Valvular Heart Disease
James S. Gammie
Aortic Stenosis
Stanley Osis is a 75-year-old man with no significant medical history. While mowing his lawn, he suffers a brief syncopal event. He is taken to the local emergency department and evaluated. A careful review of systems is unrevealing. On physical examination, his heart rate is 80 beats per minute and regular. Auscultation reveals a harsh crescendo-decrescendo systolic murmur that is best heard in the right second intercostal space and along the left sternal border. The murmur radiates to both carotid arteries. His peripheral pulse has a delayed upstroke and low volume (pulsus parvus et tardus). The lungs are clear, and the examination is otherwise normal. The electrocardiogram (ECG) shows normal sinus rhythm and left ventricular hypertrophy. The chest radiograph shows mild left ventricular enlargement.
What is the most likely diagnosis?
View Answer
On the basis of the characteristic murmur on physical examination, the most likely diagnosis is a syncopal event caused by severe aortic stenosis.
Mr. Osis is admitted to the hospital for 24 hours. Continuous ECG monitoring shows no evidence of arrhythmia. Serial creatine phosphokinase levels are normal. Carotid Doppler studies show only mild stenosis of the left internal carotid artery. A two-dimensional transthoracic echocardiogram reveals concentric left ventricular hypertrophy with severe aortic stenosis. The calculated aortic valve area is 0.7 cm2. Mitral valve function is normal, and the ejection fraction is estimated to be 60%.
What are the three most common causes of aortic stenosis, and what is the most likely cause in this patient?
View Answer
The following are the three most common causes of aortic stenosis:
Degenerative (senile) calcific aortic stenosis: the most common cause of aortic stenosis in patients undergoing aortic valve replacement. It is characterized by immobilization of the aortic valve cusps by dense deposits of calcium. The calcium prevents normal opening of the valve during systole. Degenerative aortic stenosis is seen most commonly in patients over age 65.
Congenital bicuspid aortic valve: affects as many as 2% of infants at birth. Although a bicuspid valve usually does not cause symptoms early in life, it does generate turbulent flow that ultimately leads to fibrosis of the leaflets and to valvular stenosis. Patients with congenital bicuspid aortic valves usually develop symptoms in their 50s and 60s. Congenital bicuspid aortic valve is the most common cause of aortic stenosis in patients under 65 years of age.
Rheumatic aortic stenosis: usually occurs in a previously normal valve. It is characterized by commissural fusion (attachment of the valve cusps to each other in the clefts between the cusps). Some aortic regurgitation is common in patients with rheumatic aortic stenosis, and the mitral valve is almost always diseased.
What are the three cardinal symptoms of aortic stenosis, and what is the pathophysiology of each?
View Answer
Angina, syncope, and congestive heart failure (CHF) are the three cardinal symptoms of aortic stenosis. Progressive obstruction to left ventricular outflow by the stenotic valve causes compensatory concentric left ventricular hypertrophy. This permits maintenance of cardiac output despite a significant obstruction. The hypertrophied left ventricle can maintain a large pressure gradient across the valve without a decrease in cardiac output, left ventricular dilation, or symptoms.
Angina: occurs in two thirds of patients with critical aortic stenosis and in 50% of patients it is associated with coronary artery disease (CAD). Angina is typically associated with exertion. In patients without CAD, angina results from an imbalance in myocardial oxygen supply and demand. Oxygen demand increases as a result of the increased left ventricular mass and wall tension, and myocardial blood flow is compromised by compression of intramyocardial coronary arteries.
Syncope: a result of inadequate cerebral perfusion. Syncope normally occurs during exertion, when arterial blood pressure decreases as a result of systemic vasodilation in the presence of a fixed cardiac output.
Left ventricular failure: caused by increased left ventricular filling pressures. As the ventricle progressively hypertrophies to compensate for the aortic valvular obstruction, it becomes less compliant (stiffer), and left ventricular filling pressures increase. In turn, elevated left ventricular filling pressure is transmitted to the left atrium and the pulmonary capillary bed, which results in clinical symptoms of left ventricular failure, including dyspnea with exertion, paroxysmal nocturnal dyspnea, and orthopnea. Synchronized left atrial contraction is especially important for filling of the thickened, noncompliant ventricle in aortic stenosis. The onset of atrial fibrillation and loss of this atrial kick can lead to significant hemodynamic and clinical deterioration in patients with advanced aortic stenosis.
What is the natural history of aortic stenosis, and what is the characteristic time interval between onset of each of the cardinal symptoms and death?
View Answer
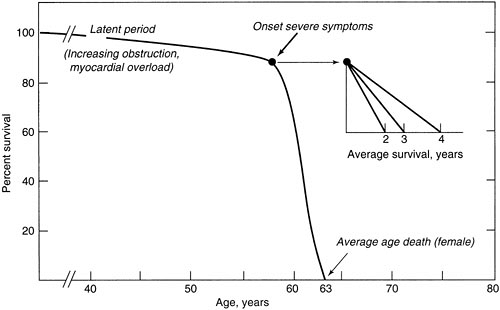
Patients with severe aortic stenosis can remain hemodynamically compensated for years. However, the onset of symptoms is an ominous event that portends a poor prognosis. Once patients develop angina or syncope, the average survival is 2 to 3 years. Average survival with CHF is 1.5 years. The characteristic natural history of this disease (prolonged compensated survival in the absence of symptoms, with rapid deterioration and dismal survival after the onset of symptoms) was defined by Ross and Braunwald (1) in a classic article in 1968 (Fig. 25.1). Death is rare among patients who have aortic stenosis but do not display symptoms. Thus, the indication for operation in a patient with severe aortic stenosis is the development of symptoms.
What is the next step in the workup for patients with aortic stenosis?
View Answer
Cardiac catheterization is mandatory to assess the coronary anatomy. Significant CAD is common among patients with aortic stenosis, and these patients require coronary artery bypass grafting at the time of aortic valve replacement. Cardiac catheterization allows simultaneous measurement of pressure within the left ventricle and in the proximal aorta. Measurement of the pressure gradient across the aortic valve allows precise determination of aortic valve area. Catheterization also allows estimation of left ventricular function and rules out coexisting valvular heart lesions.
What is the Gorlin formula?
View Answer
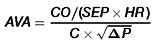
Developed by Richard Gorlin and his father at Peter Bent Brigham Hospital in Boston, this formula allows accurate calculation of the aortic valve area on the basis of hemodynamic data obtained during cardiac catheterization (2):
where AVA is the aortic valve area (cm2), CO is the cardiac output in liters per minute, SEP is the systolic ejection period in seconds per beat, HR is the heart rate, C is a constant, and (P) is the pressure gradient across the aortic valve. The normal aortic valve area is 3 to 4 cm2. Symptoms generally do not develop until aortic valve area is less than 1 cm2.
Are there any medical alternatives to aortic valve replacement?
View Answer
There are no medical alternatives to aortic valve replacement. In percutaneous balloon aortic valvuloplasty, the stenotic aortic valve is dilated with a balloon. Patients generally have a reduction of peak transvalvular gradient of about 50% and an increase in valve area of 0.5 to 1 cm2. However, 50% to 75% of patients develop restenosis within 9 months, and the 1-year mortality is 30% (3). Thus, aortic valvuloplasty should be considered only a palliative procedure applicable to patients who are not candidates for aortic valve replacement.
Mr. Osis is scheduled for elective aortic valve replacement. After a detailed discussion of the risks and benefits of surgery, he agrees to proceed.
Do patients undergoing valve surgery need any special preoperative preparation?
View Answer
All patients planning to have elective heart valve surgery should undergo a careful dental examination. Severely diseased teeth (abscess, periodontal disease) should be extracted before operation to remove a source of sepsis. Other possible sources of sepsis, such as urinary tract infections, respiratory infections, or septic processes within the gastrointestinal tract should be identified and treated before operation. Prosthetic heart valves are particularly susceptible to bacterial seeding in the first few months after surgery. Prosthetic valve endocarditis has a very high (50% to 70%) mortality rate.
What are the ideal characteristics of a valve substitute?
View Answer
An ideal replacement valve would have hemodynamic characteristics similar to those of native human cardiac valves, which are characterized by minimal transvalvular gradients during forward flow and freedom from regurgitation during closure. The perfect valve would be nonthrombogenic and durable, completely free of structural degeneration, resistant to infection, nondestructive to blood elements, readily available, and low in cost. Such a valve substitute has yet to be created.
What are the different types of replacement aortic valves? What factors guide the choice of a valve?
View Answer
The two main categories of replacement valves are mechanical prostheses and bioprostheses. In special cases, homograft aortic valves may be used.
Bioprostheses. The most commonly used bioprostheses are stented tissue valves. They consist of a round cloth sewing ring (used to suture the valve in place) and metal struts that support the three tissue leaflets. The leaflets are made from glutaraldehyde-fixed bovine pericardium or porcine valve leaflets. Stentless bioprostheses are made from a glutaraldehyde-fixed porcine aortic root (the aortic valve and the proximal ascending aorta). Durability is similar to stented tissue valves. Stentless valves are less obstructive than stented tissue valves (they have a larger effective orifice area), but stentless valves are somewhat more time-consuming to insert. Bioprostheses have the advantage of not requiring chronic anticoagulation. This benefit is offset by a limited valve lifespan, 15 to 17 years, in the aortic position. Bioprostheses last longest in older patients; therefore, they are commonly implanted in patients older than 65 years of age and in patients with a life expectancy of less than 15 years. Bioprostheses have a gradual failure mode, such that reoperation can be performed on an elective basis.
Mechanical prostheses. The most commonly implanted mechanical prosthesis is a bileaflet tilting disc valve made of pyrolytic carbon. Mechanical prostheses have excellent long-term durability, but this advantage is offset by the requirement for long-term anticoagulation with warfarin (Coumadin). This results in a 1% to 2% rate of anticoagulant-related hemorrhage per patient-year and 0.2% annual mortality. Women of childbearing age should not receive a mechanical valve because warfarin is teratogenic. In general, mechanical valves should be implanted in patients without contraindications to anticoagulation who have a life expectancy longer than 15 years and who are not intending to bear children. Both bioprosthetic and mechanical valves carry a risk of thromboembolism of 1% to 2% per year. Older patients, whose risk of anticoagulation is greater and who have a life expectancy of less than 15 years, generally benefit from a bioprosthetic valve. Most surgeons would probably recommend a bioprosthetic valve for Mr. Osis.
Mr. Osis undergoes uncomplicated aortic valve replacement. A 25-mm bioprosthesis is inserted in the aortic position. He is extubated 4 hours after surgery and is discharged home on postoperative day 4.
What are the key technical considerations in aortic valve replacement?
View Answer
After cardiopulmonary bypass is established and the heart has been arrested, the aorta is opened and the diseased valve is exposed. The valve leaflets are excised, and calcium deposits are removed. Great care must be taken to remove all loose debris to avoid a stroke. A valve of appropriate size is chosen and sutured in place.
What postoperative complications are unique to aortic valve replacement surgery?
View Answer
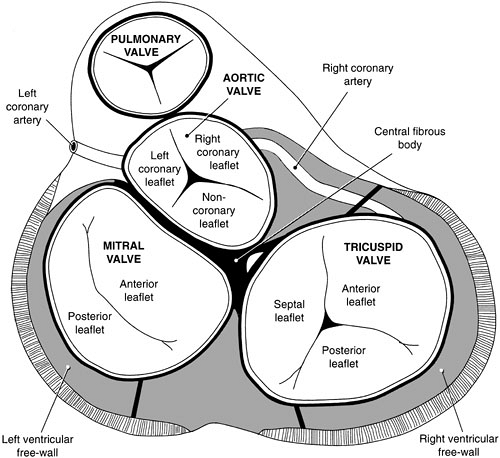
The conduction system passes inferiorly and close to the aortic annulus near the junction of the right coronary and noncoronary valve leaflets (Fig. 25.2). Sutures placed too deep at this location can produce temporary or permanent postoperative heart block. Heart block that requires a permanent pacemaker occurs in about 1% of cases.
What is the average mortality for an aortic valve replacement?
What is the long-term prognosis after aortic valve replacement?
View Answer
The 5-year survival after aortic valve replacement is 75%, and the 10-year survival is 60%.
How should anticoagulation be managed in the early postoperative period? What is the target international normalized ratio (INR) for patients with artificial heart valves?
View Answer
Most patients are given intravenous heparin starting 24 to 48 hours after operation. Warfarin is also administered, and heparin is stopped when the INR is therapeutic, which is 2.5 for prosthetic heart valves (4). Anticoagulation in patients who have received bioprosthetic valves usually consists of warfarin for 3 months after the operation and aspirin only thereafter, although some surgeons administer only aspirin after surgery. Mechanical valves require lifelong anticoagulation.
When can a patient resume normal activities after aortic valve replacement surgery?
View Answer
Light work can generally be resumed 4 to 6 weeks after operation; heavy work, such as lawn mowing, can be resumed 3 to 4 months after operation.
Aortic Regurgitation
Fred Smith is a 54-year-old peanut farmer who is referred by his family physician because of an abnormal chest radiograph, which was obtained because of a recent fever and cough. Although there is no evidence of pneumonia or pulmonary disease on radiograph, the physician thought the ascending aortic silhouette looked abnormal. Mr. Smith says he has no symptoms, and his cough has resolved. He works up to 10 hours daily on his farm and has not noted any decrease in his exercise tolerance or activity level. On physical examination, he appears in general good health. His blood pressure is 160/60 mm Hg with a heart rate of 84 beats per minute. His pulses are prominent and brisk. On auscultation, a diastolic murmur is audible, at the beginning of diastole. No systolic or carotid murmurs are appreciated, and no third or fourth heart sounds are heard. The rest of his examination is unremarkable.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree