Chapter 80 Hepatocellular carcinoma
Overview
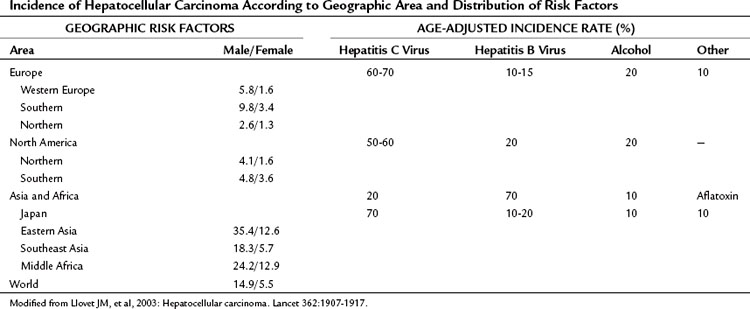
Hepatocellular carcinoma (HCC) is the most common primary liver tumor. It currently represents the sixth most common cancer worldwide and is the third major case of cancer death. Its incidence presents marked geographic differences (Table 80.1; Ferlay et al, 2010). Asia and sub-Saharan Africa constitute high-risk areas with yearly rates exceeding 20 per 100,000 population. Southern Europe and Japan have intermediate rates, and rates are low in Northern Europe and North America (Bosch et al, 2004; El-Serag, 2004; Ferlay et al, 2010). Several registries have shown that the incidence figures have changed slightly during the past several years. Vaccination plans against hepatitis B virus (HBV) and improvement in health standards have decreased the incidence in some high-risk areas, such as Taiwan (Chang et al, 1997; see Chapter 64). The incidence is decreasing in Japan, where the risk of HCC is linked significantly to chronic infection with hepatitis C virus (HCV) that was acquired decades ago, when health care was less concerned by blood-borne viral disease. By contrast, HCC incidence is increasing in the United States (El-Serag & Mason, 1999) and in Northern Europe (Bosch et al, 2004), probably reflecting the different timing in the appearance of risk factors, mostly HCV infection (Tanaka et al, 2002; see Chapter 64). Another major risk factor for HCC is alcohol intake, and this also varies widely in different countries (Morgan et al, 2004). Aflatoxin B1 uptake (Colombo & Donato, 2005), cigarette smoking, and heavy alcohol consumption are independent risk factors and may have synergistic effects (Kuper et al, 2000; see Chapter 64). Finally, recent studies have suggested that coffee intake reduces the risk (Bravi et al, 2007).
Table 80.1 Incidence of Hepatocellular Carcinoma According to Geographic Area and Distribution of Risk Factors
In all areas of the world, cirrhosis underlies HCC in almost 90% of the cases (Bruix et al, 2004; Fattovich et al, 2004). Cirrhosis is related to chronic HBV and HCV, which contribute to HCC development in 80% of cases. Alcohol consumption is a frequent cofactor, and surveys indicate that a significant proportion of HCC in cryptogenic cirrhosis is due to nonalcoholic fatty liver disease (Calle et al, 2003; Clark, 2006; El-Serag et al, 2001; Marrero et al, 2005; Regimbeau et al, 2004), a condition associated with diabetes or obesity (Day, 2002; Marchesini et al, 2003; see Chapter 65). Because of the obesity epidemic, this factor may gain greater relevance in the near future. Follow-up studies have disclosed that HCC development is now the most frequent cause of death in patients with cirrhosis (Benvegnu et al, 2004; Degos et al, 2000; Forner et al, 2009; Sangiovanni et al, 2006). Prevention, diagnosis, and treatment of this neoplasm are major areas of clinical and research activity.
The mean annual incidence of HCC in cirrhotic patients in the West is 3% to 4%, and this figure increases in parallel to liver function impairment (Benvegnu et al, 2004; Bolondi et al, 2001; Bruix & Sherman, 2011; Degos et al, 2000; Forner et al, 2009; Sangiovanni et al, 2006). Some specific characteristics have been associated with a higher risk. The most consistent are male sex; advanced age, this being a surrogate for the duration of the underlying liver disease; and increased α-fetoprotein (AFP) concentration (Bruix & Sherman, 2011). HBV and HCV coinfection imply a higher risk (Fattovich et al, 2004).
Data of patients coinfected with the human immunodeficiency virus (HIV) suggest that the incidence of HCC has increased in this population, liver function deteriorates more rapidly, and the HCC appears in younger ages and frequently in symptomatic, advanced stages (Brau et al, 2007) associated with poorer outcome (Puoti et al, 2004). In addition, several authors have reported higher potential of tumoral progression before and after liver transplantation in HIV/HCV-coinfected patients despite similar pathologic findings (Vibert et al, 2011). Further studies are needed to confirm whether HIV coinfection increases the HCC incidence, and if it is an independent prognostic factor. Irregular regeneration, high proliferative stage, and presence of dysplastic foci in biopsy samples also have been correlated with higher HCC incidence during follow-up, but the need for tissue sampling has prevented their robust validation and wide clinical acceptance (Bruix et al, 2004).
Risk Factors and Prevention
All types of cancer are the consequence of genetic changes that accumulate through time, ultimately conferring a survival advantage to cells with an abnormal phenotype. The relationship between chronic hepatitis and long-lasting liver damage with HCC development suggests that persistent inflammation is a key factor leading to cancer. Maintained injury and repair with high proliferation rate increase the risk for DNA mutation (Arbuthnot & Kew, 2001). In addition to genetic damage secondary to inflammation, HBV itself may have direct genetic or epigenetic effects, being able to integrate into the host genome (Brechot, 2004; Farazi & DePinho, 2006). This integration into the host genome is accompanied by rearrangement and increased mutagenesis that may affect crucial target genes (Farazi & DePinho, 2006; Ferber et al, 2003; Ryu, 2003; Villanueva et al, 2007). HCV is an RNA virus and does not integrate into host DNA (see Chapter 8C).
The most effective prevention of HCC is avoidance of viral infection or of toxin intake. Effective vaccination for HBV exists, and the vaccination plans in Taiwan have shown the efficacy of this action in diminishing cancer rates in children and adults (Chang et al, 1997). Proper controls in health care settings prevent HCV dissemination through blood transmission. Alcohol intake should be the target of health campaigns in the community; in addition, aflatoxin contamination of food can be prevented by avoiding grain storage in humid conditions. The epidemic of obesity with associated nonalcoholic steatohepatitis leading to HCC must be controlled by health education.
If viral infection is already present, it has been shown that a decrease in viral replication and subsequently of chronic hepatic damage could result in a reduced incidence of HCC in the long term (Liaw et al, 2004). Effective therapy is available for HBV and HCV, and prevention may be feasible for both agents; however, if chronic liver disease already has caused cirrhosis, it is unclear whether antiviral therapy has any preventive capacity, because cell damage and transformation may already be present (Bruno et al, 2007). Recent studies have shown that long-term interferon administration in does not reduce the risk of HCC in patients with cirrhosis (Bruix et al, 2011; Di Bisceglie et al, 2008). In patients with HBV infection, the genetic damage may occur in the absence of major liver insult, and this would explain the appearance of HCC in an otherwise healthy liver (Pollicino et al, 2004). This circumstance is more frequent in areas where viral transmission occurs early after birth or in childhood, stressing the benefits of HBV vaccination. Secondary prevention in patients with chronic liver disease requires the prior recognition of affected individuals, but currently no proof of the cost-effectiveness of population screening for viral liver disease has been offered, and this is not recommended (Bruix & Sherman, 2005; see Chapter 64).
Screening and Recall of High-Risk Individuals
A single randomized controlled trial performed in China recruited HBV patients to compare screening versus no screening; it showed the positive impact of screening on survival (Zhang & Yang, 1999). Unfortunately, such proof of efficacy is lacking in the West, and the establishment of surveillance for HCC in patients with cirrhosis is based on expert opinion (Bruix & Sherman, 2011; Bruix et al, 2001). The aim of screening is to detect HCC at an early stage, when the tumor is potentially curable (Bruix & Sherman, 2011; Forner et al, 2010).
Success of screening depends on several factors that include adequate ultrasound (US) performance, accurate diagnostic and staging criteria, and timely availability of curative treatments. If these requirements are not met, screening will never result in an increase of life expectancy. Even when accounting for the potential lead-time bias (i.e., patients apparently survive longer simply because their tumors were diagnosed earlier) and length-time bias (i.e., the interval between screenings misses the more aggressive tumors that will be detected when symptoms appear), it is assumed that screening results in more frequent detection of early stage HCC with more common application of effective treatment and improved outcome (Sangiovanni et al, 2004).
Effective treatment of HCC and long-term cure are feasible only with early detection; diagnosis at the symptomatic stage reflects an advanced stage and lack of effective treatment. Based on these arguments, the European Association for the Study of the Liver (EASL) panel of experts suggested abdominal US scanning and AFP measurements every 6 months for patients with liver cirrhosis who would be treated if diagnosed with HCC (Bruix et al, 2001). Because of the suboptimal accuracy of AFP (Marrero et al, 2009; Trevisani et al, 2001), the determination of this marker is no longer recommended, and screening is currently based on regular US (Bruix & Sherman, 2011). The screening recommendation excludes patients with severe associated conditions and those with advanced liver disease who are already candidates for transplantation. If this is not an option, screening for HCC aiming to detect early tumor will never be followed by treatment, and it makes no clinical sense.
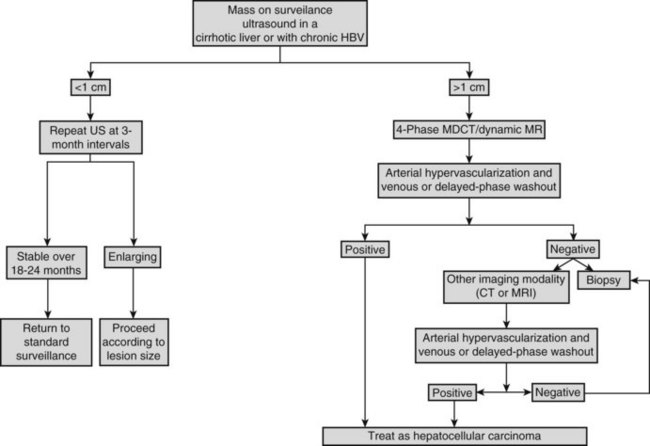
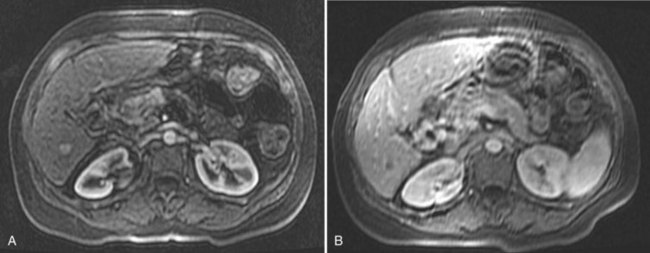
On detection of an abnormal finding, patients must be evaluated. Figure 80.1 depicts the recall strategy proposed by the American Association for the Study of Liver Diseases (AASLD) panel, and Box 80.1 summarizes proposed diagnostic criteria (Bruix & Sherman, 2011). Nodules measuring less than 1 cm in a cirrhotic liver may not correspond to a malignant focus in more than 60% of cases (Forner et al, 2008); even so, confident diagnosis is difficult with current diagnostic tools. Close follow-up to detect any increase in size is therefore recommended (Bruix & Sherman, 2005). Nodules in a cirrhotic liver that are larger than 1 cm can be diagnosed as HCC if the vascular profile on imaging techniques is characteristic of this neoplasm—that is, intense contrast uptake is seen in the arterial phase followed by contrast washout in the delayed venous phase (Bruix & Sherman, 2005; see Chapters 16 and 17). In a tumor larger than 1 cm in diameter, a single dynamic imaging technique can establish the diagnosis (e.g., computed tomography [CT] or magnetic resonance imaging [MRI]; Fig. 80.2). Contrast ultrasound is no longer recommended because it is not able to differentiate cholangiocarcinoma from HCC (Chen 2010; Vilana 2010). If the characteristic dynamic profile is not recognized by imaging techniques, it is recommended that the diagnosis be based on fine needle biopsy (Fig. 80.3; see Chapter 20). This is so even in the presence of increased AFP values, because cholangiocarcinoma and hepatic metastases may produce AFP (Bruix & Sherman, 2011; Rimola et al, 2009). It must be stressed that biopsy is not 100% sensitive, hence a negative result does not exclude HCC (Forner et al, 2008). Tumor staging is done through spiral CT and MRI (see Chapters 13, 16, and 17); MRI is the most sensitive to detect nodules smaller than 2 cm (Burrel et al, 2003). The technical advances in these techniques have made angiography unnecessary for diagnosis and staging (see Chapter 19). Other tumor markers, such as protein induced by vitamin K absence (Marrero et al, 2009), glypican-3 (Capurro et al, 2003), and AFP fractions (Marrero et al, 2009), have been proposed to be used alone or in combination with AFP, but their usefulness in routine clinical practice has not been established (Bruix & Sherman, 2011).
Box 80.1
Diagnostic Criteria for Hepatocellular Carcinoma
Noninvasive criteria (cirrhotic patients)
Focal lesion >2 cm: one imaging technique (e.g., dynamic computed tomography, magnetic resonance imaging) with arterial hypervascularization and venous washout
From Bruix J, Sherman M, 2011: Management of hepatocellular carcinoma: an update. Hepatology 53:1020-1022.
Staging and Prognostic Evaluation
Staging systems are crucial tools to stratify patients into different subgroups according to prognosis. Ideally, they should establish a link with treatment and predict outcome after therapy. A well-validated and internationally accepted staging system is lacking for HCC. In most tumors, the prognosis is related mainly to tumor stage at the time of diagnosis. The presence of underlying liver disease makes prognostic evaluation of patients with HCC more complex, because liver disease influences outcome and the possibility of treatment (see Chapter 2, Chapter 70A, Chapter 70B
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree