgery, but can make retraction and identification of anatomic structures challenging. It is for these reasons that laparoscopy in obese patients is associated with an increased risk of open conversion when compared with nonobese patients (Fazeli-Matin et al, 1999). In addition, although complication rates for laparoscopy in obese patients are higher when compared with laparoscopy in the general population (Mendoza et al, 1996), pulmonary and wound complications are lower with laparoscopy when compared with an open approach (Kapoor et al, 2004; Montgomery et al, 2005). Other factors to account for in the obese population include the increased distance to the operative field, which calls for modifying trocar location and number, as well as the use of longer instrumentation (Doublet and Belair, 2000; Jacobs et al, 2000). Consideration must also be given to the weight of the pannus, which may raise the intra-abdominal pressure and limit working space. The potential for rhabdomyolysis, a rare but devastating complication in the obese as well as very muscular individuals, must also be considered (Troppmann and Perez, 2003). For most transperitoneal renal surgery, the patient is initially positioned supine for intravenous (IV) access, the induction of general anesthesia, and endotracheal intubation. A bladder catheter and orogastric tube are placed for decompression of the bladder and stomach during insufflation, trocar placement, and dissection. Sequential compression stockings are placed for deep venous thrombosis prophylaxis. When performing transperitoneal procedures, including robotic and LESS surgeries, patients are positioned in a 30- to 45-degree flank-up position. Care is taken to pad all pressure points. The patient is secured to the operating table to allow lateral tilting of the table (Fig. 55–1). Tilting the table away from the affected kidney will help move bowel out of the operative field. There is no need to flex the table or use a kidney rest as there is with open surgery. The equipment in the operating room is situated to maximize the use of space and allow all members of the surgical team to view the procedure (Fig. 55–2). During the skin preparation and draping, the entire flank and abdomen are included in case conversion to an open procedure is required. Once a pneumoperitoneum is established, three to five trocars are initially placed to complete the dissection (Fig. 55–3). There are a variety of trocar configurations that are effective for each procedure. A 12-mm trocar is placed lateral to the rectus at the level of the umbilicus; this trocar is used for instrumentation and the passage of sutures, bulldog clamps, or staplers to secure and divide hilar vessels. In short patients, this may be placed in the midline, halfway between the umbilicus and pubis. A 10-mm trocar is placed at the umbilicus for the camera, and a 5- or 10-mm port is inserted in the midline 2 cm below the xiphoid process. In obese patients, all trocar sites are moved laterally (see Fig. 55–3C). Additional trocars for retraction may be needed for visualization or assist with organ entrapment (Fig. 55–4). Using this approach, patients are placed in a full-flank position. Modest table flexion can help increase the distance between the ribs and iliac crest to facilitate trocar placement. An axillary roll is required, as well as care in securing the patient to the bed. Arms may be secured on pillows or a purpose-built armrest. A 15-mm transverse incision is made in the posterior axillary line, midway between the tip of the 12th rib and the iliac crest (Fig. 55–5A). After deepening the dissection downward through the lumbodorsal fascia, the retroperitoneum is entered, and a working space may be developed using blunt dissection with the tip of a finger in the space between the psoas muscle and the kidney (Fig. 55–5B). A simple balloon created from two fingers of a size-8 or -9 glove may then be inserted and filed with CO2 or saline to further develop the retroperitoneal working space (Fig. 55–5C). A blunt tip trocar is then passed through the incision, and the trocar cuff is expanded and cinched to the skin to prevent leakage of CO2 (Fig. 55–5D). An alternative entry approach involves entry with the 0-degree lens and visual obturator through the initial incision (Fig. 55–6A). Entry into the retroperitoneum may be confirmed by the appearance of the characteristic yellow retroperitoneal fat, insufflation is initiated, and blunt dissection using only the laparoscope is performed to develop a working space (Fig. 55–6B). Caution must be used not to enter too anteriorly because inadvertent peritoneal entry or colon injury may occur; entering too posteriorly may result in bleeding from the quadratus lumborum or psoas muscles. Once the working space is established through either approach, pertinent structures may be identified for orientation and additional trocars placed. Typically, a 5-mm trocar is placed just off the tip of the 12th rib, and a 12-mm trocar is placed posteriorly and superiorly relative to the camera port (see Fig. 55–5A). Hand assistance offers a bridge between open surgery and laparoscopy (Nakada et al, 1997). It offers more intuitive assistance from the human hand with dissection and retraction, simultaneously providing tactile feedback to the surgeon. Several manufacturers make devices for this purpose. An incision large enough for the hand must be created and can also be used as an extraction site. This incision site is usually larger than one used for intact extraction during pure laparoscopic surgery. This technique may be advantageous for the novice laparoscopic surgeon and in patients with significant scaring about the kidney, or if a difficult dissection is anticipated. Hand assistance may also be employed in the event of an emergency, such as bleeding, by extending a trocar site and placing a hand port to assist in repair of an injury or vascular control. Indications are identical to those for laparoscopic nephrectomy or laparoscopic partial nephrectomy. The patient is positioned just as for pure laparoscopic kidney surgery. The initial incision is for the hand port that is made through the skin and fascia and into the peritoneal cavity. Location will depend on handedness of the surgeon, operative side, and body habitus of the patient (Figs. 55-7 and 55-8). Care must be taken to avoid making the incision too large, because gas may escape, making the procedure more difficult due to decreased working space. Once the hand-assisted device is placed, the peritoneum is insufflated, and the additional trocars may be placed under direct vision by placing the camera through the hand port. There are some limitations in location of port placement, and the hand may potentially get in the way of visualization or dissection instrumentation. Some leakage of gas may occur about the device, resulting in difficulty in maintaining the pneumoperitoneum. These devices exert 30 to 100 mm Hg of pressure on the arm, which may account for surgeons developing tingling, numbness, or pain in the forearm or hand (Monga et al, 2004). Most authors prefer a flank position with the table modestly flexed (Aron et al, 2008; Deane et al, 2008; Benway et al, 2009). As the robotic procedure requires more trocars than standard laparoscopy, this allows more adequate spacing of instruments and less clashing of the robotic arms. Inclining the table may provide additional space at the back of the patient for the robot and other equipment (Fig. 55–9). Figure 55–9 Operating room configured for left-sided robotic-assisted laparoscopic partial nephrectomy. Robotic trocars plus the additional assistant and camera ports are typically used for the procedure. A port-in-port technique of robotic-assisted partial nephrectomy (RaPN) has also been described, wherein the righthand and lefthand working ports are actually standard 12-mm laparoscopic ports (Aron et al, 2008). The robotic 8-mm ports are inserted through these 12-mm ports to perform RaPN. This arrangement can be advantageous in event of an intraoperative complication or robotic malfunction, wherein emergent conversion to pure laparoscopic surgery is necessary. The robot can be undocked expeditiously and standard LPN performed in the usual fashion without requiring additional time to insert new ports or working through 8-mm robotic ports that preclude passage of the necessary CT-1 or CT-X needles. A three-arm configuration includes four to five total trocars, a 12-mm periumbilical camera port, an 8-mm subcostal robotic trocar in the anterior axillary line, an 8-mm robotic trocar in the posterior axillary line placed above the iliac crest, and one 12-mm assistant trocar in the low midline to allow passage of sutures, bulldog clamps, suction, or retraction. An additional 12-mm subxiphoid trocar may be used, if necessary, for additional retraction or passage of bulldog clamps if the angle is more optimal (Fig. 55–10A). A four-arm configuration involves five to six total trocars, using the same general configuration as the three-arm technique, but shifting the robotic trocars slightly to avoid clashing of the arms (Fig. 55–10B). Laparoendoscopic single-site (LESS) surgery describes laparoscopic techniques that consolidate all ports within a single skin incision, often concealed within the umbilicus (Box et al, 2008). It has evolved to improve upon cosmesis associated with standard laparoscopic surgery and uses commercially available multichannel working ports placed in the umbilicus or below the lower abdominal hair line to minimize postoperative visible scars. Initial clinical cases of virtually every type of extirpative and reconstructive urologic procedures have now been performed by LESS surgery. Because LESS surgery remains a procedure in evolution, there is no widely accepted method of positioning or gaining access. Modified and full-flank positioning have been described and are identical to standard transperitoneal or retroperitoneal kidney surgery, respectively. Once pneumoperitoneum has been established, one may cluster multiple traditional low-profile trocars close together within a single, small extraction incision (Fig. 55–11). Alternatively, newer purpose-specific access devices (Fig. 55–12) may be used in combination with conventional laparoscopic or flexible instrumentation. The devices are secured using preplaced fascial sutures or with an inner and outer ring drawn together with a cylindrical sleeve. Characteristics of single-site access options are described in Table 55–1. (From Tracy CR, Raman JD, Cadeddu JA, Rane A. Laparoendoscopic single-site surgery in urology: where have we been and where are we heading? Nat Clin Pract Urol 2008;5:561–8.) Table 55–1 Access Options for Laparoendoscopic Single Site (LESS) Surgery These approaches are technically challenging and the most difficult of the minimally invasive techniques. To date, no differences in postoperative morbidity or recovery have been reported (Raman et al, 2009). Advances in instrumentation may increase applicability and dissemination into mainstream urologic practice. NOTES involves using a natural orifice to perform the entire operation. In the gastrointestinal (GI) and surgical literature, the mouth, vagina, and rectum have been used to remove organs such as the appendix and gall bladder (Rao et al, 2006; Zorron et al, 2007; Palanivelu et al, 2008). In urology, to date experience with pure NOTES surgery in humans is limited (Kaouk et al, 2009). Several authors have reported hybrid approaches with NOTES and standard laparoscopic techniques, using the vagina as an access and extraction site for nephrectomy (Branco et al, 2008; Alcaraz et al, 2010; Sotelo et al, 2010). Laparoscopic simple nephrectomy is indicated in the treatment of most benign renal diseases. Renovascular hypertension that is not correctable with medication or angiographic repair may be managed with a simple nephrectomy. Patients with chronic pain syndromes may benefit from nephrectomy, including symptomatic acquired renal cystic disease, autosomal dominant polycystic kidney disease (ADPKD), chronic hydronephosis not amenable to surgical repair, loin pain/hematuria syndrome. Chronic infectious processes that are recalcitrant to antibiotic therapy may also be approached laparoscopically, including chronic pyelonephritis, xanthogranulomatous pyelonephritis (XGP), and renal tuberculosis. These conditions are associated with a higher rate of conversion to open surgery due to perinephric inflammatory changes and loss of tissue planes (Gupta et al, 1997; Bercowsky et al, 1999). In some cases, subcapsular nephrectomy may be necessary to complete the procedure. Hand assistance may also be of benefit and avoid open conversion in cases with dense scar tissue formation or inflammatory reaction (Rosoff et al, 2006). Successful retroperitoneal laparoscopic simple nephrectomy has also been reported in 30 of 31 patients with nonfunctioning kidneys secondary to tuberculosis (Lee et al, 2002). Other benign conditions that may call for nephrectomy include patients with a multicystic dysplastic kidney or a symptomatic failed renal transplant. Again, in this later case, a subcapsular dissection may be required. For a left nephrectomy and all renal surgery, the line of Toldt is incised from below the lower pole of the kidney inferiorly to above the spleen superiorly (Fig. 55–13). The inferior limit of this incision may be extended inferiorly, if the bowel does not move sufficiently. The lienocolic ligament should be incised to allow the spleen to fall medially along with the pancreas and the colon (see Fig. 55–13). Care must be taken to avoid injuring the diaphragm with this maneuver. The thin colorenal attachments are incised and the colon is swept medially (Fig. 55–14). Care should be taken to avoid making a hole in the mesentery. Mesenteric fat has a brighter hue of yellow compared with the retroperitoneal or Gerotas fat, which allows for identification of the correct plane of dissection. If the operative field is not adequately visualized, a paddle retractor may be placed in an additional lower midline trocar to aid in retracting the colon, pancreas, and spleen medially (see Fig. 55–4A). Blunt and sharp dissection is necessary to move these structures off the anterior surface of the kidney and hilum. During a right-sided nephrectomy, the peritoneal incision is carried medially and parallel to the lateral border of the venacava and duodenum. A lateral (anterior axillary line) or high midline port may be needed to retract the liver anteriorly (see Fig 55–4B and C). Care must be taken to avoid thermal injury to the duodenum and gall bladder during incision of the peritoneum. Medial traction on the colon reveals colorenal attachments that must be divided to complete the colon dissection. Again, a low midline retractor may be helpful for visualization. A Kocher maneuver may be required to fully expose the medial portion of the kidney and the connective tissue overlying the renal hilum (Fig. 55–15). Once the colon has been adequately mobilized, the psoas muscle and tendon should be identified. Following this structure medially, the gonadal vessels are usually first encountered. These should be swept medially and the ureter is usually located just deep to these vessels. Peristalsis of the ureter can help differentiate between these two structures. Once identified, the ureter is elevated and followed proximally to the lower pole of the kidney. The ureter is not divided at this time, because it can be used to help elevate the kidney (Fig. 55–16). The tissue posterior to the ureter and lower pole of the kidney is swept anteriorly to further expose the psoas muscle. Care should be taken to try and stay above the psoas fascia to minimize postoperative thigh numbness. The instrument in the subxyphoid trocar is used to slide under the kidney all the way to the sidewall. This allows the surgeon to lift the kidney and place medial lymphatic and vascular attachments on stretch. Safe dissection of the renal hilum requires medial retraction of the colon and bowel by gravity or an additional retractor, as well as anterior retraction of the kidney, lifting it out of the renal fossa. With the ureter and lower pole of the kidney elevated, vessels entering the renal hilum can be identified and bluntly dissected using the tip of the irrigator-aspirator. Firm elevation of the lower pole assists in identification and dissection of the renal hilar vessels (Fig. 55–17). This is accomplished by gently placing the lateral grasper under the ureter and kidney until it abuts the abdominal sidewall. It is important to be sure that the grasper is against the muscle and not into the renal parenchyma. A gentle, layer-by-layer dissection is performed with the irrigator-aspirator until the renal vein is uncovered. There is usually an anterior bundle of connective tissue that needs to be incised in order to fully expose and visualize the vein. Gonadal, lumbar, and accessory venous branches can be clipped and divided as necessary. By clearing off inferior attachments and lymphatics, one can identify the renal artery. If the irrigator-aspirator tip is not precise enough for meticulous dissection, a hook electrode can be used to dissect the lymphatic vessels free of the vein and artery. With an endovascular gastrointestinal anastomosis (GIA) stapler, the artery is divided first, followed by the vein (Fig. 55–18). We prefer to use staples in this area because three rows of staples will be left on the vessel stump and are unlikely to be dislodged during subsequent dissection. In some instances clips may be needed. We prefer to use at least five clips on the patient side, when possible. Once the hilar vessels have been divided, the dissection continues posteriorly and superiorly to the upper pole. The adrenal gland is preserved in cases of simple nephrectomy by staying close to the upper pole (Fig. 55–19). This is accomplished by incising the Gerota fascia anteriorly, just above the hilum. Gerota fat is then gently peeled off circumferentially above the upper pole of the kidney. At this point during the dissection, it may be necessary to clip and transect the ureter. This allows the kidney to be rotated anteriorly above the liver (right) or spleen (left) to facilitate incision of the uppermost attachments under direct vision. In cases of extreme fibrosis, a subcapsular nephrectomy can be performed once the artery and vein have been controlled (Moore et al, 1998). Long, blunt instruments, such as the closed stapler or the 10-mm Ligasure Atlas (Valleylab, Boulder, CO) are particularly well suited for reaching and freeing the upper pole attachments. The kidney can be removed intact or through morcellation. When morcellation is performed, the specimen should be placed into a sturdy entrapment sac (Urban et al, 1993). This minimizes the risk of rupture during mechanical morcellation of the tissue (Landman et al, 2000; Pautler et al, 2002). Using ring forceps and a Kocher clamp, the kidney and collecting system can be morcellated and removed in small pieces (Fig. 55–20). Alternatively, the kidney can be removed intact through an incision after placement into a sac (Fig. 55–21). The kidney can be worked out of an extended trocar site or Pfannenstiel incision. For trocar sites greater than 5 mm, it is helpful to place the closure sutures before removing the kidney. Once the sutures are placed, trocars can be reinserted for kidney entrapment and removal. The postoperative results of laparoscopic nephrectomy are comparable to that of open surgery, with much less pain and shorter convalescence. Postoperative pain control requirements are approximately four times less than with traditional open incisions. Hospital stays have been decreased by 50%, and the time to full convalescence has been reported to be markedly less than with open removal. In early series, the mean operative times were greater than 300 minutes. However, with advances in techniques, experience, and equipment, current operative times have decreased dramatically (Kerbl et al, 1994a, 1994b; Nicol et al, 1994; Parra et al, 1995; Baba et al, 1996; Rassweiler et al, 1998). Renal cysts are extremely common and are present in more than one third of patients more than 50 years old (Laucks and McLachlan, 1981; Carrim et al, 2003). They rarely require surgical intervention, but indications include cyst-associated pain, infection, or obstruction (Hoenig et al, 1997; Wolf, 1998; Roberts et al, 2001; Doumas et al, 2004; Camargo et al, 2005). The increased use of cross-sectional imaging has also increased the detection of indeterminate renal cystic lesions and complex renal cysts, bringing an increased number to the attention of urologists. Classification schema have been developed to help clinicians make determinations regarding management, the most popular being the Bosniak system (Table 55–2) (Israel and Bosniak, 2005). Although these schema can be extremely useful, they are not diagnostic, and surgery may be required in some cases to exclude malignancy. Table 55–2 Renal Cyst Classification Based on Updated Bosniak Criteria From Israel GM, Bosniak MA. An update of the Bosniak renal cyst classification system. Urology 2005;66:484–8. First-line therapy and diagnosis of symptomatic renal cysts often involves percutaneous image-guided needle aspiration, with or without the use of a sclerosing agent, to prevent recurrence. If symptoms temporarily resolve and recur when fluid reaccumulates, this increases the likelihood that surgical treatment will be successful in resolving the pain (Rané et al, 2004). Caution should be exercised in use of cyst aspiration and sclerosing agents in peripelvic cysts, because fibrosis may occur (Wehle and Grabstald, 1986; Hulbert et al, 1988; Santiago et al, 1998; McDougall, 2000). In addition to causing pain, cysts may compress the renal parenchyma or other adjacent organs, cause ureteral obstruction and obstructive uropathy, spontaneously bleed, cause hypertension, or become infected. Laparoscopic decortication or unroofing may be used to treat these cysts, which are typically simple in character (Fig. 55–22). Cysts with complex appearance, such as thickened septa, calcification, or enhancement (Bosniak class III-IV), may be explored and sampled laparoscopically to rule out renal cell carcinoma due to their increased risk of harboring malignancy (Cloix et al, 1996; Santiago et al, 1998). Options include cryoablation, enucleation, partial nephrectomy, or radical nephrectomy. If there is any question of cyst proximity to the collecting system, cystoscopy and placement of an open-ended ureteral catheter may be performed to ensure the integrity of the collecting system after cyst excision. A subset of patients with autosomal dominant polycystic kidney disease (ADPKD) may develop cyst-associated pain. Laparoscopic cyst decortication, marsupialization, or unroofing can be of benefit to these patients by offering a minimally invasive treatment that is successful in relieving pain in up to 83% of cases (Lifson et al, 1998; Dunn et al, 2001; Lee et al, 2003). In patients with end-stage renal disease, bilateral synchronous laparoscopic nephrectomy may be performed in patients with enlarged, symptomatic, or infected kidneys (Gill et al, 2001; Rehman et al, 2001; Bendavid et al, 2004; Desai et al, 2008). Because an incision will be required to remove kidneys that are often quite enlarged, a hand port may be placed in the midline and used bilaterally to facilitate the dissection (Rehman et al, 2001; Jenkins et al, 2002). Depending on cyst location, a transperitoneal or retroperitoneal approach may be used as previously described. Intraoperative ultrasonography may be used to identify the cyst or cysts in question. It is usually easier to dissect out the cyst wall before evacuating fluid. The wall of the cyst can then be grasped and excised, cutting along the junction between the cyst wall and the renal parenchyma (Fig. 55–23). If suspicious lesions are noted in the base, biopsies may be taken using the 5-mm biopsy forceps. If no evidence of malignancy is seen, the remaining cyst wall may be fulgarated with either electrocautery or the argon beam coagulator. Care should be taken when ablating the surface, because inadvertent entry into the collecting system can easily occur (Cherullo et al, 1999). Moreover, these surfaces can be friable and prone to significant bleeding. Packing with hemostatic agents or suturing may be necessary. One should have a low level of suspicion for entry into the collecting system and, as such, a low threshold for leaving a drain (Fig. 55–24). If malignancy is noted, extirpative surgery or cryoablation may be used to treat the remainder of the lesion. When treating central or perihilar cysts, it may not be feasible to remove a large portion of the cyst wall. In these cases, it is helpful to place a pedicle of autologous fat into the defect to act as a wick (Nieh and Bihrle, 1993). Laparoscopic treatment of symptomatic renal cysts has been found to be effective in both decompression and pain control. A study evaluating treatment durability at a mean follow-up of 26 months demonstrated a 100% pain-free rate in patients treated for a solitary symptomatic renal cyst (Lifson et al, 1998). In a separate study with a mean follow-up of 60 months, 80% to 90% of patients have complete resolution of pain after laparoscopic cyst decortication. In patients with ADPKD, additional benefits of cyst decortication have been noted, including decreased blood pressure (Dunn et al, 2001; Lee et al, 2003). Recurrence of pain in this group is higher than with simple cysts, and durability is moderate (Brown et al, 1996). No significant changes in renal function were noted postoperatively. Patients undergoing surgery for cystic renal disease are inherently a heterogeneous group, made up of patients with simple renal cysts, complex or indeterminate cysts, and ADPKD. Interpretation of incidence reports of renal cell carcinoma in these series must take this fact into account. That said, the reported incidence of renal cell carcinoma in cystic lesions is between 3% and 20% (Rubenstein et al, 1993; Lifson et al, 1998; Roberts et al, 2001; Limb et al, 2002). Histologic information is a key component in making treatment decisions and for prognosis in patients with proteinuria or unexplained renal insufficiency (Morel-Maroger, 1982; Gault and Muehrcke, 1983; Manaligod and Pirani, 1985). Although the modality of choice is typically ultrasound-guided percutaneous renal biopsy, laparoscopic biopsy may be preferred in certain situations, such as failed percutaneous biopsy, renal anatomic anomalies, risk of bleeding complications, morbid obesity, multiple renal cysts, or solitary kidneys. The patient is placed in the full-flank position with the table flexed to increase working space between the costal margin and the iliac crest. Retroperitoneal access is preferred and obtained using techniques described later in this chapter. Renal biopsy is accomplished with the use of two trocars. With blunt dissection, Gerota fascia is opened, and the lower pole of the kidney is exposed (Fig. 55–25A). In obese patients, intraoperative ultrasonography may be required to localize the kidney when copious retroperitoneal or perinephric fat is present. A 5-mm biopsy forceps is used to take samples of cortical tissue; hemostasis is achieved with the argon beam coagulator; and adjunctive hemostatic measures are used as necessary (Fig. 55–25B). A multi-institutional series reporting on outcomes of laparoscopic renal biopsy in 74 patients over 9 years showed a mean operative time of 123 minutes, mean estimated blood loss of 67 cc, and discharge within 24 to 48 hours unless other preexisting medical conditions required a longer stay (Shetye et al, 2003). Tissue obtained from 96% of patients was adequate for diagnosis, with a complication rate of 13.5%. The authors concluded that laparoscopic renal biopsy may be performed safely with a high success rate, and, that as experience grows, the complication rate and operative duration would likely decrease. In another series of 17 patients, using balloon dilatation to create the working space demonstrated 100% success in obtaining renal tissue adequate for diagnosis. Mean operative time was 35 minutes (excluding anesthesia time), complication rate was 11%, and 15 of 17 patients were discharged within 24 hours. In published series of laparoscopic renal biopsy, hemorrhage is the most common complication. Caution should be used in resumption of anticoagulation in patients who require it postoperatively. If signs or symptoms of postoperative anemia or hypovolemia occur, a low threshold should be used to evaluate the patient with computed tomography (CT). Advances in stone therapy have made pyelolithotomy, nephrolithotomy, and ureterolithotomy almost obsolete. However, a select subgroup of stone patients will benefit from these approaches. Depending on local demographics and incidence of stone disease, the indications for laparoscopic stone extraction may vary but primarily include patients with renal stones who are undergoing simultaneous laparoscopic pyeloplasty (Ball et al, 2004; Stein et al, 2008). In these patients, a flexible cystoscope may be passed through one of the trocars to remove stones. A laparoscopic grasper may also be used to remove stones, but patients should always undergo a complete nephroscopy to ensure stone clearance. Other patients who may benefit include those with unusual anatomy (pelvic kidney) (Chang and Dretler, 1996; Harmon et al, 1996; Hoenig et al, 1997), stones resistant to fragmentation (Jordan et al, 1997), and failed extracorporeal shock wave lithotripsy (ESWL), ureteroscopy, or percutaneous procedures. Treating stone disease in ectopic kidneys can be particularly challenging. Ectopic kidneys frequently have anomalous vasculature, and therefore access into the collecting system carries a higher risk of bleeding. Laparoscopy may be used to assist in the placement of percutaneous access to minimize the risk of bleeding complications (Eshghi et al, 1985; Tóth et al, 1993; Troxel et al, 2002). A retrospective comparative study of 12 patients who underwent percutaneous nephrolithotomy (PCNL), and 16 patients who underwent laparoscopic retroperitoneal pyelolithotomy, revealed no benefit to the laparoscopic approach. Laparoscopy was also associated with longer operative time, longer recovery, inferior cosmesis, and required more advanced skills (Goel and Hemal, 2003). The authors concluded that PCNL should remain the gold standard for treating large renal stones, unless the patient is undergoing surgery for concomitant pathology. Robotic approaches to stone disease have also been described for the treatment of staghorn calculi, with or without simultaneous pyeloplasty. In a study of 12 patients undergoing robotic extended pyelolithotomy, 11 were stone free after surgery as assessed by postoperative CT scan. Mean operative time was 158 minutes, with mean estimated blood loss of 100 cc (Badani et al, 2006). Laparoscopic anatrophic nephrolithotomy has been reported in animal models and small case series (Kaouk et al, 2003; Simforoosh et al, 2008). Initial reports are encouraging, but, to date, renal functional outcomes in humans have not been evaluated with nuclear renal scans. Further studies are required to assess the feasibility of this procedure. Large or impacted ureteral stones may be managed laparoscopically with ureterolithotomy (Fig. 55–26). In a series of 93 patients undergoing this procedure, successful stone removal was achieved in 85 patients, with eight failures due to inability to locate the stone (Gaur et al, 2002). Mean operative time was reported as 80 minutes, and in many of the patients, the ureter was left open, leading to shorter operative time but more complications with prolonged urinary leakage. Other authors have also reported this approach for impacted or large ureteral stones (Hemal et al, 2003). In this series of 31 patients, success was reported in all cases, no stent was used, and closure of the ureterolithotomy in all cases resulted in a prolonged urinary leakage rate of 6.5% at 48 hours. Renal ptosis, although rare, is a real cause of chronic flank or upper abdominal pain. The precise origin of symptoms is unknown, but is likely secondary to either transient ischemia or urinary obstruction (Moss, 1997). It is usually defined as the descent of the kidney by more than two vertebral bodies, but before a definitive diagnosis is made, objective proof that is associated with pain should be obtained. The typical patient with a ptotic kidney is a young, thin female who complains of pain while in an upright position. Supine and erect intravenous pyelography (IVP) can be used for diagnosis, with the finding of interest being descent of the symptomatic kidney by two vertebral bodies (Fig. 55–27). Nuclear imaging may also quantify obstruction of blood flow or drainage in the upright position. Color Doppler sonography in both the supine and upright positions can also be used to evaluate differential blood flow. If a ptotic kidney is present, the expected finding would be diminished blood flow while in the erect position. Before surgical repair, obstruction, decreased blood flow, or significant descent correlating with pain should be documented. Figure 55–27 Intravenous pyelogram demonstrating bilateral ptotic kidneys in the supine (A) and standing (B) positions. (From El-Moula MG, Izaki H, Kishimoto T, et al. Laparoscopic nephropexy. J Laparoendosc Adv Surg Tech A 2008;18:230–6.) Surgical repair is performed using either a standard transperitoneal or retroperitoneal approach to fully mobilize the affected kidney and expose the fascia overlying the psoas and quadratus lumborum muscles (Chueh et al, 2002; Matsui et al, 2004) (Fig. 55–28). Beginning at the upper pole, interrupted sutures are placed to secure the lateral edge of the renal capsule to the fascia overlying the muscle (Fig. 55–29). Sutures may also be placed between the anterior renal capsule and the parietal peritoneum for additional support. Using sutures with preplaced Lapra-Ty clips (Ethicon Endosurgery, Cincinnati, OH), the initial pass is made through the fascia or peritoneum, and a second clip is placed on the suture after the pass through the kidney. An alternative technique describes the use of tension-free vaginal tape to secure the kidney (Hübner et al, 2004). By placing the needles such that the tape is passed around the lower pole of the kidney and out through the abdominal wall, the kidney is secured. Sometimes this is found in conjunction with a ureteropelvic junction obstruction that should be adressed simultaneously (Boylu et al, 2009). A retrospective study of 30 patients undergoing laparoscopic nephropexy with a median follow-up of 5.9 years demonstrated improvement in all patients, 11 with complete relief of symptoms (Plas et al, 2001). Significant improvement in renal function was measured by renal scan in 9 of 10 patients undergoing the study postoperatively. Two patients developed a recurrent ptotic kidney with greater than 5 cm of descent documented by IVP. An additional study of 48 patients, with a median follow-up of just more than 8 years, revealed that 94% of patients had no signs of significant ptosis on postoperative imaging, and 91% of patients had improvement in their pain symptoms (Gözen et al, 2008). Mean operative time was 95 minutes, and mean estimated blood loss was 50 cc. Patient satisfaction was high, and the authors concluded that laparoscopic nephropexy provided a minimally invasive approach to these patients with good long-term clinical outcomes. Patients with symptomatic calyceal diverticula containing stones can be managed laparoscopy. ESWL and ureteroscopy may be employed, but due to infundibular stenosis, stone clearance rates are low (Jones et al, 1991; Pang et al, 1992; Stream et al, 1992). The principles of treatment include the removal of stones and widening of the infundibular stenosis to prevent urine stasis or ablation of the diverticula cavity. In the past, symptomatic calyceal diverticula have been managed with partial nephrectomy, with marsupialization and fulgaration of the diverticulum, and occasionally simple nephrectomy. More recently, percutaneous treatment has been used, but stones or symptoms may recur if the cavity is not fully ablated (Donnellan et al, 1999). A laparoscopic approach may be indicated with large, peripheral diverticula, or for centrally located diverticula with proximity to the renal hilum. Published reports on laparoscopic calyceal diverticulectomy are limited to small series and case reports. Excellent results have been reported by numerous authors, demonstrating the definitive nature of this treatment modality (Miller et al, 2002; Gluckman et al, 1993; Ruckle and Segura et al, 1994; Harewood et al, 1996; Wolf, 2000; Canales and Monga, 2003; Wyler et al, 2005). Probably due to the rarity of this surgery, complications are also rare and have not been reported yet in the literature. Chyluria is caused by lymphatic rupture or fistulous connection into the pyelocalyceal system. It is a rare problem worldwide but is commonly seen in tropical countries when filariasis (Wuchereria bancrofti or Brugia malayi), the most common cause, is endemic (Tandon et al, 2004). Rarely, schistosomiasis may also be a cause of chyluria, and nonparasitic chyluria is rare. Other reported etiologies include tuberculosis (Wilson and White, 1976), idiopathic lymphorenal fistula (Eisner et al, 2009), prior surgery (Kim and Joudi, 2009), pregnancy (Onyeije et al, 1997), thoracic duct obstruction (Garrido et al, 1995), mesenteric adenitis (Cohen et al, 1984), renal vasculitis (El-Reshaid et al, 1998), and metanephric adenoma (McNeil et al, 2008). Patients typically present with milky white urine and may have nephrotic range proteinuria. Initial evaluation includes urinalysis and culture, urine for chyle, and complete blood count to check for eosinophila. Evaluation to localize the fistula may include cystoscopy with retrograde pyelogram, CT, magnetic resonance imaging (MRI), or lymphangiography (Fig. 55–30). (From Eisner BH, Tanrikut C, Dahl DM. Chyluria secondary to lymphorenal fistula. Kidney Int 2009;76:126.) Chyluria is often self-limited, and many patients may be managed conservatively. In filariasis-associated cases, this involves treatment with a course of diethylcarbamazine (DEC) in combination with a low-fat diet (Tandon et al, 2004). Retrograde instillation of silver nitrate or povidone iodine into the collecting system as a sclerosing agent has also been described, and these agents are commonly used as first-line treatment with comparable success rates of approximately 80% (Dalela et al, 2004a; Goel et al, 2004). When conservative management fails or if cases are particularly severe, surgical intervention is undertaken. Nephrolysis involves the complete mobilization of the kidney and skeletonization of the renal hilar vessels and upper ureter with ligation of the lymphatic channels. The procedure may be performed laparoscopically by either a transperitoneal or retroperitoneal approach and ensures complete lymphatic dissociation of the affected kidney (Chiu et al, 1995, Gomella et al, 1998). The use of an omental wrap around the hilum has also been described to provide an additional barrier against recurrence (Dalela et al, 2004b). Authors commonly performing this procedure report excellent success rates—approaching 100%. Laparoscopic approaches to malignancy have been performed for over 20 years. The indications for radical nephrectomy are similar to those for open surgery. Kidneys with tumors as large as 25 cm have been successfully removed laparoscopically, and cytoreductive nephrectomy has been performed in patients with metastatic disease (Walther et al, 1999). Moreover, tumors with low- level caval thrombi have also been removed (Martin et al, 2008). Access and trocar placement are similar to what has been described for simple nephrectomy. With larger masses, caval involvement or organ invasion, additional trocars or a hand port may be needed. The procedure for laparoscopic radical nephrectomy (LRN) is essentially identical to laparoscopic simple nephrectomy. The main distinguishing feature is that Gerota fascia is left intact during dissection. The adrenal may be removed en bloc with the kidney when indicated (Fig. 55–31A). Alternatively, this fascia is opened over the upper medial aspect of the kidney when adrenal-sparing nephrectomy is performed (Fig. 55–31B). Suspect lymph nodes may be removed, and a full hilar or retroperitoneal dissection can be carried out if deemed necessary. Excision of part of adjacent muscle or involved organs, such as the pancreas, liver, spleen, and bowel, has also been reported (Molina et al, 2004). Long-term cancer-specific survival data is now widely available from multiple centers around the world that perform laparoscopic radical nephrectomy (Table 55–3). Five- and 10-year outcomes show oncologic equivalence to open radical nephrectomy in treatment of renal cancer. Indeed, LRN has become the standard of care for most renal malignancies. A multi-institutional study from centers performing LRN compared the surgical and disease-specific outcomes between open and laparoscopic radical nephrectomy, with long-term follow-up (Portis et al, 2002). Median follow-up was 54 months, and recurrence-free survival was 91% and 92%, respectively, for the two groups at 5 years. The 5-year cancer-specific survival was 98% for the laparoscopic cohort and 92% for the open cohort. A comparative analysis of 67 patients undergoing LRN, with 54 patients undergoing open radical nephrectomy, evaluated perioperative and oncologic outcomes (Permpongkosol et al, 2005). The LRN group showed a longer mean operative time (256 vs. 193 minutes). However, this finding likely reflects the learning curve for laparoscopy, because the first 34 patients and last 33 patients in the LRN group had a significant operative time difference. Complications occurred in 15% of patients in the LRN and open groups, and blood transfusions were required in 6% and 20% of the patients, respectively. Most importantly, the calculated disease-free survival rates for laparoscopic and open radical nephrectomy were 95% and 89%, respectively, at 10 years. The actuarial survival rates for laparoscopic and open radical nephrectomy were 87% and 75%, respectively, at 10 years. These differences were not found to be statistically significant. Most recently, 10-year oncologic outcomes data following LRN were reported. Recurrence-free, cancer-specific, and overall survival rates were 86%, 92%, and 65%, respectively, at 10 years postnephrectomy (Berger et al, 2009). Of 73 patients undergoing LRN, no patient developed local recurrence, and 6 (8.2%) developed metastatic disease at a mean time to recurrence of 74 months. Although outcomes in this study were not compared with an open radical nephrectomy cohort, results are quite comparable to those for open surgery.
Patient Evaluation and Preparation
Anesthesic Considerations in Laparoscopy
Considerations in Obese Patients
Surgical Approaches and Obtaining Access
Transperitoneal Laparoscopy
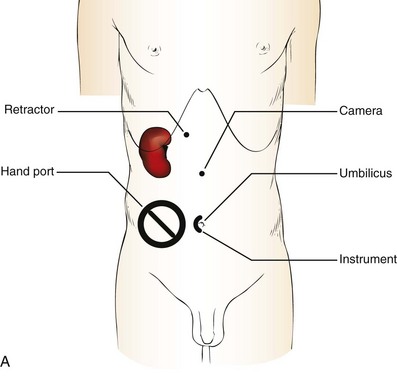
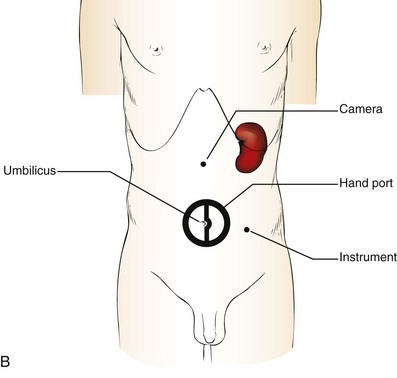
Patient Positioning and Trocar Placement
Retroperitoneal Laparoscopy
Patient Positioning and Trocar Placement
Hand-Assisted Laparoscopy
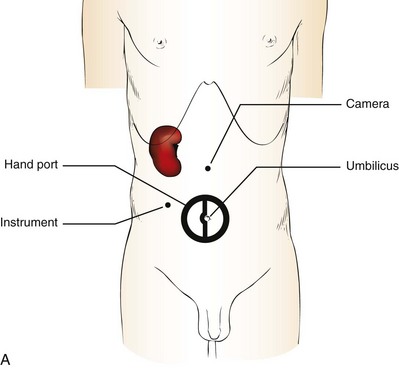
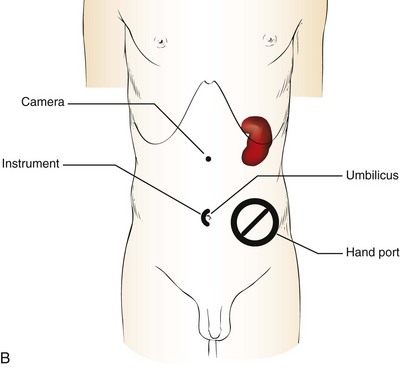
Patient Positioning and Trocar Placement
Robotic-Assisted Laparoscopy
Patient Positioning and Trocar Placement
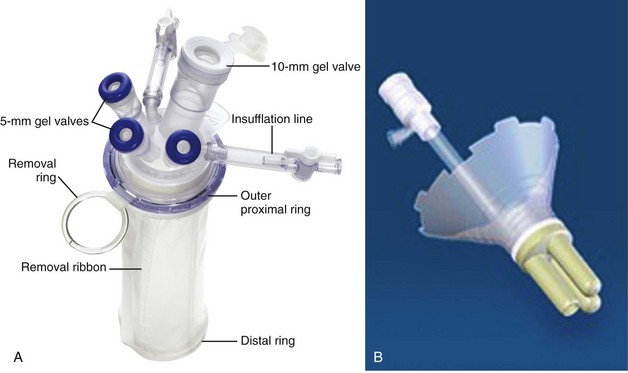
Laparoendoscopic Single-Site Surgery (LESS) and Natural Orifice Transluminal Endoscopic Surgery (NOTES)
Patient Positioning and Trocar Placement
ACCESS TYPE
DESCRIPTION
Keyhole
Use of three closely approximated periumbilical trocars placed side by side in a single skin incision or three separate incisions
No additional device required
Typically used with articulating camera and specialized instrumentation
Insufflation through trocar
TriPort (Advanced Surgical Concepts, Bray, Ireland)
Open or closed access, may be used with multiple incision sizes (5-mm to hand-assisted incision)
Anchored by inner (intra-abdominal) and outer rings drawn together with cylindrical sleeve
3-Port (one 12-mm and two 5-mm) configuration or 4-port (two 12-mm and two 5-mm) configuration available (Quadport)
Insufflation through valve housing
Uni-X (Pnavel Systems, Cleveland, OH)
Open access technique, requires 2-cm fascial incision
Anchored with preplaced fascial sutures
Single port encompassing three 5-mm access ports
Typically used with articulating camera and specialized instrumentation
Insufflation through valve housing
Gelport (Applied Medical, Rancho Santa Margarita, CA)
Open access technique, requires 2.5- to 5-cm fascial incision
Anchored by inner and outer rings drawn together with cylindrical sleeve
Can accommodate all trocar sizes
May allow for wider spacing of trocars
Insufflation through trocar placed through device
Simple Nephrectomy
Benign Indications
Procedure
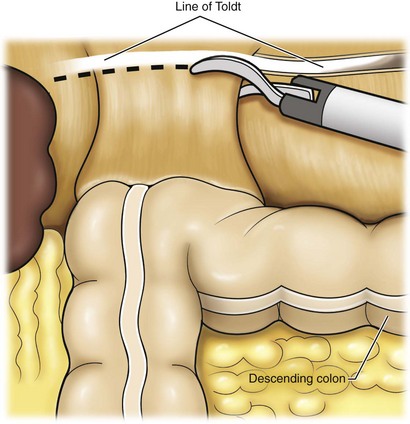
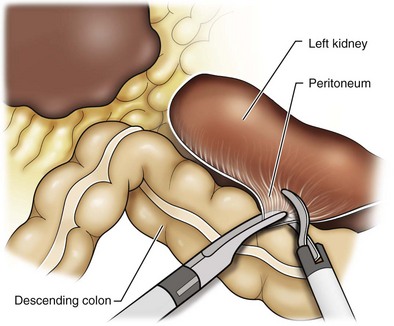
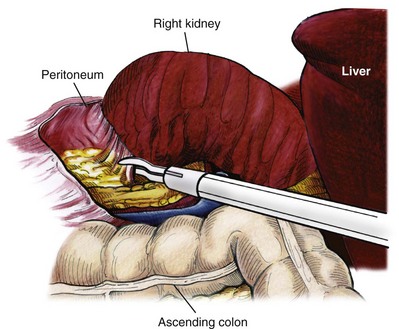
Reflection of the Colon
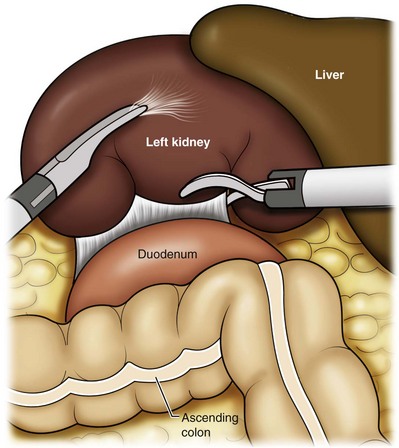
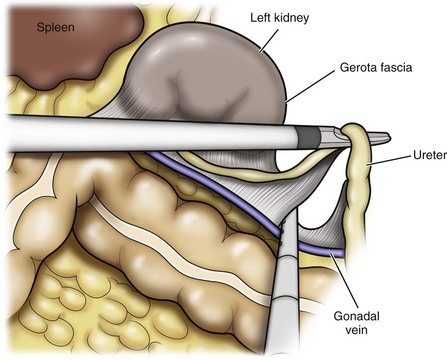
Dissection of the Ureter
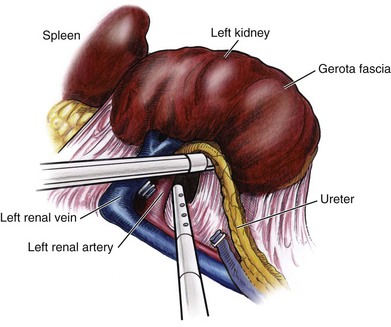
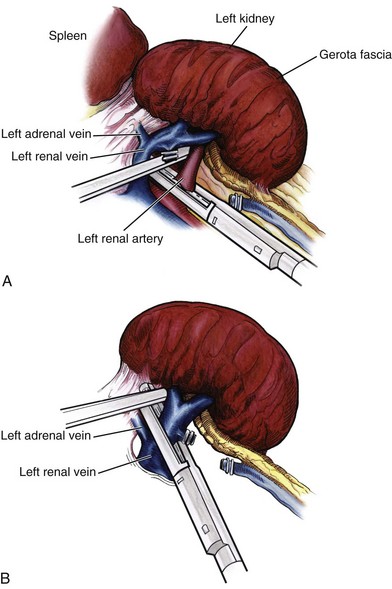
Identification of the Renal Hilum
Securing the Renal Blood Vessels
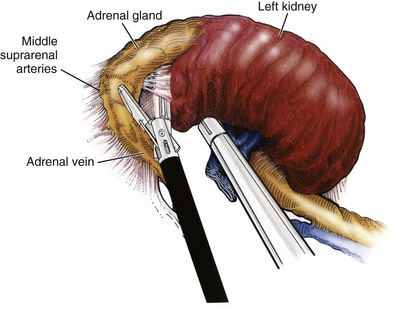
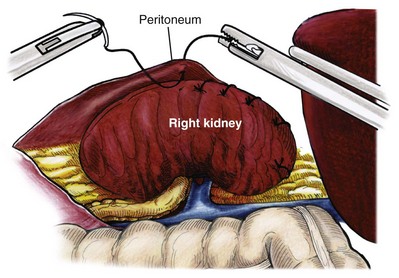
Isolation of the Upper Pole
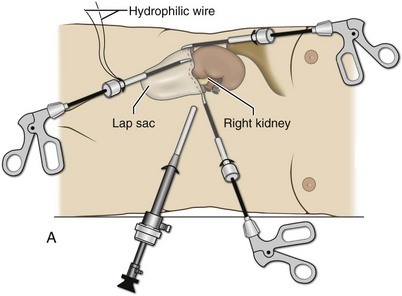
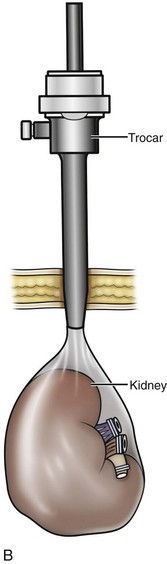
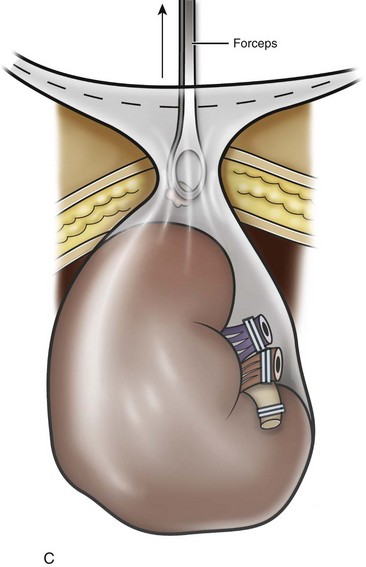
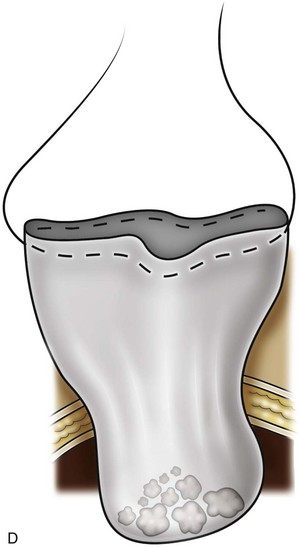
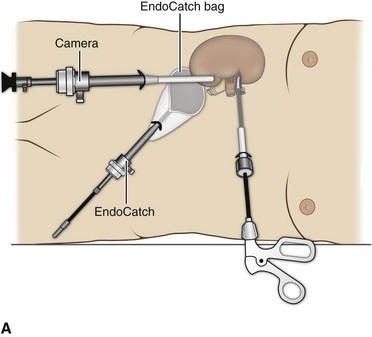
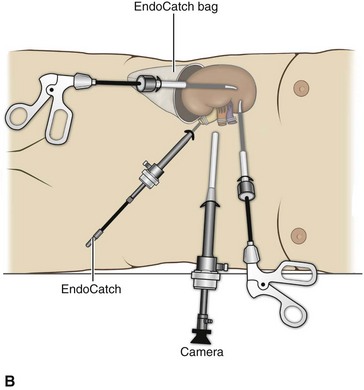
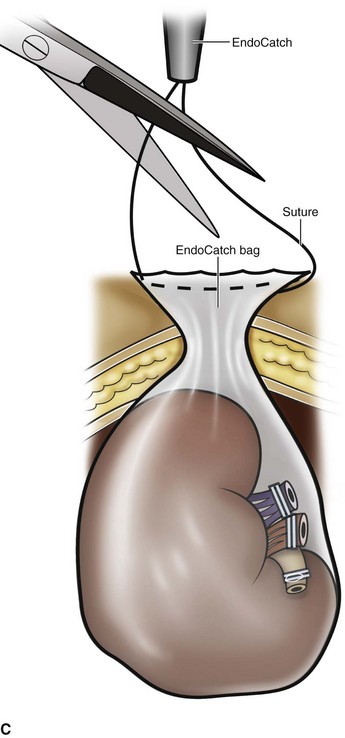
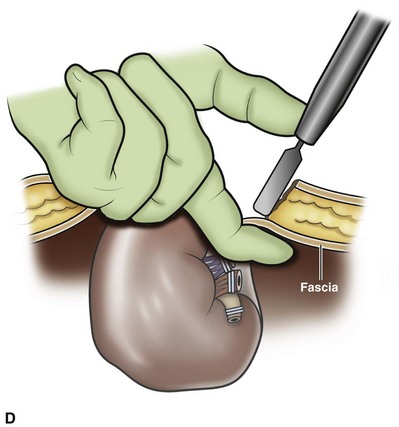
Organ Entrapment and Extraction
Results
Renal Cystic Disease
TYPE
DESCRIPTION
RECOMMENDED MANAGEMENT
I
A benign simple cyst with a hairline thin wall that does not contain septa, calcifications, or solid components. It measures water density and does not enhance.
No follow-up necessary
II
A benign cyst that may contain a few hairline thin septa in which “perceived”* enhancement may be present. Fine calcification or a short segment of slightly thickened calcification may be present in the wall or septa. Uniformly high-attenuation lesions (3 cm) (so-called high-density cysts) that are well marginated and do not enhance are included in this group.
No follow-up necessary
IIF
Cysts that may contain multiple hairline thin septa or minimal smooth thickening of their wall or septa. Perceived enhancement of their septa or wall may be present. Their wall or septa may contain calcification that may be thick and nodular, but no measurable contrast enhancement is present. These lesions are generally well marginated. Totally intrarenal nonenhancing high-attenuation renal lesions >3 cm are also included in this category.
Follow-up imaging required
III
“Indeterminate” cystic masses that have thickened irregular or smooth walls or septa in which measurable enhancement is present. These are surgical lesions, although some will prove to be benign (e.g., hemorrhagic cysts, chronic infected cysts, and multiloculated cystic nephroma), some will be malignant, such as cystic renal cell carcinoma and multiloculated cystic renal cell carcinoma.
Surgical treatment
IV
These are clearly malignant cystic masses that can have all the criteria of category III, but also contain enhancing soft tissue components adjacent to, but independent of, the wall or septum. These lesions include cystic carcinomas and require surgical removal.
Surgical treatment
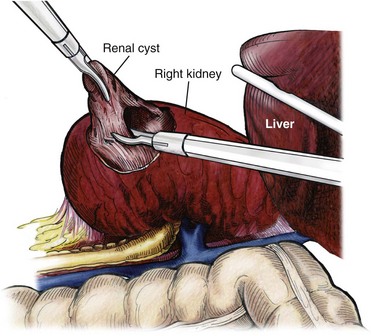
Indications
Procedure
Results
Renal Biopsy for Medical Renal Disease
Procedure
Results
Pyelolithotomy, Nephrolithotomy, and Ureterolithotomy
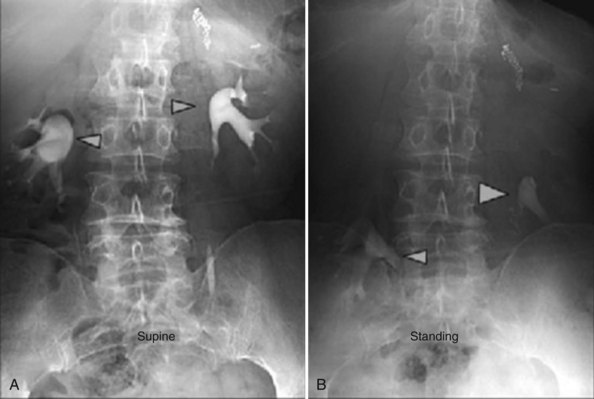
Nephropexy
Procedure
Results
Calyceal Diverticulectomy
Results
Nephrolysis
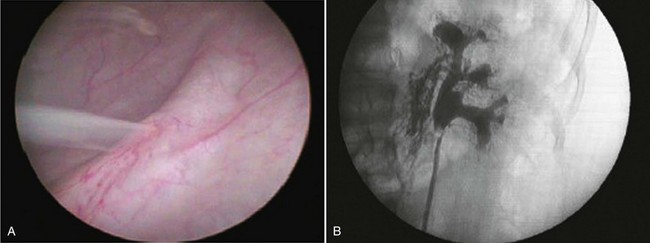
Diagnosis
Treatment
Procedure and Results
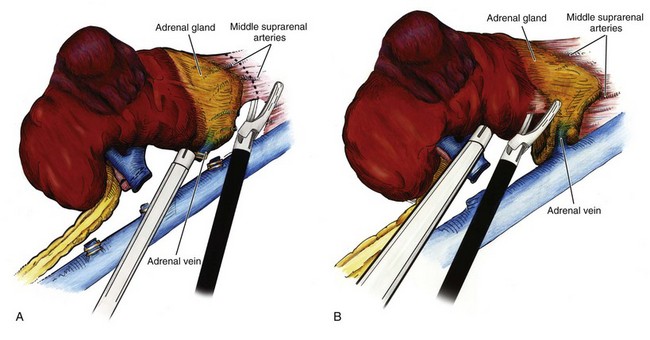
Radical Nephrectomy
Indications for Malignancy
Transperitoneal
Procedure
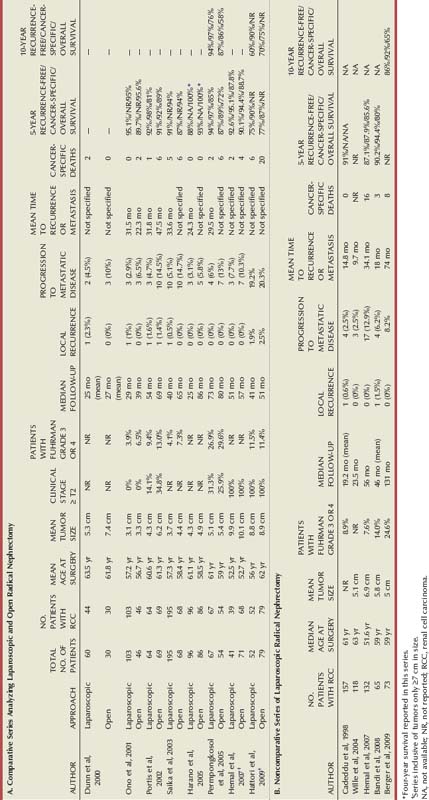
Results
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Laparoscopic Surgery of the Kidney