CHAPTER 29 Medical Aspects of Urolithiasis
![]() How common is nephrolithiasis?
How common is nephrolithiasis?
The overall prevalence of stone disease is approximately 8%.
![]() How many Americans experience a bout of renal colic caused by nephrolithiasis annually?
How many Americans experience a bout of renal colic caused by nephrolithiasis annually?
Approximately 1,200,000.
![]() How does region affect the incidence of nephrolithiasis?
How does region affect the incidence of nephrolithiasis?
More common in the southeast United States and less common in the northwest.
![]() Is the worldwide incidence of kidney stones increasing, decreasing, or about the same and why?
Is the worldwide incidence of kidney stones increasing, decreasing, or about the same and why?
The rate of kidney stone disease is increasing worldwide. This is thought to be the result of more cultures adopting a Western diet as well as global warming contributing to increased dehydration.
![]() Is nephrolithiasis more common in men or women and by what ratio?
Is nephrolithiasis more common in men or women and by what ratio?
The male:female ratio is shifting, in the last few years it has been closer to 3:1, but is currently 1.7:1. Roughly 7% of women and 13% of men will develop at least one stone at some point in their lifetimes.
![]() What type of calculus is equally common in men and women?
What type of calculus is equally common in men and women?
Cystine and uric acid stones.
![]() What type of calculus is more common in women than men?
What type of calculus is more common in women than men?
Struvite stones are twice as likely to occur in women because of the higher incidence of urinary tract infections in females.
![]() What is the most common age at presentation with nephrolithiasis?
What is the most common age at presentation with nephrolithiasis?
A peak incidence for nephrolithiasis occurs from 30 to 60 years old with 35 to 45 being the peak decade.
![]() What are the most common associated (non-chemistry) reasons for formation of stones in children, women, and men, respectively?
What are the most common associated (non-chemistry) reasons for formation of stones in children, women, and men, respectively?
• Children:
![]() Voiding dysfunction
Voiding dysfunction
![]() Anatomic abnormalities (ie, valves)
Anatomic abnormalities (ie, valves)
• Women:
![]() Prolapse
Prolapse
![]() Neurogenic bladder
Neurogenic bladder
![]() Foreign bodies
Foreign bodies
![]() Urinary tract infection
Urinary tract infection
• Men:
![]() BPH, urethral stricture, bladder diverticulae
BPH, urethral stricture, bladder diverticulae
![]() Neurogenic bladder
Neurogenic bladder
![]() Bladder stones make up what percentage of all urinary calculi?
Bladder stones make up what percentage of all urinary calculi?
5%.
![]() What is the current estimated yearly cost of kidney stone disease in the United States?
What is the current estimated yearly cost of kidney stone disease in the United States?
More than 2 billion dollars a year and rising.
![]() Does family history of stones increase patient’s risk of stone formation?
Does family history of stones increase patient’s risk of stone formation?
Yes, 3-fold increased risk.
![]() What is the risk for stone recurrence after an initial symptomatic episode of urolithiasis? How does risk of stone formation change over time?
What is the risk for stone recurrence after an initial symptomatic episode of urolithiasis? How does risk of stone formation change over time?
• Increasing risk each year:
![]() Year 1: 10% to 15% risk of additional stone formation
Year 1: 10% to 15% risk of additional stone formation
![]() Year 5: 50% to 60%
Year 5: 50% to 60%
![]() Year 10: 70% to 80%
Year 10: 70% to 80%
![]() What are some risk factors for stone formation?
What are some risk factors for stone formation?
• Dehydration
• Increased weight (obesity)
• Family history
• High sodium diet
• High animal protein diet
• Previous history of stones
![]() What populations of patients necessitate treatment of asymptomatic renal calculi?
What populations of patients necessitate treatment of asymptomatic renal calculi?
• Pediatric
• Woman planning pregnancy
• Solitary kidney or transplant
• Occupational hazard—military, pilot, etc
![]() What is the recommended antibiotic prophylaxis for stone manipulation?
What is the recommended antibiotic prophylaxis for stone manipulation?
• Single systemic quinolone
• Cephalosporin
![]() What effect, if any, does obesity have on kidney stone formation?
What effect, if any, does obesity have on kidney stone formation?
Obesity is a strong predictor of stone recurrence especially in first-time stone formers. Obesity causes changes in urine chemistry reflecting increased calcium, uric acid, sodium, and decreased citrate in the urine. Urine of obese patients has also been found to have a lower pH. Obesity is associated with a higher risk for both uric acid and calcium oxalate stones.
![]() How does obesity affect nephrolithiasis?
How does obesity affect nephrolithiasis?
Through purine gluttony, dietary excesses, diabetes, and insulin resistance.
![]() How might insulin resistance contribute to stone forming?
How might insulin resistance contribute to stone forming?
Insulin resistance at the cellular level results in hyperinsulinemia, which in turn causes defects in renal production of ammonia, lowering the pH of the urine.
![]() What medications can be used to facilitate ureteral stone passage in urolithiasis?
What medications can be used to facilitate ureteral stone passage in urolithiasis?
α1-Adrenergic antagonists facilitate stone passage. Tamsulosin and other α-blockers, used often in BPH, facilitate passage of stones less than 10 mm. The rate of stone passage increases approximately 30% with the use of α-blockers, and the need for narcotics and surgical intervention decreases. Steroids may be used to reduce ureteral edema for easier stone passage. In addition, calcium channel blockers such as nifedipine have been shown to cause ureteral dilatation and relaxation resulting in increased spontaneous stone passage rates.
![]() What percentage of patients requires intervention for passage of a stone?
What percentage of patients requires intervention for passage of a stone?
It is 20% overall; 50% of patients with stones ≥5 mm. A proximal ureteral stone will have a lower chance of spontaneous stone passage (50%) than a distal ureteral stone (85%). In general, a stone 4 mm or less in size has a 90% chance of eventual passage.
![]() In a patient presenting with a ureteral stone, is antibiotic therapy indicated?
In a patient presenting with a ureteral stone, is antibiotic therapy indicated?
Patients with bacteriuria and a ureteral stone should be treated with antibiotics given the increased risk of infectious complications and urosepsis (according to American urologic association (AUA) guidelines). Patients with planned interventions should be treated with antibiotics prior to the intervention.
![]() In patients presenting with a ureteral stone, with well-controlled symptoms, what are the recommendations for medical expulsion therapy?
In patients presenting with a ureteral stone, with well-controlled symptoms, what are the recommendations for medical expulsion therapy?
• Patients should be comfortable with this treatment and have good renal reserve.
• Patients should be informed that α-blocker therapy is used “off label.”
• Patients should have radiologic follow-up to assess for progression.
![]() What is the average time to spontaneous stone passage?
What is the average time to spontaneous stone passage?
The time to spontaneous passage correlates with stone size. The average time to stone passage is 1 week for <2 mm, 2 weeks for 2 to 4 mm, and 3 weeks for >4 mm.
![]() How long should one wait for an asymptomatic stone to pass or move before considering doing a surgical procedure?
How long should one wait for an asymptomatic stone to pass or move before considering doing a surgical procedure?
For stones <10 mm approximately 95% will pass spontaneously within 4 to 6 weeks, following that time, if the stone has not passed spontaneously, one should consider intervention. After that time, the risk of possible long-term complications, such as strictures, increases.
![]() What are the indications for hospitalization in the acute setting of renal colic?
What are the indications for hospitalization in the acute setting of renal colic?
Severe pain requiring continuing parenteral pain medication, fever and infection, intractable vomiting, renal insufficiency or rising creatinine, and a solitary kidney. Urinary tract infection with an obstructed system is an indication for emergent decompression of the collecting system with ureteral stenting or placement of a percutaneous nephrostomy tube.
![]() What medication decreases the need for narcotics to control renal colic?
What medication decreases the need for narcotics to control renal colic?
Ketorolac (Toradol) IV has been demonstrated to decrease the need for narcotics in acute renal colic. IV Acetaminophen has also been used.
![]() Can stone disease be ruled out in the absence of hematuria?
Can stone disease be ruled out in the absence of hematuria?
No, approximately 15% of patients with ureteral stones have no RBC/HPF on microscopic urinalysis.
![]() What diagnostic imaging should be used if there is clinical suspicion of urolithiasis?
What diagnostic imaging should be used if there is clinical suspicion of urolithiasis?
Noncontrast spiral/helical CT. Patients with BMI <30 may have low-dose noncontrast CT as this will reduce the amount of exposure to radiation in these patients. Obese patients should have standard noncontrast CT as their body habitus precludes a low-dose study. CT can also assist in diagnosing other nonrenal causes of flank pain.
![]() Should a kidneys, ureter, bladder (KUB) be performed when the CT scan has already identified a stone? Why or why not?
Should a kidneys, ureter, bladder (KUB) be performed when the CT scan has already identified a stone? Why or why not?
Yes, a KUB should be immediately performed when the CT scan has demonstrated a stone for several reasons. First, it provides a baseline study to easily track progress of the stone. Next, it quickly separates radiopaque from radiolucent stones. Finally, it provides a clear idea of the shape and size of the stone as well as its surgical orientation.
![]() What is the recommended imaging for follow-up of ureteral stones for patients that are being managed expectantly?
What is the recommended imaging for follow-up of ureteral stones for patients that are being managed expectantly?
For radio-opaque stones, ultrasound and KUB should be the preferred method of imaging. If stones are not radio-opaque, low-dose noncontrast CT should be employed.
![]() Compare modalities of imaging as diagnostic tests for urolithiasis.
Compare modalities of imaging as diagnostic tests for urolithiasis.
• Ultrasound detects 50% to 60% of stones.
• KUB detects 60% to 70%.
• Intravenous urogram (IVU) detects 70% to 90%.
• Noncontrast CT scan detects 90% to 100%.
![]() What differential diagnoses with urolithiasis may be evaluated using noncontrast CT?
What differential diagnoses with urolithiasis may be evaluated using noncontrast CT?

![]() What are some of the limitations of CT imaging of renal stones?
What are some of the limitations of CT imaging of renal stones?
CT cannot identify pure indinavir stones. It also cannot differentiate between radiolucent and radiopaque stones, and it does not always give an accurate rendering of the exact size or shape of some stones. It is also expensive and has significant radiation exposure so it cannot be used in pregnancy.
![]() How accurate is CT in diagnosing urolithiasis?
How accurate is CT in diagnosing urolithiasis?
98% sensitivity, 95% specificity, 98% positive predictive value, and 95% negative predictive value.
![]() Is MRI a useful imaging study in urolithiasis?
Is MRI a useful imaging study in urolithiasis?
MRI does not visualize stones and should not be used to make the diagnosis of urolithiasis. MR urography may have a role in the evaluation of flank pain in pregnancy, although ultrasound is the first choice in imaging.
![]() What is first- and second-line imaging modality for stones in pregnancy?
What is first- and second-line imaging modality for stones in pregnancy?
• US
• IVP
• MRI can also be used, it is not very effective in visualizing stones, but may pick up filling defects
![]() What risk factors contribute to stones in pregnancy?
What risk factors contribute to stones in pregnancy?
• Multiparity
• Caucasian
• Prior stone episode
• Renal disease
• Hypertension
![]() During what trimester is a pregnant woman most likely to have a symptomatic stone?
During what trimester is a pregnant woman most likely to have a symptomatic stone?
Second and third.
![]() What is the recommended treatment for a symptomatic ureteral stone in a pregnant woman?
What is the recommended treatment for a symptomatic ureteral stone in a pregnant woman?
Ureteroscopy is acceptable treatment in the second and third trimester.
![]() According to AUA guidelines, what is the first-line treatment for pediatric stones?
According to AUA guidelines, what is the first-line treatment for pediatric stones?
While both ureteroscopy and extracorporeal shock wave lithotripsy (ESWL) can be used, some literature favors ESWL for first-line treatment of pediatric renal/ureteral stones.
![]() Are forced fluids helpful in the management of acute renal colic?
Are forced fluids helpful in the management of acute renal colic?
A recent randomized trial demonstrated that fluid bolus in the emergency department did not impact pain or stone passage. As such, forceful hydration may not help with stone expulsion although many patients with acute renal colic will be dehydrated and may need IV fluid replenishment for other reasons.
![]() What fluids and drinks should generally be avoided in stone formers?
What fluids and drinks should generally be avoided in stone formers?
Phosphoric acid in soda drinks will acidify the urine and increase the risk of kidney stones. Performance drinks are often high in sodium content and will increase the risk of stones.
![]() How does dietary sodium impact the risk of stone formation?
How does dietary sodium impact the risk of stone formation?
Every teaspoon of salt increases urinary calcium by 23 mg. It leads to higher rates of bone resorption and fluid retention while decreasing urinary citrate levels by 20%.
![]() Is significant oral calcium restriction recommended for calcium stone formers?
Is significant oral calcium restriction recommended for calcium stone formers?
Aggressive calcium restriction increases the risk of stone formation. Calcium is important for bone homeostasis and to bind oxalate in the bowel. A moderate limitation of calcium intake is reasonable.
![]() What complementary approaches are available for patients with hypercalciuria?
What complementary approaches are available for patients with hypercalciuria?
Fish oil supplementation (Eicosapentaenoic acid, n-3 fatty acid) competes with arachidonic acid (n-6) resulting in a decrease in prostaglandin E2. This leads to a decrease in renal calcium excretion, increase of renal calcium reabsorption, and decreased bone resorption. It has been demonstrated to decrease urinary supersaturations for both calcium phosphate and calcium oxalate.
![]() What dietary changes can influence calcium excretion in the urine?
What dietary changes can influence calcium excretion in the urine?
Increased sodium intake results in increased urinary calcium. An increase of 100 mEq of dietary sodium will increase urinary excretion by 50 mg/day.
![]() What oral medication can effect hypercalciuric stone formation?
What oral medication can effect hypercalciuric stone formation?
Thiazides are used to blunt the increased calcium excretion in the urine. High sodium diet can block this effect.
![]() How does low calcium diet effect stone formation?
How does low calcium diet effect stone formation?
Low calcium diets increase urinary oxalate, therefore encouraging stone formation. Low calcium diet is NOT recommended.
![]() How does dietary oxalate affect stone formation?
How does dietary oxalate affect stone formation?
Urinary calcium and oxalate are excreted in a bound state, thereby staying in solution. If there is less calcium excreted, oxalate is not bound and is free to facilitate oxalate stone formation.
![]() What food products contain high levels of oxalate?
What food products contain high levels of oxalate?
Chocolate, green leafy vegetables (spinach), and nuts.
![]() What effect does a high animal protein diet have on calcium, uric acid, pH, and citrate?
What effect does a high animal protein diet have on calcium, uric acid, pH, and citrate?
• Increase in calcium and uric acid in the serum.
• Decrease in urinary pH and urinary citrate.
![]() Which patients should undergo a full metabolic evaluation for nephrolithiasis?
Which patients should undergo a full metabolic evaluation for nephrolithiasis?
1. Pediatric stone formers
2. Stone composition: cystine, uric acid, and struvite
3. Nephrocalcinosis
4. Renal failure and transplant patients
6. High stone recurrence (> 1 per year)
7. Strongly motivated to follow through on long-term treatment
![]() What else is included in a full metabolic evaluation for nephrolithiasis?
What else is included in a full metabolic evaluation for nephrolithiasis?
1. History and physical—including diet history, fluid intake, medications, history of recurrent urinary tract infections, personal and family history of stone events
2. Stone analysis—chemical analysis of stones or fragments recovered
3. Urinalysis
4. Serum electrolyte/metabolic panel—serum sodium, potassium, chloride, bicarbonate, creatinine, uric acid, phosphate, magnesium, calcium, and alkaline phosphatase
5. Parathyroid hormone level (intact) and 25 (OH) vitamin D depending on calcium level
6. 24-hour urine collection on normal diet
7. 24-hour urine collection with dietary changes and increased fluid. (Some literature suggests that a single 24-hour urine may be sufficient for metabolic analysis of most patients, and some practitioners use 2 random 24-hour urine collections.)
![]() What are the components of a complete 24-hour urine evaluation?
What are the components of a complete 24-hour urine evaluation?
1. Total urine volume
2. pH urine
3. Calcium urine
4. Oxalate urine
5. Uric acid urine
6. Citrate urine
7. Sodium urine
8. Sulfate phosphorus urine
9. Magnesium urine
10. Ammonium urine
11. Potassium urine
12. Creatinine urine
13. Calcium oxalate
14. Brushite
15. Sodium urate
16. Struvite
17. Uric acid
![]() What is the most common reason for an inaccurate 24-hour urine evaluation?
What is the most common reason for an inaccurate 24-hour urine evaluation?
Inadequate collection, not collecting for full 24 hours.
![]() In what percentage of patients can at least one metabolic abnormality predisposing to urolithiasis be identified?
In what percentage of patients can at least one metabolic abnormality predisposing to urolithiasis be identified?
A probable metabolic etiology for nephrolithiasis is found in 97% of patients who are properly evaluated.
![]() What is the most common type of stone formed?
What is the most common type of stone formed?
Calcium oxalate stones make up approximately 70% of all renal calculi.
![]() What types of crystal are depicted in this urine sample?
What types of crystal are depicted in this urine sample?
Figure A.
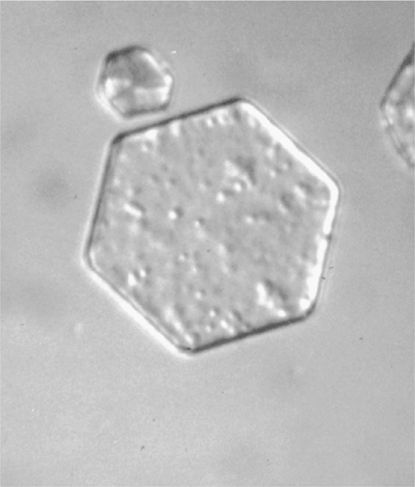
Cystine: hexagonal shape.
Figure B.
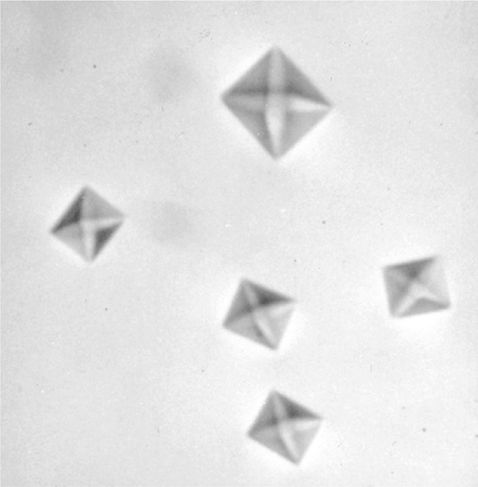
Calcium oxalate: star-box shape.
Figure C.

Calcium phosphate: coffin shape.
![]() What are the 5 most important urinary chemical abnormalities found in stone formers?
What are the 5 most important urinary chemical abnormalities found in stone formers?
Hypercalciuria, hypocitraturia, hyperuricosuria, hyperoxaluria, and low urinary volume.
![]() What are some promotors of urinary stone formation?
What are some promotors of urinary stone formation?
1. Calcium
2. Sodium
3. Oxalate
4. Urate
5. Cystine
6. Low urine pH
7. Tamm-Horsfall proteins
8. Low urine flow/volume
![]() What are some inhibitors of stone formation?
What are some inhibitors of stone formation?
1. Citrate
2. Magnesium
3. Pyrophosphate
4. Tamm-Horsfall protein
5. Urinary prothrombin fragment 1
6. Protease inhibitor
7. Glycosaminoglycans
8. Osteopontin (uropontin)
9. Renal lithostathine
10. Bikunin
11. Calgranulin
12. High urine flow/volume
![]() How does potassium citrate aid in prevention of stone formation?
How does potassium citrate aid in prevention of stone formation?
Potassium citrate corrects acidosis and increases urinary citrate levels. Citrate is a potent inhibitor of stone formation, inhibiting calcium stone crystallization and lowering the saturation of calcium in urine.
![]() Citrate is an important inhibitor of stone formation. What drink is highest in citrate content?
Citrate is an important inhibitor of stone formation. What drink is highest in citrate content?
Grapefruit juice (14,500 mg/L of citrate). However, grapefruit juice is also moderately high in oxalate content. The most effective means to obtain dietary citrate is 4 oz of concentrated lemon juice per day, usually as lemonade.
![]() How does citrate supplementation affect citrate levels in the urine?
How does citrate supplementation affect citrate levels in the urine?
Citrate absorbed from the intestines is converted to bicarbonate in the liver. This increased alkali load stimulates renal excretion of citrate, raises the urinary pH, and increases urinary citrate levels.
![]() What role does citrate play in urinary stone formation?
What role does citrate play in urinary stone formation?
Citrate is an stone inhibitor for calcium oxalate stones.
![]() What physical states result in hypocitraturia?
What physical states result in hypocitraturia?
• Metabolic acidosis
![]() Distal RTA (Type 1)
Distal RTA (Type 1)
![]() Chronic diarrhea
Chronic diarrhea
![]() Physical exercise (lactic acidosis)
Physical exercise (lactic acidosis)
![]() High protein diet
High protein diet
![]() Hydrochlorothyazide (HCTZ) use, potassium wasting
Hydrochlorothyazide (HCTZ) use, potassium wasting
• Urinary tract infection
![]() Citrate is degraded by organism
Citrate is degraded by organism
![]() How does acidosis effect stone formation?
How does acidosis effect stone formation?
Acidosis increases calcium resorption in the GU tract and from the bone, therefore there is more circulating calcium, and decreased renal resorption of calcium in the kidney. Calcium is then excreted in the urine.
![]() In what ways does citrate inhibit calcium stone formation?
In what ways does citrate inhibit calcium stone formation?
Citrate binds calcium, forming a soluble complex in urine and competing with its binding with oxalate or phosphate. Citrate will also alkalinize the urine, thereby inhibiting the formation of both calcium oxalate and uric acid crystals.
![]() What is the optimal level of oral citrate supplementation and how can this be determined clinically?
What is the optimal level of oral citrate supplementation and how can this be determined clinically?
An optimal concentration of 320 to 340 mg of citrate/L of urine and 24-hour urine levels ≥600 to 650 mg of citrate have been suggested as long as the urinary pH remains at or below 7. While a 24-hour urine test for citrate would be optimal, it is impractical to perform this test too often. Another method is to maximize the potassium citrate supplementation until the patient reaches maximum tolerance or the pH exceeds 7. FDA-approved urinary dipsticks to specifically monitor urinary pH are now available commercially (www.uridynamics.com).
![]() What is the most important chemical mediator of intestinal absorption of calcium?
What is the most important chemical mediator of intestinal absorption of calcium?
The most important chemical mediator of calcium absorption is calcitriol (1,25-dihydroxyvitamin D3), which increases calcium transport across the intestinal lumen. The conversion of precursors to calcitriol is initiated by hypophosphatemia and parathyroid hormone (PTH).
![]() What additional effects does PTH have on calcium homeostasis?
What additional effects does PTH have on calcium homeostasis?
PTH is a polypeptide that stimulates the osteoclasts to resorb bone, releasing calcium and phosphate into the bloodstream. PTH also acts upon the kidney to resorb calcium from the urine into the blood.
![]() PTH increases calcium resorption in the kidney, so explain why we find hypercalciuria in patients with hyperparathyroidism.
PTH increases calcium resorption in the kidney, so explain why we find hypercalciuria in patients with hyperparathyroidism.
The calcium resorption effect of PTH is overcome in hyperparathyroidism by the extremely high serum calcium levels.
![]() True/False: The incidence of stone formation in hyperparathyroid patients is unrelated to the degree of serum calcium or PTH elevations.
True/False: The incidence of stone formation in hyperparathyroid patients is unrelated to the degree of serum calcium or PTH elevations.
True.
![]() What percentage of hyperparathyroid patients will have nephrolithiasis as their presenting or initial symptom?
What percentage of hyperparathyroid patients will have nephrolithiasis as their presenting or initial symptom?
22%.
![]() In what ways are hyperparathyroid stone formers different from hyperparathyroid patients without nephrolithiasis?
In what ways are hyperparathyroid stone formers different from hyperparathyroid patients without nephrolithiasis?
Hyperparathyroid stone formers are more likely to be younger and have hypercalciuria. Their hyperparathyroidism is almost always caused by parathyroid adenomas as opposed to hyperplasia.
![]() How much dietary calcium is absorbed on a daily basis?
How much dietary calcium is absorbed on a daily basis?
Approximately 33% of the total dietary calcium (600-1200 mg or 15 mg/kg) is absorbed and 100 to 200 mg is secreted into the intestinal tract for a net absorption of approximately 100 to 300 mg. Calcium ions that are complexed in the gastrointestinal (GI) tract to phosphate, oxalate, citrate, and sulfate are not absorbed.
![]() How is hypercalciuria defined? (Give at least 3 definitions.)
How is hypercalciuria defined? (Give at least 3 definitions.)
(1) The excretion of greater than 200 mg/24 h on a diet of 400 mg calcium and 100 mEq sodium for a week.
(2) On an unrestricted diet, hypercalciuria can be defined as >275 mg calcium/day for men, >250 mg/day for women. (3) Another definition is more than 4 mg of calcium/kg body weight/day
![]() What is the estimated percentage of patients with calcium oxalate stones who will demonstrate hypercalciuria?
What is the estimated percentage of patients with calcium oxalate stones who will demonstrate hypercalciuria?
Approximately 35%.
![]() What are the 3 basic types of hypercalciuria?
What are the 3 basic types of hypercalciuria?
1. Absorptive hypercalciuria caused by increased intestinal absorption of calcium.
2. Renal leak hypercalciuria caused by an abnormality in renal absorption of calcium.
3. Resorptive hypercalciuria caused by augmented bone demineralization.
Hypercalciuria types:
• Absorptive hypercalciuria
![]() Increased calcium absorption in the GI tract leading to increased plasma calcium, decreased PTH, and increased urinary calcium.
Increased calcium absorption in the GI tract leading to increased plasma calcium, decreased PTH, and increased urinary calcium.
• Renal leak hypercalciuria
![]() Increased calcium excretion into the urine, decreased calcium in plasma, increased PTH and increased GI calcium absorption, and increased bone absorption.
Increased calcium excretion into the urine, decreased calcium in plasma, increased PTH and increased GI calcium absorption, and increased bone absorption.
• Resorptive hypercalciuria (primary hyperparathyroidism)
![]() Increased PTH → increased bone resorption and increased GI Ca absorption, increased plasma calcium, and increased urinary calcium
Increased PTH → increased bone resorption and increased GI Ca absorption, increased plasma calcium, and increased urinary calcium
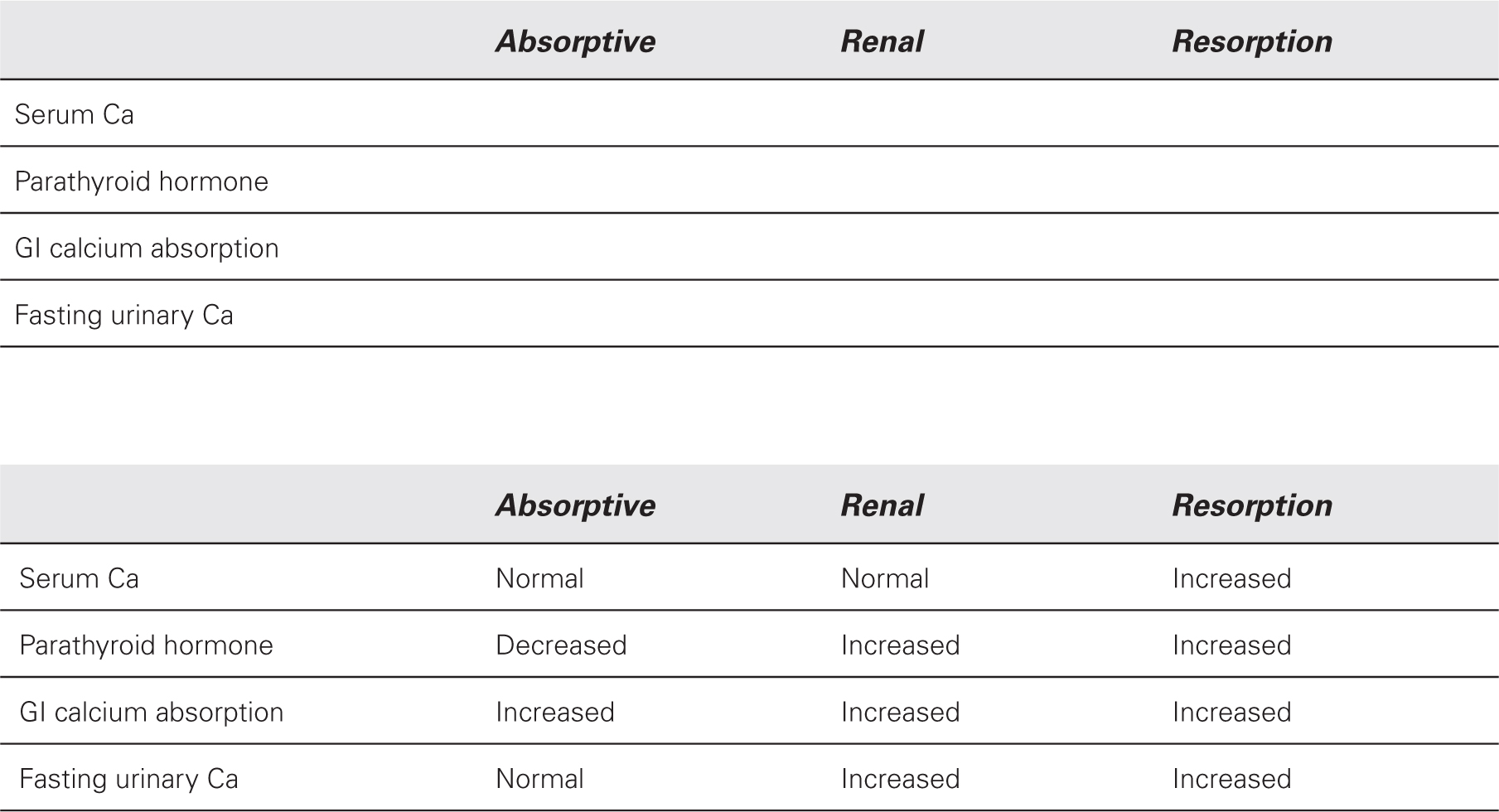
Using the below table, test your knowledge of hypercalciuric states. Options are “Normal”, “Increased”, or “Decreased.”
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree



