William R. Mulley, John Kanellis
Evaluation and Preoperative Management of Kidney Transplant Recipient and Donor
Renal transplantation provides superior long-term outcomes compared with dialysis, in both quantity and quality of life, although the benefit gained varies among individuals.1 The improvement in outcomes coupled with the shortage of available organs has led to an expansion of donor criteria and an increasing tendency for centers to accept marginal donor kidneys.2 In this chapter, we review current recommendations for the evaluation and preoperative management of both the kidney transplant donor and the recipient.
Recipient Evaluation
Many units now accept patients who were previously excluded from transplantation, such as those with human immunodeficiency virus (HIV) infection, obesity, and diabetes. This is because of the availability of newer treatment options for some conditions and a greater understanding of the impact of these conditions on patient and graft survival along with changing societal attitudes regarding equality of access to transplantation. Some absolute contraindications to transplantation remain (Table 102-1), including significant current infection or malignant disease, noncompliance or substance abuse, and any condition likely to severely limit life expectancy (<1 to 2 years).3–5
Table 102-1
Contraindications to renal transplantation.
CDC, Complement-dependent cytotoxicity; HIV, human immunodeficiency virus.
| Contraindications to Renal Transplantation | |
| Current Absolute Contraindications to Transplantation | Previous Contraindications to Transplantation* |
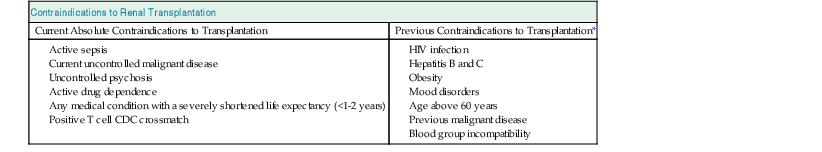
* These conditions are now acceptable under certain circumstances—see text.
Whereas the application of guidelines for transplant suitability may be relatively straightforward for patients with a single comorbidity, it is not as simple for those with multiple medical conditions, who represent a growing group of potential transplant recipients. Determination of suitability in such patients often requires input from specialists in a variety of medical and surgical disciplines along with allied health professionals. The final decision needs to be a joint one between clinician and patient after full and open discussion of the likely risks and benefits followed by regular reassessment of suitability while the patient awaits transplantation.
A summary of guidelines published by national and international transplantation associations3–5 is presented in Box 102-1. Some of the important areas to consider in evaluating the transplant recipient are discussed here.
Cardiovascular Disease
Cardiovascular disease is a significant problem in patients with end-stage renal disease (ESRD) and a major cause of death in transplant recipients. Hence, cardiovascular evaluation is critical in the evaluation of the transplant recipient.
Coronary Heart Disease and Left Ventricular Dysfunction
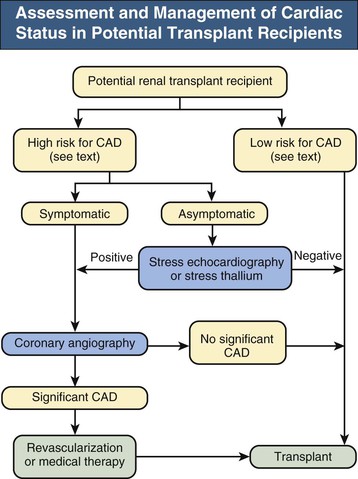
Chronic kidney disease (CKD) itself is a major risk factor for coronary heart disease. However, the vascular lesion, clinical features, and response to treatment may be quite different in patients with ESRD compared with the normal population. The role of pretransplantation screening and intervention for coronary heart disease is controversial, making definite recommendations difficult. However, given the high incidence of cardiac events in the peritransplantation period and its major contribution to post-transplantation mortality, we favor aggressive screening and intervention in at-risk patients while avoiding unnecessary tests and procedures in low-risk candidates. Patients may be stratified into risk groups on the basis of history and examination, resting electrocardiography, and chest radiography. Further screening is unnecessary in asymptomatic patients without risk factors because of a very low incidence of coronary events.6 Further investigation is recommended, however, in patients with abnormal test results or significant risk factors, such as previous cardiac ischemic events, diabetes, smoking, age older than 50 years, hypertension, prolonged dialysis duration (>2 years), or a family history of coronary heart disease.3–5
Symptomatic patients should proceed directly to coronary angiography; noninvasive functional testing should be used to screen for the need for angiography in asymptomatic patients.7 Exercise echocardiography or myocardial perfusion scintigraphy is preferred; however, pharmacologically driven testing may be necessary if exercise is not possible. Whereas a normal result does not exclude coronary heart disease, both testing modalities have negative predictive values for myocardial infarction or cardiac death in excess of 96% in patients with renal failure.8 If significant coronary heart disease is identified, treatment before transplantation should be instituted. Treatment consists of medical management including aggressive risk factor modification, angioplasty and stenting or coronary artery bypass grafting in patients with significant stenoses.7 A suggested approach is presented in Figure 102-1.
In patients with clinical or radiologic evidence of left ventricular dysfunction, transthoracic echocardiography should be performed to assess the severity and nature of the dysfunction. A cause should be sought and treated when possible. Severe left ventricular dysfunction may improve significantly after transplantation; therefore it is not in itself an absolute contraindication to transplantation. It is, however, associated with reduced post-transplantation survival; thus in patients with significant comorbidities it does represent a contraindication to transplantation unless, in rare instances, combined heart and kidney transplantation is appropriate.3–5
Cerebrovascular Disease
Patients with a history of recent transient ischemic attack or stroke are at greatest risk of recurrence early after the primary event; and because stroke after transplantation is associated with a high rate of mortality,9 a waiting time of 6 months is recommended. Meanwhile, aggressive risk factor modification should be undertaken to limit the likelihood of further stroke. Routine screening for cerebrovascular disease is not advocated in asymptomatic patients. Evidence suggests a benefit in further investigation of patients with a carotid bruit and treatment with carotid endarterectomy if a significant stenosis is found.10 Patients with polycystic kidney disease at high risk of cerebral aneurysm rupture, such as those with a previous personal or family history of cerebral hemorrhage, should be screened for cerebral aneurysm before transplantation; however, there is no conclusive evidence to support doing so in those without such risk factors. Risk of cerebral aneurysm in polycystic kidney disease is discussed further in Chapter 46.
Peripheral Vascular Disease
Asymptomatic patients with strong femoral and peripheral pulses generally require no further investigation. Patients with diabetes, history of claudication, or reduced pulses require vascular imaging beginning with Doppler ultrasound. Significant disease involving the iliac vessels may make transplantation difficult or impossible and may worsen ischemia in the distal leg. Although it is not an absolute contraindication to transplantation, peripheral vascular disease is associated with increased mortality11 and should be considered in conjunction with the patient’s other comorbidities.
Cancer
Cancer is a major cause of death in renal transplant recipients. Further increases in the incidence of malignant disease are likely with increased graft survival and acceptance of older recipients. The incidence of malignant disease is also increased in transplant recipients compared with the general population.12 However, the effect of transplantation on different types of cancers is not uniform, nor is the effect of different immunosuppressive agents.13 Some cancers, such as nonmelanoma skin cancers (61% to 82% at 20 years) and lymphoma, have a markedly increased incidence in transplant recipients compared with the general population; the incidence of other malignant neoplasms, such as breast and prostate, is not as markedly increased.14 In patients with a previous malignant neoplasm, guidelines have been devised on the basis of the likelihood of recurrence after transplantation (Box 102-2). In general, the longer the cancer-free interval before transplantation, the smaller the recurrence risk. For most malignant neoplasms, a period of 2 to 5 years is recommended.3–5 There are several exceptions. A longer waiting time (≥5 years) is recommended for breast cancer with nodal involvement, melanoma, and colorectal cancer worse than Dukes stage B1; no waiting time is thought necessary for nonmelanocytic skin cancers confined to the skin, in situ cancers of bladder and cervix, focal microscopic low-grade prostate cancer, and small (<7 cm) incidentally discovered and surgically removed renal cell carcinomas. Given the heterogeneity of malignant neoplasms, waiting periods before transplantation need to be individualized, taking into account the patient’s other comorbidities.
Although extensive screening of all potential recipients is not warranted, guidelines appropriate for the general population should be adopted in screening for breast, cervical, prostate, and colorectal cancer. Chest radiography is performed as part of the routine assessment. More comprehensive and targeted evaluation is recommended in patients with a strong family history or suggestive clinical features of malignant disease or conditions associated with an increased risk of malignant disease, such as renal imaging in patients with acquired cystic disease of the kidney for possible renal cell carcinoma.3,4
Infectious Complications
All patients are screened for previous exposure to Epstein-Barr virus and cytomegalovirus (CMV) to assess the risk of infection, either primary or reactivation. This guides the appropriate use of prophylactic antiviral agents. For example, patients who are negative for CMV IgG who receive a kidney from a CMV-positive donor are at the highest risk of infection and may benefit from prolonged prophylaxis compared with the lower-risk CMV-negative donor to a CMV-negative recipient (see Chapter 105). Screening for other infections should be tailored to geographic location; a guide for screening is presented in Table 102-2. Immunization against hepatitis B should be undertaken in all potential recipients. Immunization against encapsulated organisms (pneumococci, Haemophilus influenzae, and meningococci) should also be considered in patients at high risk of antibody-mediated rejection in case rescue therapy—for example, splenectomy or complement inhibition with eculizumab—is required.
Before highly active antiretroviral therapy (HAART), HIV infection was considered an absolute contraindication to transplantation because of very poor patient survival.15 In the HAART era, HIV infection is seen as a manageable chronic disease, and transplantation is offered to HIV-positive patients at many centers that have experience in managing HIV infection and transplantation. Reports of short- to medium-term results suggest excellent patient and graft survival in carefully selected recipients.16 Longer-term studies are under way to further explore these outcomes. Patients with sustained CD4 counts above 200/ml and undetectable HIV viral loads, without an acquired immunodeficiency syndrome (AIDS)–defining illness, can be considered for transplantation. Patients with hepatitis B may be considered for renal transplantation if there is no evidence of active viral replication (hepatitis B virus [HBV] DNA or hepatitis B early antigen [HBeAg] positive), advanced liver disease or cirrhosis (as determined by liver biopsy), or hepatocellular carcinoma.3–5 Immunosuppression can increase HBV replication; hence, treatment before transplantation is indicated in patients with active disease, and although data to support prophylactic antiviral therapy, such as entecavir, after transplantation are scarce, it is commonly practiced while immunosuppression is at its highest (initial 12 to 24 months). Early reports suggest that mortality may be increased in HBV-positive patients after transplantation compared with HBV-negative recipients.17 The significance of these findings is unclear in the current era with more effective treatment options and if only patients with inactive disease undergo transplantation. Frank disclosure of possible risks involved is recommended.3–5
Patients with hepatitis C should be assessed by measurement of hepatitis C virus (HCV) viral load and a liver biopsy. Treatment should be instituted before transplantation; however, candidates who do not clear the virus may still proceed with transplantation because mortality in this group is improved compared with those remaining on dialysis.18 HCV-positive patients may in some units, with informed consent, receive a kidney from an HCV-positive donor. Any possible increased risk may be offset by a significantly reduced waiting time. Patients with hepatitis B and C should be screened every 12 months for hepatocellular carcinoma by liver ultrasound and serum alpha fetoprotein. Those with cirrhosis may be considered for combined kidney-liver transplantation.
Patients at high risk of tuberculosis reactivation after transplantation (previous tuberculosis, abnormal chest radiograph, or positive tuberculin skin test result; residence in an endemic area) who have not been previously treated should receive prophylactic isoniazid after transplantation (see Chapter 105). Recently, interferon-γ (IFN-γ) ELISPOT assays have been proposed to replace or supplement the tuberculin skin test because they appear more sensitive for detecting latent tuberculosis infection.19 The need to screen patients is determined by the likelihood of previous exposure.
Previous graft loss caused by polyoma (BK) viral nephropathy is not a contraindication to repeated transplantation. Waiting for serum and urine BK polymerase chain reaction test results to become negative appears to be preferable,20 whereas the value of graft nephrectomy before repeated transplantation is unclear. Vigilant screening for recurrence is recommended.5
Obesity
Transplantation in obese patients (body mass index [BMI] >30 kg/m2) generally improves survival compared with matched waiting list controls, but inferior outcomes for patient and graft survival, delayed graft function and wound healing, and infective complications have been reported in obese compared with nonobese patients, particularly those with a BMI above 36 kg/m2.21 There are several reports to the contrary, however, including a recent multivariate analysis of registry data suggesting that obesity is not associated with worse graft and patient survival when comorbidities are controlled for.22 Nevertheless, the overweight or obese patient is more likely to develop new-onset diabetes after transplantation, which can adversely affect graft and patient survival. Potential transplant recipients with obesity should be advised to lose weight as a means to decrease this risk before transplantation.
Recurrent Disease
The risk of disease recurrence needs to be discussed as part of the informed consent process, particularly in certain primary renal diseases (e.g., focal segmental glomerulosclerosis). Graft loss attributed to recurrent disease has increased in recent years but still accounts for only 5% of graft loss.23 The risks and management of recurrent disease are discussed in Chapter 108.
Gastrointestinal Disease
Screening for gastrointestinal disease is not warranted in the asymptomatic patient.4,5 Patients with active acute or chronic pancreatitis should not undergo transplantation until they have been clear of symptoms for 12 months. Patients with active peptic ulcer disease should be treated before transplantation with proton pump inhibitors, and this should be continued to prevent ulceration after transplantation. Patients with symptomatic diverticular disease require colonoscopy and potential colonic resection in severe cases before transplantation because they are at increased risk of perforation on immunosuppressive medications.4 Whereas symptomatic cholecystitis should be treated surgically before transplantation, asymptomatic cholelithiasis does not require surgery before transplantation because cholecystectomy after transplantation is required in less than 10% of these patients and results in no increased mortality or morbidity compared with pretransplantation cholecystectomy and no deleterious effects on graft function.24
Genitourinary Disorders
Screening for genitourinary disorders before transplantation is indicated in those with a history or renal ultrasound suggestive of urinary obstruction, especially in children, in whom urologic problems are a major cause of ESRD. If obstruction is found, urologic assessment, which may include voiding cystourethrography and urodynamic studies, is indicated to determine the best course of action to ensure bladder emptying and limit bladder pressures after transplantation; this may involve bladder augmentation, urinary diversion, or self-catheterization.
Native nephrectomy before transplantation should be considered for recurrent or persistent renal sepsis, particularly in the setting of nephrolithiasis. Very large polycystic kidneys may need to be removed to accommodate the transplant kidney. Whether previous grafts should be removed before repeated transplantation is controversial. Nephrectomy of a failed graft is commonly performed on withdrawal of immunosuppression in patients with early graft failure (<12 months)25 to alleviate symptoms such as pain over the graft, fever, and weight loss.26 Other indications include graft sepsis and allowance of room for the new graft. However, unless there is a convincing reason to remove the graft, it is generally left in situ. In these circumstances the patient may need to stay on minimal immunosuppression (e.g., prednisolone, 5 mg/day) for 3 to 6 months to minimize graft tenderness and inflammation. Graft nephrectomy may be associated with an increased risk of human leukocyte antigen (HLA) sensitization,25 perhaps relating to loss of antigen-dependent tolerogenic mechanisms27; this, however, is not a universal finding. Another advantage of leaving the previous transplant in situ is preservation of any residual renal function and urine output.
Pulmonary Disease
Initial assessment by physical examination and chest radiography is indicated for all potential recipients; further testing, such as pulmonary function tests or computed tomographic scanning, is performed if it is clinically indicated. Recent guidelines suggest that patients with a short life expectancy associated with pulmonary disease, such as cor pulmonale, uncontrolled asthma, and severe obstructive lung disease (FEV1 <25% of predicted or Po2 <60 mm Hg [8 kPa] on room air), or those needing home oxygen should be excluded from transplantation.5 Many units require patients to cease smoking before acceptance because smokers have an increased risk of death and graft loss. Cessation of smoking demonstrates positive lifestyle behavior and good compliance, suggesting that these factors will be optimized in the post-transplantation period.
Psychosocial Issues
Psychosocial issues can have a major impact on transplant outcomes. Patients should be evaluated by a health professional experienced in judging capacity to consent and assessing likely compliance with a transplant medication regimen. Compliance with the post-transplantation treatment regimen is vital to minimize premature graft loss. Predicting compliance can be challenging and may be based on pretransplantation compliance, such as adherence to dialysis management regimens. If compliance is unlikely, as judged by medical, psychiatric, psychological, and social work assessment, transplantation should not proceed.3–5
Cognitive impairment is not an absolute contraindication to transplantation as long as appropriate supports and proxy arrangements are in place. Patients with psychiatric illnesses including depression, bipolar affective disorder, and psychosis require assessment by a psychiatrist to determine transplant suitability and to devise a management plan to cope with possible consequences of immunosuppressive medications such as corticosteroids.3–5 Drug and alcohol addiction should be addressed with rehabilitation and demonstrated abstinence before the patient is listed for transplantation.
Presence of Multiple Comorbidities
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree