EPIDEMIOLOGY OF ACUTE KIDNEY INJURY
Acute kidney injury (AKI) is a complex syndrome associated with several etiologic factors. AKI occurs in a variety of settings with clinical manifestations ranging from a minimal elevation in serum creatinine (SCr) to anuric kidney failure.
1,
2,
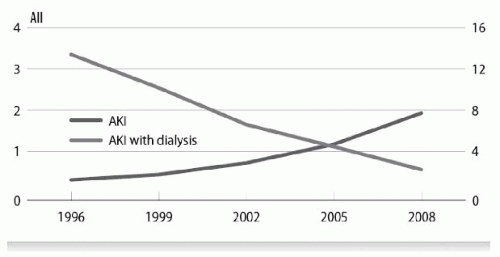
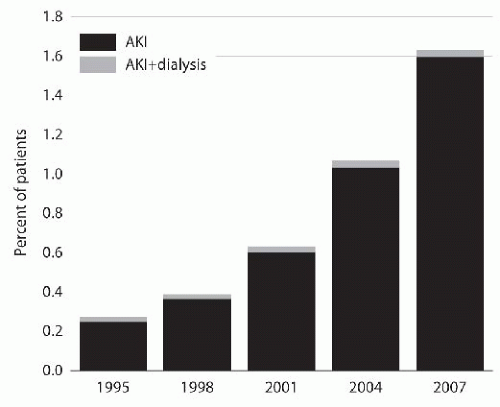
3 The incidence of AKI with and without need for dialysis has been progressively increasing in the last 15 years
4 (
Fig. 28.1) and is more pronounced in older hospitalized patients. Data from the USRDS shows that 1.6% of patients age 66 and older who were continuously enrolled in Medicare inpatient/outpatient had the diagnosis of AKI
4,
5 (
Fig. 28.2). The increasing incidence can also be explained by the growing awareness by the medical community about AKI as a main risk factor for mortality and an important contributor for chronic kidney disease (CKD).
The reported incidence of AKI is widely variable in different regions of the world (
Table 28.1). In the developed world AKI is seldom a community-acquired disease; the condition develops primarily in hospitalized patients. In these regions the incidence of hospital-acquired AKI exceeds that of community-acquired AKI by 5 to 10 times, having an estimated yearly incidence of 0.15% to 7.2%.
6 In the developed world more than 20% of AKI cases occur in the intensive care unit (ICU) setting. On the other hand, in the developing world a number of cases of AKI are found in rural areas. The demographics, etiologies, and outcomes of AKI in rural settings differ from those in more developed areas. In rural regions AKI is caused predominately by snake, spider, caterpillar, or bee envenomations, or by specific infections such as leptospirosis, tetanus, or severe malaria. Those patients are managed by primary caregivers who have limited resources.
The age of AKI patients is another difference between developed and developing countries. Elderly patients predominate in the developed world whereas in the developing world AKI is generally a disease of the young. Children are more often affected in developing countries, constituting more than 15% of patients in some studies. Age differences might partially account for differences in reported survival rates between the developed and developing countries. Paradoxically, patients in developing countries might have a better chance of survival. Here younger patients develop AKI as a result of a single disease (e.g., leptospirosis or malaria) rather than from multiple organ failure, but are more prone to complications secondary to poor nutrition and resource availability (
Table 28.1).
The first step for the nephrology community to better understand and quantify the clinical importance of AKI was to develop a uniform definition. Before 2004 acute renal failure (ARF) had no accepted definition resulting in more than 30 different definitions reported in the literature.
7 Consequently, epidemiologic studies used different clinical and physiologic endpoints making it difficult to compare the results between studies (
Table 28.1). This lack of a uniform definition yielded discrepancies in AKI incidence, prevalence, and outcomes in various clinical settings. The reported incidence ranged from 1% to 31% and mortality from 28% to 82%.
8,
9,
10 The formation of the Acute Dialysis Quality Initiative (ADQI) group in 2000 was the beginning of a process to establish consensus and evidence based guidelines in ARF. In 2004, ADQI formulated the Risk, Injury, Failure, Loss, and End-stage Kidney (RIFLE) classification for ARF.
7 The RIFLE classification system provides three grades of severity for AKI based on the maximal increase in SCr or decrease in urine output from the baseline condition: injury—risk (class R), injury (class I), and failure (class F)—and two outcome classes (loss and end-stage renal disease [ESRD]) (
Table 28.2).
The clinical predictive ability of the RIFLE classification has been extensively validated in large general and ICU populations.
8,
11,
12,
13,
14,
15 In a single center retrospective cohort study, including 5,383 patients admitted during a 1-year period, the incidence of AKI was 67.2%. In that cohort AKI was associated with an increased risk for hospital mortality compared to those who never developed AKI. The higher incidence that might have been considered unusual before the RIFLE era was confirmed in other studies that applied the criteria.
8,
11,
12,
13,
14,
15The ADQI group, associated with representatives of nephrology societies (ASN, ISN, and NKF) and the European
Society of Intensive Care Medicine, created the Acute Kidney Injury Network (AKIN) as an independent collaborative network intended to facilitate international, interdisciplinary, and intersocietal collaborations.
16 One of the tasks proposed by the AKIN was to further refine the AKI definition. In 2007, a modified version of the RIFLE classification was published, also known as the AKIN classification (
Table 28.2).
17 The terms Risk, Injury, and Failure were replaced by stages 1, 2, and 3, respectively. An absolute increase in creatinine of at least 0.3 mg per dL was added to stage 1. Patients starting renal replacement therapy (RRT) are automatically classified as stage 3, regardless of their SCr or urine output. The outcome categories Loss and ESRD were eliminated. Another difference between RIFLE and the AKIN classification is the 48-hour time frame within which the diagnosis of AKI is made, “AKIN criteria … change in creatinine should occur within 48h.”
17 However, after the diagnosis is established, staging should be applied with no time frame constraint. Additionally, the glomerular filtration rate (GFR) criteria were eliminated.
In the RIFLE and AKIN classification systems patients are classified based on the worst category achieved. This is intended to describe the change or trend in AKI severity over time. Several studies in the ICU population have validated the concept verified in clinical practice that patient outcome progressively worsens with the maximal severity of AKI achieved.
8,
12,
14,
18,
19,
20 Over 71,000 patients were included in published studies with the RIFLE classification system; these studies showed a stepwise increase in relative risk (RR) for death going from Risk (RR: 2.40) to Injury (RR: 4.15) to Failure (6.37).
21 Osterman and Chang
22 performed a retrospective analysis of a database of 41,972 patients admitted to ICU. AKI based on RIFLE occurred in 35.8% of patients: 17.2% Risk, 11% Injury, and 7.6% Failure. Patients with Risk, Injury, and Failure had a hospital mortality of 20.9%, 45.6%, and 56.8%, respectively, compared to 8.4% among non-AKI patients. Abosaif et al.
23 retrospectively applied the RIFLE classification in order to evaluate its sensitivity and specificity to predict renal and patient outcomes in 183 critically ill patients with AKI. Mortality rate in the ICU (60 days, 74.4%) and 6-month mortality rate (86%) were significantly greater in the RIFLE-Failure group compared with all groups. Cruz et al.
15 conducted a prospective multicenter study in 19 ICUs in northeastern Italy. Of 2,164 ICU patients who were admitted during the study period, 234 (10.8%) developed AKI whereas 3.3% were treated with RRT. Of the AKI patients, 19% were classified as Risk, 35% as Injury, and 46% as Failure. Overall, ICU mortality was higher among those in RIFLE class Failure (49.5% vs. 20% in R, 29.3% in I).
Hoste and colleagues
14 performed a retrospective singlecenter study on 5,383 patients admitted during a 1-year period in seven ICUs. AKI occurred in 67% of ICU admissions, and 12% reached a maximum RIFLE class of Risk, 27% Injury, and 28% Failure. Interestingly, among the patients that reached a level of Risk, 56% progressed to either Injury or Failure. Patients with maximum RIFLE class Risk, Injury, and Failure had hospital mortality rates of 8.8%, 11.4%, and
26.3%, respectively, in contrast to 5.5% in non-AKI patients. RIFLE classes were still associated with hospital mortality after adjusting for multiple covariates (baseline severity of illness, case mix, race, gender, and age). These findings showed that patients with RIFLE-Risk are indeed at significant risk of progression to more severe AKI. Patients with RIFLE class Injury or Failure incur a significantly increased length of stay and an increased risk of in-hospital mortality compared with those who do not progress past Risk or those who never develop AKI.
The increasing severity of illness in critically ill patients with AKI is one of the contributors to the persistently high mortality rate associated with this syndrome. Observational studies suggest that critically ill patients with AKI are increasingly older, have more comorbid diseases, have a higher incidence of septic, and have greater severity of illness and organ failure scores. Two severity of illness scoring systems are widely used: the Acute Physiology and Chronic Health Evaluation (APACHE) score and the Sequential Organ Failure Assessment (SOFA) score.
24,
25 Although patients developing AKI have shown a significant decrease in mortality rate in the last decade,
25,
26,
27 the persistently high mortality in patients with multiorgan system failure remains a challenge. This emphasizes the need for early assessment and intervention in all cases of AKI. It is apparent that AKI is associated with substantial morbidity, mortality, and cost.
The financial costs of AKI are high. Fischer et al.
28 performed a multicenter analysis in 23 Massachusetts hospitals for a 2-year period (1999 to 2000). They identified 2,252 records of patients hospitalized with uncomplicated ARF. Patients hospitalized with uncomplicated ARF incurred median direct hospital costs of $2,600, median hospital length of stay (LOS) of 5 days, and mortality of 8%. Dialysis was independently associated with significantly greater hospital costs and LOS for patients with uncomplicated ARF (
P <0.05) compared to patients with other commom medical diagnoses.
28The cost of RRT for patients with AKI is high; however, information on the costs of the three dialytic techniques for AKI is minimal. In a Canadian ICU, the cost of dialysis was $3,486 to $5,117 (Canadian) per week for continuous renal replacement therapy (CRRT).
29 For intermittent hemodialysis (IHD), major costs include the need for supervision by a trained dialysis nurse, which can become an economic issue if IHD is performed on a frequent or daily basis. For CRRT, major costs include disposables and replacement fluids. Most investigators have found that CRRT costs are somewhat greater than IHD.
30,
31 An evaluation of total hospital costs
32 showed that from the start of RRT to hospital discharge patients on CRRT total cost was US$57,000 more than that for those on IHD. A recent cost analysis of the of RRT for patients with AKI estimated that mean adjusted total costs were U$1,342/week for IHD compared to U$3,486/week for CRRT
30 and no difference was found in the outcome, renal recovery at hospital discharge. However, there was a nonsignificant statistical trend toward enhanced renal recovery in the CRRT group despite a significantly lower mean arterial pressure and a trend toward higher Acute Physiology and Chronic Health Evaluation (APACHE) II scores. Considering that nonrecovery of renal function would adversely affect quality of life, a modality that enhanced the rate of renal recovery would offer an important advantage, even if there were no difference in survival across modalities.
Parameters for Acute Kidney Injury Diagnosis
Serum Creatinine
Therapeutic interventions are generally based on an evaluation of clinical data and diagnostic information. In AKI the importance of timing of diagnosis was poorly appreciated as therapeutic interventions have generally been lacking, and the small number of studies reported have failed to improve
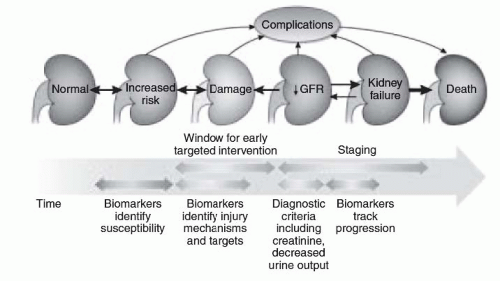
outcomes. In the last decade, the concept of interventions based on “windows of opportunity” coupled with targeted therapy became evident in other ischemic events such as acute chest pain syndromes and stroke. The importance of finding early diagnostic information in AKI has since been highlighted and the development of technology has facilitated the search for new biomarkers of kidney injury.
The current criteria for AKI diagnosis and classification, RIFLE and AKIN, are still based on SCr incremental concentrations and decreased urine output. Many characteristics other than renal function, such as age, muscle mass, cata bolic rate, and race, influence SCr concentrations. In addition, SCr levels depend not only on renal elimination but also on creatinine generation and volume of distribution.
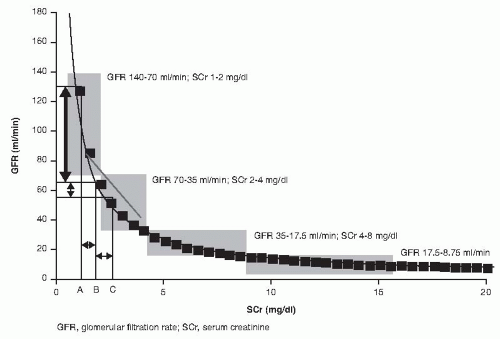
33 Given the exponential relation of SCr and GFR, significant decreases in GFR are reflected as small increases in SCr in the early phases of injury (
Fig. 28.3).
In a steady state setting a reasonable approximation is that each time the GFR halves, the SCr concentration doubles. Thus, steady state GFRs of 100, 50, 25, 12.5, and 6.25 mL per minute are associated with increasing SCr concentrations ranging from 1 to 16 mg per dL; however, there is wide range depending on the level of extrarenal clearance that is accentuated as kidney function declines and may contribute to up to 40% of total clearance. AKI often occurs in a nonsteady state in which the three determinants of SCr concentration (production, volume of distribution, and renal elimination) fluctuate.
33 Computerized models derived from AKI patients demonstrate that several patterns of change in GFR occur during development and recovery from AKI. These GFR changes are poorly reflected by daily changes in SCr concentration.
33 Moreover, the rise in SCr that occurs in AKI is a post facto finding. In critically ill patients, a nonsteady state condition and the positive cumulative fluid balance enhances the insensitivity of SCr as a parameter of renal dysfunction.
34 Hoste et al. showed that in a group of recently admitted ICU patients with normal SCr the 1-hour urinary creatinine clearance revealed values lower than 80 mL/min/1.73 m
2 in 46.2% of the patients.
35 These data suggest that SCr is not a reliable tool to detect even moderate kidney dysfunction in AKI patients.
Blood Urea Nitrogen
Blood urea nitrogen (BUN) is also used as a parameter to evaluate renal function. However, elevations in BUN level are often, but not always, due to a decrease in GFR. Some factors enhance urea production, such as gastrointestinal bleeding, corticosteroid therapy, and high-protein diet. In conditions of decreased intravascular effective volume like decompensated heart failure, increases in BUN are not proportional to the rise in SCr level and fall in GFR. The usual BUN:SCr ratio is about 10:1 and the BUN and SCr increase by 10 to 15 and 1.0 to 1.5 mg/dL/day, respectively, in the absence of GFR. Increases in the basal metabolic rate that occur with fever or glucocorticoid administration enhance these daily rates. Although an increase in the BUN/
creatinine ratio has been used for many years to help to differentiate between prerenal and renal azotemia, BUN can increase independently from SCr in situations characterized by decreased glomerular perfusion pressure such as heart failure. Some studies have already shown elevations in BUN independently from SCr levels and demonstrated that these two parameters are not only a reflection of the severity of renal dysfunction, but rather the consequence of two distinct pathologic processes.
36,
37,
38 The activation of renin-angiotensin-aldosterone system (RAAS) and sympathetic nervous system is responsible for decreasing the glomerular perfusion pressure and GFR. The increment in vasopressin levels upregulates aquaporin-2 and urea transporter expressions and increases water and urea reabsorption. Urea, in contrast to SCr, is not secreted but reabsorbed by the renal tubules. The increased reabsorption of sodium and water, rather than the reduced GFR, enhances reabsorption of urea and increases BUN levels. Thus, BUN levels and BUN/creatinine ratio could be a more effective way to assess circulatory volume than GFR, which is regulated by the pressure difference between glomerular afferent and efferent arterioles.
39 In heart failure and possibly in other settings where underfilling is part of the physiopathologic process, the rise in BUN greater than any fall in GFR is a marker of the neurohumoral axis activation.
40In selected circumstances, it may not be clear if an elevated BUN:SCr ratio is due to an acute or chronic process. In this circumstance review of previous records is helpful.
Oliguria
Although the hydration status, osmolar excretion, as well as a large dose of diuretics will influence urine volume, and severe AKI can occur with normal urine output, the urinary flow rate also may provide helpful information about the cause of AKI. Sustained periods of anuria suggest urinary tract obstruction as the cause of AKI. Other rare causes of anuria include rapidly progressive glomerulonephritis, mechanical occlusion of renal blood flow, and diffuse renal cortical necrosis. Nonoliguric varieties of AKI are common, ranging from 25% to 80% of all cases of AKI with 33% nonoliguric at AKI diagnosis.
41,
42,
43 The nonoliguric state may be present in all types of AKI including those following surgery, trauma, hypotension, nephrotoxins, and rhabdomyolysis. Several factors may contribute to the devlopment of nonoliguric AKI: use of volume expansion, high-dose potent diuretic agents, high osmolar loads, and renal vasodilators. Another contributory factor is aggressive fluid resuscitation and improved supportive management of critically ill patients. However, nonoliguria may mask the underlying severity of AKI and lead to delayed recognition.
The definition of oliguria changed after the RIFLE and AKIN classification system. The AKIN group proposed the hourly measurement of urine volume, providing an opportunity to treat urine flow as a continuous rather than as an interval physiologic variable, with more time points for the diagnosis of oliguria and detection of AKI. Although fluctuations in this parameter can result from external influences, such as drug administration, the pattern of change can be detected earlier with more frequent observations. Experimental studies conducted in laboratory models of AKI, as well as clinical studies, have clarified the pathophysiologic aspects underlying the variations of urine flow rate in AKI.
44,
45 In a study of 25 patients with predominantly renal ischemia-associated AKI, Rahman and Conger found that the urine flow rate strongly correlated with residual GFR.
45 In that study urine flow rate did not correlate with selected aspects of renal tubular function such as urine:SCr or the fractional excretion of sodium.
However, because of the difficulties in measuring and recording the hourly urine output, the AKIN oliguria criterion was evaluated in fewer studies than the SCr criterion. Of the studies evaluating the urine output criterion most were retrospective and used a modified definition of evaluating urine volume in 2 to 12 or 24 hours and shortening the time of observation to the first 24 hours of ICU admission or postoperative period. In a retrospective study Barrantes et al.
46 evaluated the outcomes of hospitalized patients classified by the AKIN criteria, using both the urine output (UO) and SCr criteria in the first 48 hours after ICU admission. Comparing patients that developed AKI using the UO, SCr, or both criteria, they found that UO criterion did not affect the AKI associated mortality. Joannidis et al.,
47 using the SAPS 3 database of ICU patients, assessed 24-hour urine volume for 48 hours. Patients with AKI defined by the lowest urine volume had higher mortality compared to non-AKI patients. In a prospective cohort of cardiac surgery patients, Haase et al.
48 subclassified the AKI patients based exclusively in the UO criterion (over a 48-hour period). Only those patients that reached a RIFLE-F or AKIN stage 3 had significantly longer ICU and hospital lengths of stay and a higher mortality rate (compared to non-AKI patients and those with maximum RIFLE-R/I and AKIN stage 1/2). In a systematic review,
21 the relative risk for death among studies that used both SCr and UO criteria was lower than in those using only the SCr criterion. Hoste and Kellum,
19 in a review including 10 studies, showed that patients in the RIFLE Risk class defined by the SCr criterion were more severely ill than those in the same class defined only by the UO criterion. Hoste et al.
14 also observed that patients in Failure based on the RIFLE GFR criterion had a slightly higher mortality than those in Failure based on the UO criterion. In a study by Cruz et al., RIFLE classes (using creatinine and UO criteria together) were the strongest predictor of ICU mortality in multivariable analysis.
15 In that study, the analysis was based only on the SCr criterion; the RIFLE class was sustained as an independent predictor of ICU mortality but with inferior statistical power. Based only on the UO criterion, RIFLE class did not emerge as an independent predictor.
These clinical observations, and a large body of experimental data, suggest the residual level of GFR is the primary determinant of urine flow in patients with AKI. The higher level of residual GFR in nonoliguric patients is
compatible with improved survival and lower morbidity in these patients. However, one needs to be aware that there is a distinction between spontaneous nonoliguria from diuretic induced urine flow with respect to underlying GFR and nonoliguria does not automatically imply a higher GFR.
Biomarkers
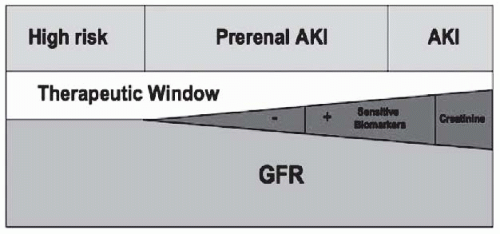
The importance of early detection of AKI has been emphasized as an earlier diagnosis would provide a wider window to perform supportive and therapeutic interventions (
Fig. 28.4). The recognition of the insensitivity to detect AKI by the most commonly used surrogates of kidney function (SCr and UO) has led to extensive efforts to identify alternative biomarkers for AKI diagnosis, including urine and serum biomarkers. Compared to the use of more sensitive biomarkers of kidney injury, SCr delays the AKI diagnosis by 48 to 72 hours.
49,
50,
51,
52 Most of the studies have focused on the ability of these biomarkers to detect AKI earlier than the classical parameters, but biomarkers may be also useful to predict the course and prognosis of AKI (
Fig. 28.5). Several promising candidates have emerged, demonstrating reasonable diagnostic performance for AKI up to 48 hours prior to a significant change in SCr.
53,
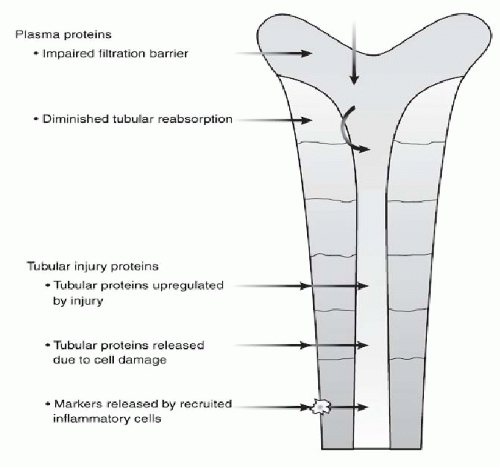
54 Although the commercial platforms are becoming available for research use, the knowledge for the clinical application, utility, and diagnostic value of these early biomarkers remains to be validated. These new candidates are being tested in different AKI clinical scenarios. The different abilities to detect renal injury and estimate GFR are associated with their pathophysiology (
Fig. 28.6). AKI biomarkers differ on the basis of how they
enter the urine, either through filtration, upregulation, and secretion or through leakage from damage of a constitutive marker. The time sequence and magnitude of biomarker elevations are unique and may depend on the nature and severity of injury.
In addition to potentially facilitating an earlier diagnosis, these biomarkers will also allow for retesting preventive and therapeutic drugs that have failed in clinical trials that used SCr as a parameter to guide intervention. Another area of possible application for the new biomarkers is assisting in decisions to initiate RRT in patients with AKI. The main biomarkers being tested and some clinical data on their use are summarized below.
Cystatin C
Cystatin C is a low molecular weight protein produced at a constant rate by all nucleated cells. Because of its low molecular weight it is freely filtered at the glomerulus and reabsorbed and catabolized, but not secreted by the renal tubule. Plasma levels correlate with GFR and, unlike creatinine, are not significantly affected by age, gender, race, or muscle mass. In human studies, both pCyC and uCyC have been shown to predict AKI, although its superiority over SCr has not been a universal finding.
55,
56,
57 Recently, Herget-Rosenthal et al.
58 compared cystatin C with creatinine for the diagnosis of AKI in a series of 85 patients. Increases in serum levels of cystatin C were detectable 1 to 2 days earlier than comparable changes in SCr. In another study measuring plasma Cr (PCr) and pCyC in 444 adults on ICU admission, of whom 124 already had AKI on entry, pCyC moderately predicted death or RRT (area under curve [AUC] 0.61, 95% confidence interval [CI], 0.53-0.68) and performed similarly as PCr (AUC 0.60, 95% CI, 0.51-0.67).
59 In a cohort of 151 AKI patients from a small multicenter study, Royakkers et al.
60 used the RIFLE classification system to define AKI and compare the performance of sCyC and uCyC as early biomarkers for AKI. Urinary CyC had no diagnostic value during the days prior to AKI diagnosis by SCr (AUC <0.50). In addtion, sCyC and uCyC determined on the first day of AKI diagnosis were poor predictors for the need for RRT (AUC =0.66).
60
Neutrophil Gelatinase-Associated Lipocalin (NGAL)
Neutrophil gelatinase-associated lipocalin (NGAL) is a 25-kD protein, originally characterized in and secreted by neutrophils, that is bound to gelatinase. The physiologic role of NGAL in the kidney is unknown; however, it is believed to play a role in renal morphogenesis.
61 High throughput functional genomic studies have identified NGAL as one of the most upregulated transcripts in whole kidney tissue very early after acute injury. Downstream proteonomic studies using animal models have also revealed the 25-kD NGAL protein to be one of the earliest and most robustly produced proteins in the kidney after ischemic or nephrotoxic AKI.
62 Several clinical studies have suggested that urine NGAL expression may serve as an early marker of AKI.
63,
64,
65,
66 In a clinical trial of 71 children who underwent cardiac surgery, urinary NGAL increased within 2 hours of cardiopulmonary bypass to a level of >50 µg per L in all 20 children who had
an increase in SCr of >50% (RIFLE Risk) and in only one of the 51 children who did not meet the RIFLE definition of AKI.
67 Urinary NGAL also was evaluated in adult patients who underwent cardiac surgery, with far less impressive results.
68 In a cohort of 81 such patients at a single institution, 16 developed AKI, as defined by RIFLE. Preoperative urinary NGAL levels were comparable among patients who did and did not develop AKI and were not significantly different immediately after surgery. However, within 1 hour after surgery, the urinary NGAL concentration began dropping in patients who did not develop AKI but continued to rise in patients with AKI, peaking at 3 hours and remaining elevated for 24 hours.
68 In one study evaluating urinary NGAL levels in emergency room patients, the AUC for NGAL to detect AKI (0.948) did not significantly differ from the curve for SCr (0.921). Nevertheless, there was very little overlap in NGAL values in patients with AKI and prerenal failure, whereas SCr values overlapped significantly in AKI patients and in those that reversed the condition within 48 hours.
64 Urinary NGAL was measured in hospitalized patients with established AKI at study inclusion and after 2 days. Of the 145 patients analyzed, 75 had intrinsic AKI, 32 had prerenal AKI, and 38 patients could not be classified. Urinary NGAL levels effectively discriminated between intrinsic and prerenal AKI (AUC 0.87). An NGAL level over 104 µg per L indicated intrinsic AKI (likelihood ratio 5.97), whereas an NGAL level of 47 µg per L made intrinsic AKI unlikely (likelihood ratio 0.2). A logistic regression analysis showed that NGAL independently predicted a composite outcome (worsening RIFLE severity class within 7 days, need for RRT, and in-hospital mortality) after correcting for demographics, comorbidities, creatinine, and RIFLE class. The authors concluded urinary NGAL was useful in classifying and stratifying patients with established AKI.
69Several studies have examined the clinical endpoint of RRT initiation using NGAL.
70 Urine or plasma/serum NGAL have been studied in various clinical settings including children and adults (
Table 28.3). In a meta-analysis of studies that evaluated NGAL accuracy for diagnosis and prognosis, 1,948 patients from nine studies were included. The overall incidence of RRT was 4.3%, and the pooled analysis yielded an AUC of 0.782 (95% CI, 0.648-0.917) for discriminating patients who would receive RRT associated with AKI. For a cut-off in NGAL of 278 ng per mL, the sensitivity was 76% and specificity was 80%.
70 However, these studies included diverse patient populations, and the specimens and assays used in obtaining this estimate varied, making it difficult to translate the results to bedside clinical use.
N-acetyl-β-D-glucosaminidase (NAG)
N-acetylβ-D-glucosaminidase (NAG) is a lysosomal enzyme (>130 kDa) that has been localized to lysosomes in several human cells including the renal tubules. The large molecular weight precludes glomerular filtration implying that urinary
elevations are from tubular origin. Increased activity suggests injury to tubular cells or may reflect increased lysosomal activity. NAG catalyzes the hydrolysis of terminal glucose residues in glycoproteins and is the most active glycosidase found in proximal tubular epithelial cell lysosomes. Urinary NAG activity remains elevated during different kinds of active renal disease.
71 The diagnostic and prognostic ability of nine urinary biomarkers, including NAG, was evaluated in a cross-sectional study with 102 patients with established AKI and compared to 102 subjects without AKI.
72 The non-AKI subjects included healthy controls, ICU patients and subjects who underwent coronary angiography, whereas AKI patients were recruited at initial nephrology consultation. An age-adjusted analysis, using log-transformed biomarker values, showed NAG to be a significant predictor for RRT, mortality, and composite endpoint. The median normalized NAG level in AKI patients who underwent RRT was 0.06 U per mg Cr, versus 0.02 U per mg Cr in those who did not.
In another study including 635 patients presenting to the emergency room, urine NAG was not predictive of a composite outcome of nephrology consultation, ICU admission, RRT initiation, and mortality on multivariable analysis which included SCr and BUN.
64
Kidney Injury Molecule-1 (KIM-1)
Kidney injury molecule-1 (KIM-1) is a type I transmembrane glycoprotein with a cleavable ectodomain localized in the apical membrane of dilated tubules in acute and chronic injury. KIM-1 and its soluble ectodomain in urine (90 kDa) are believed to play a role in the regeneration processes after epithelial injury. It is undetectable in normal kidney tissue but expressed at very high levels in proximal tubule epithelial cells in human and rodent kidneys after ischemic or toxic injury.
73 A small study in six patients with confirmed acute tubular necrosis (ATN) showed KIM-1 expression via immunohistochemistry on kidney biopsy. The same investigators subsequently examined urinary KIM-1 in 40 patients and found KIM-1 levels elevated to a higher degree in patients with ischemic acute tubular necrosis (ATN) compared to patients with contrast nephropathy, other forms of AKI, CKD patients, and normal controls.
74 In a cohort of adults undergoing coronary artery bypass graft (CABG), urinary KIM-1 levels were predictive of subsequent AKI (sensitivity 0.74, specificity 0.9, AUC 0.83) at 12 hours postsurgery. Other studies also suggest that urinary KIM-1 may be useful in identifying ischemic ATN.
49,
75,
76In a study by Liangos et al., the AUC for prediction of RRT or death for KIM-1 was 0.61 (95% CI, 0.53-0.61), comparable to that of SCr and UO.
77 On adjusted analysis, patients in the highest KIM-1 quartile had a 3.2-fold higher odds (95% CI, 1.4-7.4) for a composite outcome compared to patients with the lowest quartile. However, the result was no longer significant when adjusted for multiple factors. In another study by the same authors, KIM-1 was not a significant predictor for RRT, but was a significant predictor for mortality.
72
Interleukin-18
Interleukin (IL)-18 also has been considered as a candidate biomarker for acute renal injury. IL-18 is a pro- inflammatory cytokine generated by caspase-1-mediated cleavage in injured proximal tubules and released into the urine.
78 It can also enter the urine by glomerular filtration. In animal models, IL-18 has been shown to exacerbate tubular necrosis, and neutralizing antibodies to IL-18 reduced renal ischemic injury in mice.
78 In humans urinary IL-18 levels were measured in 72 individuals, 14 with ATN, 8 with prerenal failure, 5 with urinary tract infections, 12 with CKD, 22 who received a kidney transplant, and 11 healthy control subjects. Patients with ATN had significantly higher urinary IL-18 levels as compared with control subjects and patients with other forms of kidney disease.
66 Similarly, patients who underwent transplantation and had delayed graft function had higher urinary IL-18 levels than patients with prompt graft function. Using samples collected on days 0, 1, and 3 of the Acute Respiratory Distress Syndrome (ARDS) Network trial, urine IL-18 levels of >100 pg per mL were associated with a 6.5-fold increased risk for development of AKI, defined by RIFLE.
52Many other factors beside GFR determine biomarker elevation: heterogeneity of clinical settings, underlying patient characteristics, severity of illness, and potential reasons for decreased renal function. It is more likely that a panel of biomarkers will provide a better understanding about the timing, nature, and the severity of an acute renal insult. Further work is clearly needed to inform the time course and performance of AKI biomarkers in various situations, to identify the pathways involved, to define clinical endpoints, including prerenal states, and to improve adjudication of biomarker data with respect to functional changes.
79
DETERMINING REVERSIBILITY
Although epidemiologic studies have shown that even small increases in SCr are predictive of nonrenal outcomes, SCr remains an insensitive and delayed marker of AKI. A key unanswered question is whether reversible AKI or prerenal AKI is also associated with increased complications and worse outcomes. For decades the differentiation from intrinsic AKI and prerenal AKI was based on urine analysis and microscopy parameters, but could only be confirmed retrospectively by the reversibility of creatinine increase or improvent of urine output. In the era of the new biomarkers new paradigms may arise to answer this question.
Physiology of Reversibility
The traditional classification of AKI includes causes associated with a decrease in renal blood flow (
Table 28.4), intrinsic renal parenchymal diseases (
Table 28.5), or obstruction of urine flow (
Table 28.6). Although obstructive AKI is usually easier to diagnose, prerenal and intrinsic renal causes can be difficult to distinguish in the clinical setting. Another
way to approach AKI classification is to ascertain the possibility of reversibility.
Prerenal AKI has been accepted as a reversible form of renal dysfunction, caused by factors that compromise renal perfusion. The term has been used as part of a dynamic process that begins with a reversible condition, the prerenal state, and can progress to an established disease, ATN. Experimental models have largely informed our current understanding of the physiology of the kidney injury associated with prerenal failure. Before the onset of clinically evident prerenal azotemia, the kidney passes through a phase of remarkable compensation called pre-prerenal azotemia.
80 Three main steps are involved in this compensatory mechanism: (1) the cardiac output fraction that reaches the kidney; (2) plasma filtration by the glomerulus (filtration fraction); and (3) proportion of the glomerular filtrate that is reabsorbed by the tubules. Renal blood flow (RBF) depends on the tone of renal vascular resistance (RVR) in relation to systemic vascular resistance (SVR): if the RVR increases in relation to the SVR, the RBF decreases. At reduced levels
of cardiac output, intrarenal factors are triggered, increasing renal arterial vascular tone and, consequently, decreasing the RBF. In order to maintain the intraglomerular pressure, efferent arteriolar resistance increases, preserving the filtration pressure even when the pressure in the afferent arteriolar decreases to levels low enough to cease filtration. Augmented activity of the sympathetic nervous system, RAAS, and vasopressin secretion increases the amount of filtered fluid and Na
+ that are reabsorbed.
These three mechanisms, control of blood flow to the kidney, the filtration fraction, and amount of fluid and solutes reabsorbed by the kidney, are the components responsible for the kidney reserve. However, the efficiency of these mechanisms has limits imposed by structural changes and the severity of the insult. The reserve is diminished by the presence of underlying arterial and intrinsic renal diseases that interfere with the control of renal blood flow, filtration fraction, and reabsorbtion functions, as well as by drugs that interfere with the vascular or neural humoral control of these mechanisms. When these compensatory mechanisms are overwhelmed, a prerenal state is discernible.
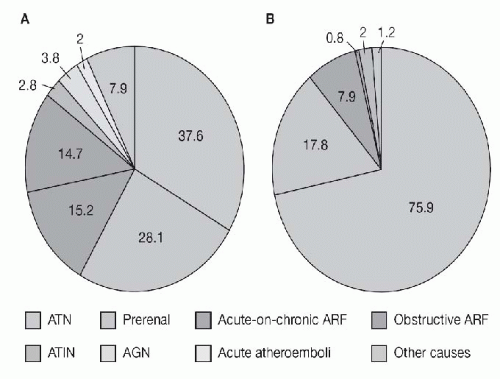
The frequency of prerenal azotemia as a cause of AKI varies with the clinical setting. A prospective study by Hou et al.
81 found prerenal azotemia to be the single most common cause of AKI in a general medical-surgical hospital. Liano found that prerenal causes of AKI among the elderly accounted for 48% of community-acquired AKI and 58% of hospital-acquired AKI (
Fig. 28.7).
82,
83 Brivet observed that in critically ill patients, prerenal causes accounted for 17% of cases of AKI.
84 Prerenal forms of AKI also appear to be common causes of community-acquired AKI and constituted 70% of all such cases as reported by Kaufman and associates.
85The terms “reversible AKI” or “prerenal failure” refer to all these different conditions that vary considerably in pathophysiology and course, including intravascular volume depletion, relative hypotension, compromised cardiac output, or hepatorenal syndrome (HRS). Although these terms are usually defined as an elevation of SCr or a reduction of UO that is easily reversible with improved renal perfusion
pressure, there is no agreement on the amount, nature, and duration of fluid resuscitation needed to eliminate a prerenal state. In most cases, the effect of fluid expansion or hemodynamic effect on renal function is retrospective and frequently evaluated by trial and error. The return of renal function to the previous baseline within 24 to 72 hours is considered to represent a prerenal or reversible condition. Diagnostic strategies have usually been based on demonstrating a fluid responsive change in renal function. The AKIN group recommended the “exclusion of urinary tract obstructions or … easily reversible causes of decreased urine output” and application of the diagnostic criteria “following adequate resuscitation when applicable.”
17 This modification in the criteria was intended to exclude transient changes in creatinine or UO due to volume depletion or other easily reversible causes of renal hypoperfusion. Although ideally created for prospective application, most studies seeking to validate the criteria were retrospective. In those studies, the exclusion of urinary tract obstruction and prerenal failure are impossible to verify. One small retrospective study attempted to evaluate the outcome of patients that met the adequate resuscitation criteria in comparison to those patients in which data on fluid challenge was not available.
86 Detailed fluid challenge information was available in 123 patients who met AKI criteria. They found that the association of AKI with in-hospital mortality was still significant even when the appropriate fluid challenge requirement was discarded.
Several clinical scenarios are often associated with a potentially reversible or prerenal form of AKI. The use of nonsteroidal anti-inflammatory drugs (NSAIDs) in patients with volume depletion, hypoalbuminemia, an edematous disorder, advancing age, underlying chronic renal failure, or recent diuretic use are all contributing factors for prerenal AKI.
87 A similar reversible form of AKI can complicate angiotensin-converting enzyme (ACE) therapy in the presence of decreased RBF from severe bilateral renal artery stenosis, renal artery stenosis in a solitary kidney, and other high-renin, high-angiotensin II states (i.e., edematous states and volume depletion disorders). In these cases ACE inhibition, with a resultant decrease in both renal perfusion pressure and efferent arteriolar constriction, can precipitously decrease GFR. About one third of patients with severe congestive heart failure experience an abrupt rise in SCr concentration following ACE inhibitor therapy.
88 In the setting of heart failure, this increase in SCr following ACE inhibition tends to be mild and readily reversible on discontinuation of the drug.
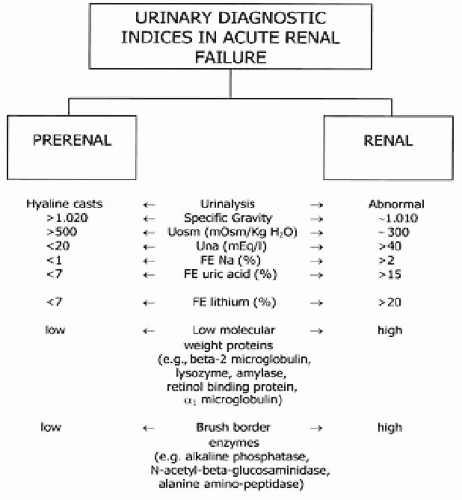
Although laboratory tests to distinguish reversible prerenal conditions from ATN have been used, these diagnostic parameters present frequent exceptions and the distinction between prerenal and renal causes are frequently not accurate (
Fig. 28.8). The plasma (P) urea/creatinine ratio, urine (U) osmolality, U/P osmolality, U/P creatinine ratio, urinary Na level, and fractional excretions of Na (FE
Na) are the most frequently used tests. Serum U/P creatinine ratio
helps to identify whether the oliguria is a result of water reabsorption (U/PCr >20) or loss of tubular function (U/ PCr <20). In reversible states the reabsorption of sodium is increased, not only from the increase in proximal tubular reabsorption of water, but also by the increase in aldosterone level secondary to hypovolemia. The frequent use of diuretic therapy limits the value of FE
Na. The fractional excretion of urea ((FE
UN) can be helpful in these cases. FEUN relates inversely to the proximal reabsorption of water; urea reabsorption leads to a decrease in (FE
UN and an increase in the BUN/creatinine ratio. Carvounis et al.
89 found that FEUN has a high sensitivity (85%), a high specificity (92%), and a high positive predictive value; in that study a FEUN less than 35% was associated with a 98% chance of reversible failure. Still, there are limitations for the use of (FE
UN. In osmotic diuresis and with the use of mannitol or acetazolamide, the proximal tubular reabsorption of salt and water is impaired, so there can be an increase in (FE
UN even in states of hypoperfusion. The same can occur when a patient is given a high protein diet or presents with excessive catabolism. Urinary osmolality is also used to evaluate the urinary concentration ability, a function that becomes impaired in the early process of tubular dysfunction. A value greater than 500 mOsm per kg indicates that tubular function is still intact, although there are also some considerations about this index; a low protein diet or low protein absorption by intestinal edema can impair the concentration ability of the urine and show a low osmolality even in reversible states.
There are some promising new biomarkers for AKI that may be helpful in distinguishing between reversible and established AKI.
64,
69,
90 During the reversible state, the persistent vasoconstriction associated with metabolic changes and inflammation promotes the release of cell functional markers that can be detected in the blood and urine. However, at the current time, there are no specific markers representing reversible conditions.
Obstruction to Urine Flow
Obstruction of urine flow is generally considered a less common cause of AKI. In several series, obstructive uropathy is encountered in 2% to 10% of all cases in AKI.
83,
84 However, obstructive uropathy is more common in selected patient populations, such as the very young or older men with prostatic disease, and patients with a single kidney or intraabdominal cancer, particularly pelvic cancer. Obstructive uropathy is most frequently encountered in community- and hospital-associated AKI and is less common in ICU-related AKI.
82,
83 For example, obstructive uropathy constitutes 20% to 40% of all community-acquired AKI. Finally, the cause of obstructive uropathy is often amenable to therapy. Thus, obstructive uropathy should be considered in each case of AKI.
Obstruction of urinary flow can occur anywhere from the kidneys to the urethral meatus. Certain points along this path are more susceptible to obstruction. The three points of narrowing along the ureter include the ureteropelvic junction, the crossing of the ureter over the area of the pelvic brim, and the ureterovesical junction. The cause of obstruction of urine flow can be classified as intrarenal or extrarenal (
Table 28.6). Intratubular deposition of either crystalline or proteinaceous material can increase intratubular pressure, thereby decreasing effective glomerular filtration pressure. For example, intratubular precipitation of uric acid can cause tubule obstruction and AKI. Acute uric acid nephropathy is most often seen following chemotherapy for leukemias and lymphomas. In this setting, the liver converts the purine load generated by cytolysis into uric acid. The high filtered load of uric acid and tubular reabsorption combine to produce high tubular concentrations of soluble urate and uric acid. Acidification of tubular fluid converts urate to uric acid, which can crystalize and occlude tubular lumens.
Abrupt exposure of the kidneys to high filtered loads of other insoluble crystalline substances can also cause an intrarenal form of obstructive uropathy. For example, AKI associated with calcium oxalate crystalluria can accompany ethylene glycol ingestion, administration of the anesthetic agent methoxyflurane, chronic pancreatitis, and use of gastrointestinal lipase inhibitors.
91,
92,
93,
94 Administration of high doses of methotrexate can be associated with AKI, possibly owing to intratubular precipitation of the insoluble 7-hydroxy metabolite of methotrexate.
95 Other crystalline substances that can potentially precipitate within renal tubules and lead to AKI include acyclovir, triamterene, sulfonamides, and protease inhibitors such as indinavir.
96Another cause of intratubular obstruction is the deposition of immunoglobulin light chains in plasma cell dyscrasias. Immunoglobulin light chains are low molecular weight proteins that are filtered through the glomerulus and reabsorbed into the proximal tubular epithelium by initially binding to a heteromeric receptor complex composed of megalin and cubilin.
97 Saturation of this receptor- mediated endocytotic process results in the presence of free light chains in the distal nephron and urine. Nephrotoxicity of the metabolism of monoclonal light chains causes tubulointerstitial nephritis and cast nephropathy (also known as “ myeloma kidney”), resulting in AKI and progressive CKD from tubular obstruction.
97Extrarenal lesions are the most common cause of postrenal AKI and are listed in
Table 28.6. Several factors determine renal response to extrarenal obstruction. The site, degree, and rapidity of onset of obstruction are all important. Without a complicating infection, substantial improvement in renal function can follow decompression of the urinary tract after several days of complete obstruction. In men, prostatic obstruction is by far the most common cause of postrenal AKI because of its critical location at the bladder outlet. Obstruction of the upper urinary tract is a less common cause of AKI because it requires simultaneous obstructions of both ureters or unilateral ureteric obstruction with either absence of or severe disease in the contralateral kidney. Intraureteric obstruction can be due to stone, released necrotic papillae, tumor, pus, blood clots, and fungal balls. Papillary necrosis can occur in the
setting of sickle cell disorders, chronic urinary tract infections, analgesic abuse, and obstructive uropathy. Extraureteric lesions producing obstruction include retroperitoneal fibrosis, adenopathy, and tumors. Retroperitoneal fibrosis is often idiopathic but may be encountered in response to retroperitoneal neoplasia as well as in the setting of some pharmacologic agents ( methysergide, methyldopa
β β-blockers), prolonged peritoneal dialysis, and some connective tissue diseases. It has been reported that a high frequency of AKI occurs because of prostatic carcinoma in males and pelvic carcinoma (predominantly cancer of the cervix) in females causing ureteric occlusion. Less commonly encountered causes of extrinsic ureteric obstruction include inflammatory bowel disease (predominantly right-sided obstruction), an inflammatory reaction resulting from a leaking abdominal aortic aneurysm, and the late stages of pregnancy.
Acute obstruction can also be related to use of pharmacologic agents with potential anticholinergic effects (e.g., tricyclic antidepressants, phenothiazines, antihistamines) and cold remedies containing β-adrenergic agents (e.g., phenylpropanolamine) often precipitate acute urinary retention by impairing detrusor function and enhancing bladder sphincter tone, respectively.