Chapter 38 FASCIA LATA SLING
Pubovaginal sling cystourethropexy is considered one of the gold standards of female stress urinary incontinence (SUI) surgery. The bladder neck sling can be performed using autologous, allograft, or synthetic material. The two autologous methods use either rectus or fascia lata fascia. This chapter summarizes the literature on the indications, surgical technique, and outcomes of using fascia lata to treat female SUI.
HISTORY
The autologous sling used to affect the urethra as a method of urinary control was first mentioned in 1907 by Von Giordano, who described the use of gracilis muscle to wrap around the urethra.1 The pyramidalis muscle was used to suture behind the urethra by Goebell in 1910.2 Squier referred to the use of the levator ani muscles to augment bladder outlet resistance.3 In 1942, Aldridge described the use of bilateral strips of rectus fascia passed inferiorly through the rectus abdominis muscles to support the urethra posteriorly.4 In 1978, McGuire and Lytton described using an autologous fascial sling in 52 patients with SUI.5 Forty-two of the patients had had previous corrective incontinence surgery. Postoperative urethral pressure measurements showed urethral pressure increase but no occlusion. Fifty patients had a successful outcome, and there were 2 failures. These results were further confirmed by Blaivas and Jacobs in 1991, when they observed 67 patients who had undergone a pubovaginal fascia sling repair and reported that 82% were completely dry and 9% were dramatically improved.6
The fascia lata sling was first described by Price in 1933.7 Beck and colleagues examined the use of fascia lata as a suburethral sling for the treatment of recurrent urinary incontinence.8–10 They first described the use of the fascia lata sling in 19748 and in 1988 reported their results in 170 patients.10 They used the fascia lata suburethral sling after failed primary incontinence surgery in 69 cases and after multiple failed prior anti-incontinence repairs in 101 cases. The overall success was reported at 98.2%.10
Cadaveric fascia lata (CFL) has been used to repair tissue defects for a number of years, but its first use for incontinence surgery was reported in 1996.11 Handa and colleagues used CFL in 16 patients, 14 with genuine SUI and 2 who had a previously placed synthetic graft that became infected. Eighty-eight percent were subjectively improved, and 79% of the patients with genuine SUI were objectively improved.11
The use of xenograft material for sling surgery has been reported as well. In 2002, a pilot study examined the use of bovine pericardium placed as a suburethral sling in 22 females.12 At 20 months, 21 of 22 patients were cured with no incidences of erosion or rejection. Another study recently looked at the use of processed porcine small intestine submucosa (SIS) as a pubovaginal sling in 152 female patients with SUI.13 In this 4-year series, there was a 93.4% cure rate, with three additional patients demonstrating marked improvement. There was no erosion, infection, or rejection in the group.
Synthetic materials have been used for incontinence since 1961, with the use of nylon strips fashioned into a suburethral sling.14 Numerous materials have been tried since then, including Silastic,15 Gore-Tex,16 Mersilene,17 Marlex,18 Prolene,19 and Vicryl.20
INDICATIONS
ISD has been associated with multiple etiologies. The presence of previous SUI corrective surgeries is a risk factor. A neurologic insult to the sacral area may result in compromise of the bladder’s innervation and ISD. This insult could come from damage to the nerve roots as they leave the spine or via major pelvic surgery such as a radical hysterectomy. In addition, patients with diabetic neuropathy may also have peripheral neurologic problems interfering with sphincteric function. Patients who have undergone previous radiation treatment are at risk of developing ISD-based SUI.21 Another possible cause of ISD is estrogen deficiency: as women age, lower levels of estrogen may predispose to ISD.22 Horbach and Ostergard examined 263 consecutive patients with urodynamic studies for urinary leakage.23 In this population, 132 women had ISD, defined as urethral closure pressure lower than 20 cm H2O, and the only independent risk factor for ISD was age greater than 50 years.
ISD is classically diagnosed using video urodynamics, which shows an open bladder neck at rest. In addition, a low Valsalva (abdominal) leak-point pressure (VLPP) is seen (<60 cm H2O) in patients with ISD. Incontinent women with a VLPP greater than 90 cm H2O usually have type 2 SUI according to fluoroscopic assessment.24 There is also correlation between worsening SUI subjective symptoms and a lower VLPP.25 Urethral pressure profilometry has been used to define SUI in women. Weber performed an extensive review of published articles using urethral pressure profilometry and found the technique to be nonstandardized and poorly reproducible.26
The recommendations from the American Urological Association clinical guidelines panel summary report published in 1997 suggested that the standard patient should be informed about the four major surgical correction categories: retropubic suspension, transvaginal suspensions, anterior repairs, and sling surgeries.27 According to the panel, the patient should understand that retropubic suspensions and sling surgeries carry a higher complication rate along with their higher rate of longer-term cure. The panel suggested categorizing patients as having urethral hypermobility, ISD, or both, although they acknowledged that there was no standardized method to make this diagnosis. These recommendations decreased the popularity and use of needle suspensions and anterior repairs as treatment options for women with SUI. In May 2007 the Urinary Incontinence Treatment Network, led by Albo and colleagues, published a large randomized study to examine the differences between a Burch colposuspension and an autologous fascial sling.28 Six hundred fifty-five women were randomized to either the Burch or a fascial sling and followed for 24 months with standardized assessments. Seventy-nine percent of the women completed the assessments. Success rates were higher for the women who underwent the sling procedure versus the women who had a Burch (47% vs. 38%, p = 0.01). Specific to stress incontinence, there was a 66% success rate in the sling group whereas the Burch group had a 49% success rate. Complications were higher in the sling group, with more urinary tract infections, voiding difficulty, and postoperative urge incontinence. Performance of prolapse repair with the stress incontinence surgery did not affect the outcomes between the groups.
The recommendation to apply a particular surgery depending on the type or degree of SUI was suggested by Sand and associates in 1987.29 They found a 54% failure rate in patients who underwent a Burch procedure and who had a mean urethral closure pressure (MUCP) lower than 20 cm H2O, whereas the patients with a MUCP greater than 20 cm H2O had an 18% failure rate. Koonings and coworkers compared 19 women with SUI and low urethral pressure to 106 patients with SUI and a normal urethral pressure.30 The patients underwent a modified Pereyra procedure or a Burch colposuspension. The women with low urethral pressure preoperatively had a higher failure rate, regardless of the surgical procedure used, compared to patients with normal urethral pressure profilometry. These data suggest the limitations of colposuspension and needle suspensions but do not limit the indication of the pubovaginal sling.
Several studies have examined the use of pubovaginal slings in groups of female patients with type 2, type 3, or a combination of both SUI types. Kreder and Austin examined 28 female patients with SUI.31 Eleven had ISD alone, and 16 had a combination of urethral hypermobility and ISD. The diagnostic definitions were obtained with the use of multichannel video urodynamics. ISD existed if the VLPP was lower than 60 cm H2O and the bladder neck was open at rest. Urethral hypermobility was defined via a pelvic examination with the patient performing a Valsalva maneuver and a cough. Overall, the patients in the sling group (17 with rectus fascia and 11 with fascia lata) had a 70% dry rate at an average of 22 months. The patients with ISD alone did poorly, with a 55% dry rate, whereas the patients with combination type 2/3 SUI had an 81% dry rate. One patient in the ISD group had a complete surgical failure.
In another study, by Zaragoza, 60 consecutive female patients with SUI were examined with multichannel video urodynamics.32 ISD was defined in 22 patients, who had a VLPP lower than 60 cm H2O (mean, 42 cm H2O), and 38 patients diagnosed with type 2 SUI had a mean VLPP of 95 cm H2O. At an average of 25.1 months after a rectus fascia pubovaginal sling procedure, 95% of the patients were completely continent. The three failures were due to urgency incontinence, which was present in all three patients preoperatively. The success rate was almost identical between the patients diagnosed with type 2 and those with type 3 SUI.
Finally, Cross and colleagues examined a large group of female patients diagnosed with SUI, all treated with a rectus fascia pubovaginal sling.33 There were 134 patients who were contacted at an average of 22 months after surgery, and their initial diagnosis was type 2 SUI (45 patients), type 3 SUI (73 patients), or a combination of both (16 patients). The overall dry rate was 93%, as confirmed by video urodynamics: 96% of the type 2 patients, 89% of the type 3 patients, and 100% of the type 2/3 patients.
CHOICE OF FASCIA LATA SLING AND SOURCE
Autologous rectus and fascia lata are commonly used for pubovaginal slings as well as allogenic fascia. The advantage of autologous material is the lack of potential disease transmission and low erosion rates. Fascia lata offers advantages over rectus fascia such as greater tensile strength and lack of abdominal wall hernias related to graft harvest; moreover, it is rare that the patient’s past surgical history would interfere with harvesting of a long strip of fascia lata.34 The harvesting of autologous fascia adds operative time and possible morbidity to sling surgery, compared with synthetic and allograft fascia sources.
Allograft fascia lata is from a cadaveric source and has been used in sling surgery since 1996.35 There is a theoretical advantage for the use of CFL compared with a synthetic source in that there is a lower risk of erosion, whereas the disadvantage is a chance of disease transmission.36 Choe and Bell examined 16 allograft sources of tissue, including 8 freeze-dried gamma-irradiated CFL allografts.37 Fourteen of the allografts contained DNA material, as determined from standardized DNA extraction techniques. FitzGerald and colleagues found donor antigens still present in CFL prepared via the freeze-dried and the Tutoplast techniques.38 Hathaway and Choe took four commercially available human cadaveric allografts and subjected 10 of each type to DNA extraction. Thirty-nine (97.5%) of the allografts contained DNA with variable amounts.39 The significance of the presence of DNA and antigens in the allograft material is unknown. The risk of contracting Creutzfeldt-Jacob disease from allograft material is approximately 1 in 3.5 million,40 and the risk of human immunodeficiency virus (HIV) transmission is estimated to be 1 in 8 million.41
CFL is mainly made via two techniques: solvent dehydration with gamma irradiation (Tutoplast) or freeze-drying (tissue banks and FasLata). Both of these preparations require 15 to 30 minutes to rehydrate in saline before implantation.36 A study by Lemer and associates examined the tensile strength of two types of CFL (solvent-dehydrated and freeze-dried), autologous rectus fascia, and cadaveric dermal grafts.42 They found no significant difference between solvent-dried fascia lata, dermal graft, and autologous rectus fascia. Freeze-dried fascia lata had a significantly lower maximum-load-to-failure rate than either of the other types tested. In contrast, Sutaria and Staskin examined the tensile strength after three methods of CFL preservation (freeze-dried/gamma-irradiated, freeze-dried, and solvent-dehydrated/gamma irradiated).43 They found no significant difference in the tensile strength among the three CFL preparations. Choe and colleagues studied the biomechanical properties of full-strip slings and patch-suture slings using autologous tissue, cadaveric tissues, and synthetic materials.44 For the full-strip slings, cadaveric tissues (including CFL) had the best tensile strength, followed by synthetics and then autologous tissue. In the analysis of patch-suture slings, the synthetic material was the strongest, followed by CFL and then autologous rectus fascia. Dora and coworkers examined the biomechanical properties of several sling materials in a time-dependent fashion.45 They placed the materials in rabbit models and tested them before implantation and again at 12 weeks. The tensile strength of polypropylene mesh and autologous fascia remained consistent with baseline. CFL and porcine xenografts decreased in tensile strength by 60% to 89%. Walter and colleagues further reviewed the change in tensile strength of three lots of freeze-dried CFL after implanting it into a rabbit model.46 They demonstrated significant preimplantation variability in the tensile strength of the CFL. At 12 weeks, the samples were explanted and retested and showed a 90% decrease in tensile strength. The significance of variations in tensile strength among the various sling materials has not been shown to be clinically significant. Based on the earlier failure of some sling materials, the presence of tensile strength variations and degradation may play a role in long-term clinical success.
CADAVERIC FASCIA LATA OUTCOMES
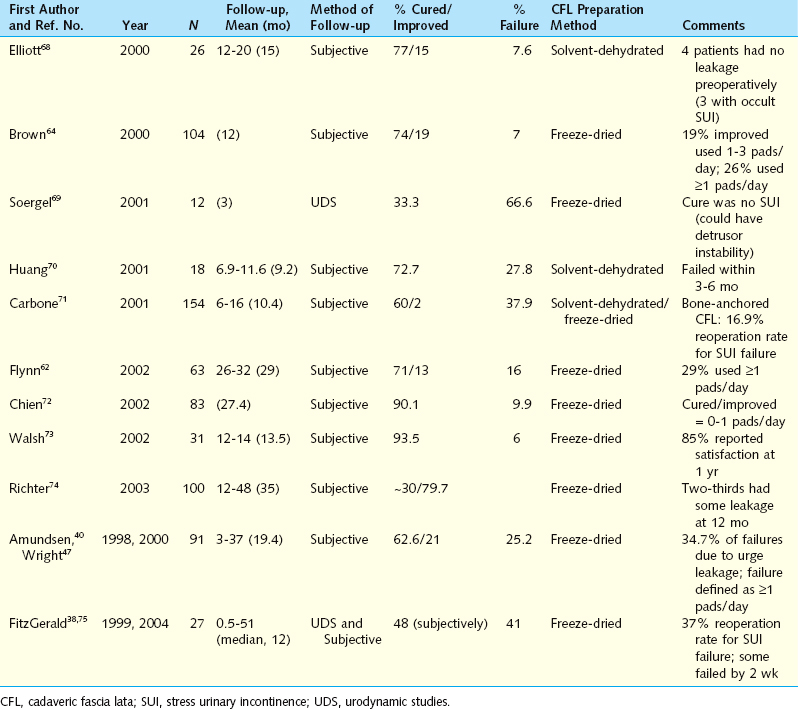
Using CFL for pubovaginal slings offers the advantage of decreased operative time and less patient morbidity.47 The results from performing the procedure are less favorable than with their autologous counterparts. Table 38-1 displays the results of several series of patients undergoing CFL sling cystourethropexy. Overall, the follow-up periods were short and most studies used subjective assessment methods with varying definitions of postsurgical success. Numerous studies showed high failure rates using CFL slings, with postsurgical SUI rates ranging from 6% to 66.6% as early as 3 months postoperatively.
O’Reilly and Govier examined eight patients who had recurrence of SUI after CFL sling placement.48 Five of the eight patients underwent urodynamic studies, which demonstrated ISD with lower VLPPs than the patients had had preoperatively. No other reason for failure was identified.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree