Chapter 67 SACROSPINOUS LIGAMENT SUSPENSION FOR VAGINAL VAULT PROLAPSE
Several techniques are used for suspension of the vaginal vault, including the sacrospinous vaginal vault suspension, abdominal sacrocolpopexy, bilateral uterosacral vaginal vault suspension, McCall culdoplasty, iliococcygeus vault suspension, levator myorrhaphy, and many tension-free mesh techniques. The optimal surgical approach for advanced prolapse of the vaginal apex remains a subject of debate and is not well studied. Some surgeons prefer the abdominal route for younger women with advanced prolapse; however, these women are at increased risk for morbidity from the surgery, subsequent volvulus from abdominal adhesions, and foreign body reactions due to graft materials. Other surgeons prefer the vaginal route to decrease postoperative pain and morbidity and for women who are at increased operative risk.
What constitutes success or failure is not delineated in the literature, partly because indications for prolapse surgery are not uniform and the degree and site of prolapse are not always clearly defined. The Pelvic Organ Support Study (POSST) was a multicenter, observational study of 1004 women between the ages of 18 and 83 years that evaluated the prevalence of pelvic organ prolapse in a general gynecologic office setting. The investigators found that 24% of the women had stage 0 prolapse, 38% had stage I, 35% had stage II, and 2% had stage III.1 The prevalent findings of stage II prolapse suggest that a stage II recurrence may not necessarily represent an anatomic failure after reconstructive pelvic surgery. These data support the need for the evaluation of anatomic and functional outcomes. According to some surgeons, the correction of all anatomic defects and restoration of normal anatomy should be the overarching goal. Others regard a compensatory postoperative anatomy as acceptable and perhaps even preferable in certain cases to achieve long-term functional outcomes with minimal risk of recurrent or de novo prolapse.
HISTORY
The vaginal vault suspension was first described in 1892 by Zweifel,2 who tried to suspend the vaginal vault to the sacrotuberous ligament. Richter and Albrich3 later described successful application of this procedure. Nichols introduced the operation to the United States in 1971.4
Several modifications of the sacrospinous vaginal vault suspension have been described. The original technique described by Nichols involves suspension of the vaginal vault to the sacrospinous ligament through the posterior compartment. A modification of this technique was introduced by Winkler and colleagues5 in 2000, suspending the vault to the sacrospinous ligament through the anterior compartment. This technique was developed to address the limitations of the posterior sacrospinous vault suspension by reducing postoperative proximal vaginal narrowing and lateral deviation of the upper vagina.6 In 2003, Kearney and DeLancey7 described the Michigan four-wall sacrospinous suspension, which suspends four points of the open vaginal apex to the sacrospinous ligament. The excess vagina is removed to avoid laxity in the anterior and posterior wall.7 In 1993, Kovac and Cruikshank8 described successful outcomes, even after childbirth, with sacrospinous hysteropexy, in which the uterus was suspended bilaterally to the sacrospinous ligaments.
UNILATERAL AND BILATERAL SACROSPINOUS LIGAMENT SUSPENSIONS
Bilateral suspension has been emphasized in earlier reports.3,9 Pohl and Frattarelli10 reported no recurrent apical prolapse among 40 women treated with bilateral sacrospinous ligament suspension after 6 to 40 months of follow-up. Cespedes11 reported good success after bilateral sacrospinous fixation by the anterior vaginal approach; at 17 months, no recurrent vault prolapse was seen in 27 of 28 women with grade 3 or 4 prolapse. However, there is little evidence to suggest that bilateral suspension improves outcomes compared with the unilateral approach.12 Moreover, compared with the unilateral sacrospinous ligament suspension, the bilateral procedure entails more extensive dissection and requires generous width at the vaginal apex to avoid causing tension between fixation points. Because of these additional challenges and its lack of clear advantage, the bilateral technique is used far less often than unilateral fixation in post-hysterectomy vault prolapse.
INDICATIONS
The most common indication for sacrospinous ligament vaginal vault suspension is resuspending a symptomatic prolapsed post-hysterectomy vaginal apex. The incidence of post-hysterectomy vault prolapse requiring surgery has been estimated at 3.6 per 1000 years.13 The risk increases from 1% to 5% 15 years after hysterectomy. The risk also increases for women who had prolapse as the primary indication for their initial surgery.14,15 For women with pelvic ligaments and connective tissue supports that are severely attenuated or absent, the sacrospinous ligament provides a consistent and strong site for apical fixation. Sacrospinous ligament fixation may be performed at the time of hysterectomy in cases of severe apical prolapse if the uterosacral ligaments are attenuated. The use of the sacrospinous ligament fixation prophylactically at the time of hysterectomy, however, is not widely accepted. Surgeons argue that there is an increase in time and morbidity associated with the procedure, which may not be warranted by the outcomes compared with resuspension using the uterosacral ligaments. Cruikshank and Cox16 performed sacrospinous suspensions at the time of hysterectomy on 48 of 135 women without apparent excess in morbidity. In contrast, Colombo and Milani17 retrospectively compared 62 women who underwent sacrospinous vaginal vault suspensions with 62 women who underwent McCall culdoplasty during vaginal hysterectomy and pelvic reconstruction operations and found significant increases in operative time and blood loss in the sacrospinous group. The rate of recurrent vault prolapse was similar for the two groups.17
Another indication for sacrospinous suspension occurs when women desire to preserve fertility or preserve the uterus. It may also be used to decrease operative time and morbidity in elderly patients who have no evidence of endometrial hyperplasia or cancer. There is an increasing body of evidence that uterine hysteropexy may be just as effective as vaginal hysterectomy for recurrent prolapse. Sacrospinous ligament uterine suspension has been successfully used for women with symptomatic uterovaginal prolapse.8,18,19
Kovac and Cruikshank8 evaluated 19 women (average age, 27 years) who underwent uterine-sparing sacrospinous ligament suspension and reported that 4 of 5 women subsequently had successful pregnancies and vaginal deliveries without recurrent apical prolapse. Hefni and colleagues18 evaluated 109 women (61 with uterine suspension and 48 with vaginal hysterectomy and concomitant sacrospinous suspension). The mean age of these cohorts was 60 years, and after a mean follow-up period of 34 months, success rates were comparable with regard to vault prolapse and cystocele recurrences. They found significantly less blood loss, shorter operative time, and fewer complications with uterine preservation.18 Maher and coworkers19 retrospectively compared 34 women with sacrospinous ligament hysteropexy with 36 women with hysterectomy and sacrospinous vaginal vault suspension for symptomatic uterine prolapse. The mean follow-up was 36 months for the hysterectomy group and 26 months for the hysteropexy group. Subjective and objective success rates were similar, and they found a significant reduction in operative time and blood loss with uterine preservation.19
The sacrospinous hysteropexy entails fixation of the distal uterosacral ligaments and paracervical tissue to the sacrospinous ligament. Bilateral rather than unilateral suspension was performed in 15 of the 19 subjects in Cruikshank’s series to avoid lateral deviation of the uterus and any adverse effect it might have on reproductive capacity.8 Alternatively, Hefni and associates18 performed all of their hysteropexies unilaterally.
ANATOMY
The sacrospinous ligament is an excellent support structure for the apex of the vagina. In 1992, DeLancey20 described the anatomic aspects of vaginal eversion after hysterectomy and found that the structures supporting the vagina could be divided into three levels, primarily vertical in orientation. The first level of support, which is suspensory in nature, occurs in the upper vagina adjacent to the cervix and is suspended above by long connective tissue fibers.20 These suspension points should lift the apex of the vagina to the level of or above the ischial spine. These apical or level I vaginal supports are increasingly recognized as a critical component in overall pelvic floor support.21 Loss of level I support can occur after abdominal or vaginal hysterectomy in as many as 2% to 45% of patients due to failure to resuspend the vaginal cuff to the uterosacral ligaments. Resulting vaginal vault prolapse can occur with or without an enterocele.21,22 The sacrospinous ligament provides a good option for level I support.
A good understanding of the anatomy of the sacrospinous ligament helps to minimize complications at the time of surgery. Few structures are at risk for injury in a correctly performed procedure. The sacrospinous ligament attaches laterally to the ischial spine and medially to the lateral margins of the sacrum and coccyx.23 The pudendal nerve leaves the pelvis through the greater sciatic foramen, medial to the sciatic nerve and internal pudendal vessel, between the piriformis and coccygeus muscles and passes under the sacrospinous ligament close to its attachment to the ischial spine.24 Verdeja found that the pudendal neurovascular bundle ranges from 0.90 to 1.30 cm medial to the ischial spine; the sciatic nerve is located 3.10 to 3.30 cm medial to the spine.25 Based on these anatomic relationships, neurovascular injury seems most likely along the lateral third of the sacrospinous ligament when suturing. Another series of cadaver dissections, performed by Barksdale and colleagues,26 showed the pudendal neurovascular bundle to be relatively shielded from injury by the ischial spine and sacrospinous ligament. The inferior gluteal artery—with a more perpendicular course relative to the ligament—was the vascular structure whose location appeared most vulnerable to injury. These researchers referred to three elements of the operation that may carry particular risk: suture placement along the posterior ligament, retractor placement beyond the ligament, and overly aggressive denuding of the ligament surface.26
PREOPERATIVE AND PERIOPERATIVE CONSIDERATIONS
Richardson27 has described site-specific defects in the endopelvic fascia in the anterior, posterior, and apical vaginal wall as the anatomic basis for cystocele, rectocele, and enterocele, respectively. He believed that the surgeon should be able to recognize and repair all existing pelvic floor support defects at the time of surgery. He described the post-hysterectomy enterocele as a defect in the endopelvic fascia at the vaginal apex due to failure to reconstitute pubocervical and rectovaginal fascia in this region. This allows small bowel and peritoneum to herniate into the rectovaginal septum and come in direct contact with vaginal epithelium without intervening muscle or fascia.21,27 An unrecognized enterocele is often to blame for the rare case of recurrent apical prolapse after sacrospinous vaginal vault suspension. Cruikshank and Muniz28 suggested the importance of the site-specific repair in a longitudinal study of 695 patients who underwent sacrospinous vaginal vault suspensions over a 16-year period. Their cohort was divided into three groups. The first group, treated between 1985 and 1990, had only sacrospinous suspensions with or without a hysterectomy (n = 173); the second group, treated between 1991 and 1994, had anterior and posterior colporrhaphies and high cul-de-sac closures performed concomitantly with the sacrospinous suspensions (n = 221); and the third group, treated between 1995 and 2000, had a more inclusive, site-specific approach with the suspensions (n = 301). Recurrence rates dropped significantly between the first and third groups, with the third group having the highest success rates.28 Appropriate patient selection and flexibility in the operating room can maximize the odds of success.
Preoperative evaluation should include assessment of bony and soft tissue pelvic anatomy. Markedly foreshortened bony pelvic dimensions, such as a true platypoid pelvis, may limit the surgeon’s ability to preserve adequate vaginal length and weigh against the choice of sacrospinous suspension for sexually active patients. When considering soft tissue, the length of the vaginal vault should be sufficient to avoid tension after sacrospinous fixation, which may predispose to suture pullout, anatomic distortion, and possibly unmasking potential urinary incontinence due to posterior displacement of the urethra from the pubic bone.29 In the absence of adequate length, operative revision of the cuff or suspension to a more anterior or distal structure, such as iliococcygeus fascia, may be performed.30 Consideration of concomitant sacrospinous vaginal suspension on the right, with an iliococcygeal suspension on the left at a slightly more distal point, may lead to improved apical support and diameter.
Preoperative planning should include assessment of estrogen status for postmenopausal women and consideration of local estrogen therapy before surgery if the vaginal cuff epithelium appears thin or poorly vascularized. Estrogen improves vaginal vascularization, epithelial thickness, and collagen in the connective tissue.31 A well-estrogenized vaginal epithelium can facilitate healing at the site of suture fixation and reduce the likelihood of subsequent surgical failure resulting from suture pullout from the vaginal side.
TECHNIQUE
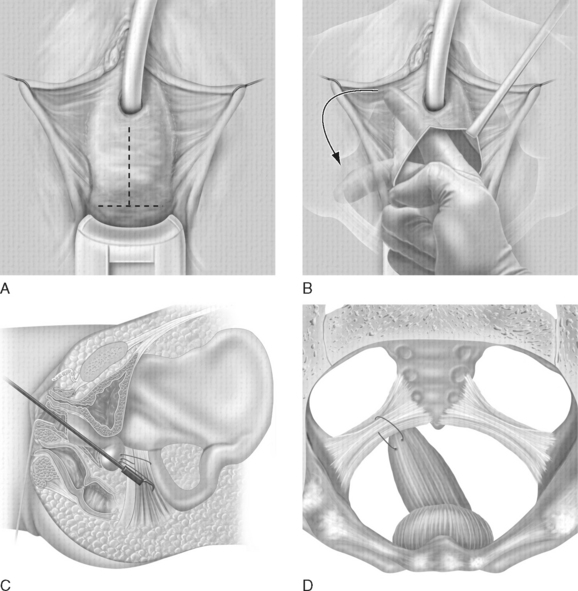
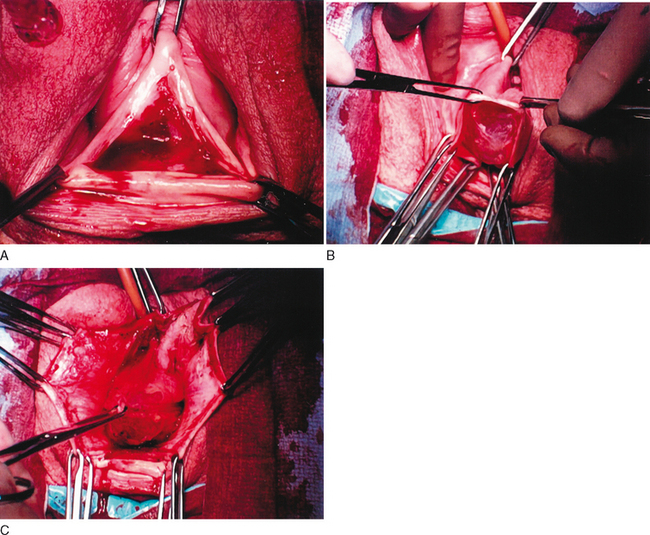
Exposing the Ligament
Access to the sacrospinous ligament and coccygeus muscle can be achieved by several means. The most common approach, as described by Nichols,4 involves a posterior vaginal incision and posterior colporrhaphy dissection, facilitating perforation of the rectal pillar near the ischial spine. With blunt dissection of the pararectal space medial to the ligament, the coccygeus muscle and sacrospinous ligament are exposed. The anterior sacrospinous suspension technique (Fig. 67-1)5,6 involves perforation into the retropubic space through an anterior vaginal wall incision, as would be performed for an anterior colporrhaphy, and dissection of the ipsilateral paravesical and paravaginal area from the level of the bladder neck to the ischial spine along the arcus tendineus fasciae pelvis (Fig. 67-2). The dissection is performed in a manner similar to that for a vaginal paravaginal repair. This dissection opens a large space for the vaginal apex and avoids the narrowing often found in posterior approaches with perforation through the rectal pillars. For cases involving mainly anterior compartment defects with no rectocele, the anterior approach facilitates suspension of the vaginal apex without a posterior vaginal incision.