ESSENTIALS OF DIAGNOSIS
ESSENTIALS OF DIAGNOSIS
High fever, frequent bloody stools, severe abdominal pain, dehydration, and no improvement after 3–4 days of initial supportive treatment are worrisome features.
Sigmoidoscopy and biopsy are indicated in patients with bloody dysenteric stools and tenesmus lasting more than 3–4 days.
Upper endoscopy and biopsy are indicated in patients with persistent diarrhea and evidence of malabsorption.
Routine stool cultures aid in identifying Salmonella, Shigella, and Campylobacter but rarely provide useful information if diarrhea develops 2–3 days after hospitalization.
Clinical features of shigellosis, salmonellosis, and Campylobacter colitis (diarrhea, tenesmus, fever, abdominal cramps) often overlap.
Consider Clostridium difficile infection after both recent and remote (within 3 months) use of antibiotics and if diarrhea develops during hospitalization.
Risk factors for severe C difficile infection include age >65, renal failure, immunosuppression, and white blood cell count >20,000/μL.
Consider enterohemorrhagic Escherichia coli (E coli O157:H7) in patients with bloody diarrhea, abdominal pain, leukocytosis, and little or no fever, especially if uremia or microangiopathic anemia develops; if suspected, avoid antibiotics.
Giardiasis and cryptosporidiosis are best diagnosed using stool immunoassays directed against Giardia and Cryptosporidium antigens which are more sensitive than microscopic stool examination.
Up to 10% of patients who have had infectious diarrhea may develop a postinfectious irritable bowel syndrome.
ACUTE DIARRHEA
Acute diarrheal diseases remain a major global public health problem, responsible for an estimated 2 million or more deaths annually. Most deaths occur in developing countries and many occur in infants and young children. The majority of acute diarrheal episodes reflect gastrointestinal infections, but medications, food intolerances, or the abrupt onset of chronic disease may be causative (Table 5–1). In the United States, acute diarrheal diseases are a major health and economic problem resulting in between 150 and 300 million total episodes, 900,000 hospitalizations, and approximately 6000–12,000 deaths per year.
Viral infections Bacterial infections Parasitic infections Medication related Laxatives Antibiotics Antacids Nonsteroidal anti-inflammatory agents Nutritional supplements Others (colchicine, gold, and many more) Food related Allergies (shellfish) Additives (sulfites) Sorbitol Carbohydrate intolerances Abrupt onset of chronic disease Inflammatory bowel disease Celiac disease Irritable bowel syndrome |
Acute diarrheal diseases are second only to respiratory infections as a cause of time lost from work in the United States. As a symptom, diarrhea can be defined as an increase in frequency, volume, and often urgency of the passage of stool and as a decrease in stool consistency. More objectively as a sign, diarrhea is an increase in stool mass to greater than 200 g/24 h, the upper limit of normal stool weight. Acute diarrheal disease is generally defined as having begun within 2 weeks of presentation; diarrhea that persists for between 2 and 4 weeks is considered persistent and diarrhea that lasts more than 4 weeks is considered chronic.
[PubMed: 14702526]
Under normal circumstances, adults ingest approximately 2 L of fluid per day. An additional 7 L of endogenous secretions from salivary, gastric, pancreatic, biliary, and enteric sources enter the intestine for an approximate 24-hour load of 9 L. Ingested nutrients are dissolved or suspended in this 9-L fluid load. The healthy small intestine will absorb about 7.5 L, largely in the duodenum and jejunum. Of the 1.5 L that traverse the ileocecal valve, the colon absorbs approximately 1.3 L, resulting in a stool mass of no more than 200 g/24 h. The maximum absorptive capacity of the small intestine is about 12 L and that of the colon, 4–6 L, for a total of 18 L; roughly double the normal daily fluid load.
An increased fluid load sufficient to overwhelm the intestinal and colonic absorptive capacity of about 12 L usually results in diarrhea. Excessive endogenous fluid secretion is the usual culprit, but excessive fluid intake may contribute. Examples of diseases that cause diarrhea by excessive secretion include cholera, toxigenic E coli infection, or, rarely, a vasoactive intestinal peptide-secreting tumor (Table 5–2).
Epithelial cell absorptive and digestive function in the small and large intestine is impaired and permeability of the epithelial barrier is increased by the mucosal damage caused by a variety of gastrointestinal viral, bacterial, and protozoal infections. In addition, the available absorptive surface may be reduced as in, for example, rotavirus and norovirus enteritis and in giardiasis. Increased intraluminal osmolality is a cause of acute diarrhea. Ingestion of poorly absorbed or nonabsorbable laxatives such as magnesium citrate or magnesium hydroxide or polyethylene glycol 3350 are the classic examples, but impaired nutrient absorption in some intestinal infections contributes to diarrhea by increasing intraluminal osmolality. All of the mechanisms detailed above may be causative in acute diarrheal diseases in which there is substantial mucosal inflammation as occurs in most invasive and some toxigenic bacterial enterocolitides. If mucosal inflammation is mild or absent, as in cholera, enhanced fluid secretion, altered permeability, enhanced motility, or, in the case of laxative ingestion, increased intraluminal osmolality are the major mechanisms.
A careful history and physical examination are crucial in the evaluation of patients presenting with acute diarrhea and should provide information as to the possible nature, underlying cause, and severity of the diarrhea. Historical points that should be explored include (1) any recent travels; (2) the nature of recent food ingestion, including type, preparation (fully cooked, rare, or raw), and location (home, restaurant, or street vendor); (3) occurrence of a similar illness among recent contacts; (4) medication history, especially new medications or past or current use of antibiotics; (5) sexual history; and (6) predisposing conditions, such as compromised immune status.
The character of the diarrhea also provides clues as to the site of involvement by the process causing the diarrhea. A large stool volume with little urgency, no tenesmus, and only a moderate increase in stool number suggests involvement primarily of the small intestine and more proximal colon, whereas frequent, low-volume dysenteric stools containing blood and mucus associated with urgency and tenesmus suggest disease involving the rectum and distal colon. Although a patient’s subjective description of the nature of the diarrhea is helpful, examination of the stool by the clinician is most useful.
On physical examination, the presence or absence of fever should be determined. Hypotension, orthostasis, tachycardia, poor skin turgor, and dry mucous membranes indicate dehydration, a major cause of morbidity and mortality in acute diarrheal diseases, especially in the young and the elderly. A careful abdominal examination is essential to assess for evidence of localized abdominal infections (eg, appendicitis or diverticulitis) that may be associated with diarrhea. The abdomen should also be assessed for distention, which might suggest megacolon, and for the presence of peritoneal signs.
[PubMed: 15123984]
[PubMed: 22080825]
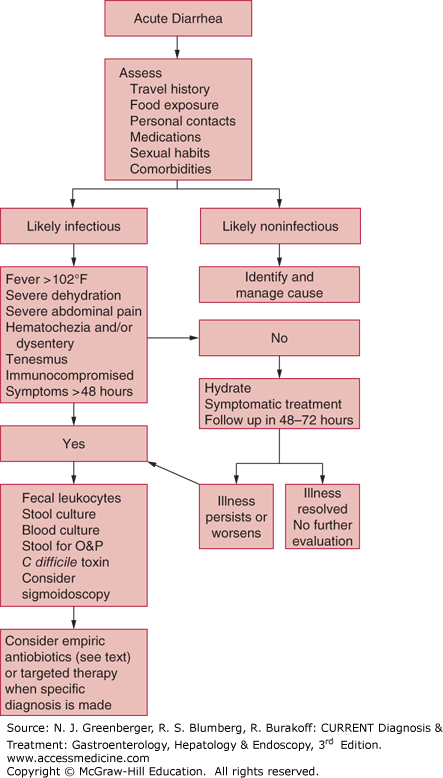
The majority of patients with acute diarrhea in developed countries have relatively mild, self-limited illness, which resolves in a day or two and requires no specific diagnostic studies. A major goal of the initial assessment, including the history and physical examination, is to identify patients who have more serious disease meriting prompt and specific diagnostic studies and aggressive treatment (Figure 5–1). Worrisome features include high fever, dehydration, frequent bloody stools with tenesmus (dysentery), severe abdominal pain, an immunosuppressed host, and no improvement in or progression of symptoms after 3–4 days of supportive treatment (see Figure 5–1). Useful tests for further evaluation include assessment of stool for fecal leukocytes or leukocyte markers such as lactoferrin or calprotectin, stool culture, blood culture, stool examination for ova and parasites, stool testing for C difficile toxin, endoscopy and mucosal biopsy, and, in selected cases, abdominal imaging studies.
Examination of stool suspensions stained with methylene blue for the presence of fecal leukocytes is a time-honored test for evaluation of patients with acute diarrhea. Neutrophils are usually present in patients with dysenteric stools (containing blood and mucus) caused by infection with invasive bacteria (Table 5–3) and colitides unassociated with infection, as can occur in acute onset or during acute exacerbations of inflammatory bowel disease (IBD). In other words, the presence of neutrophils indicates the presence of an inflammatory colitis or enterocolitis but not its cause. Thus, the test lacks specificity for infectious colitis, and its reported sensitivities for invasive bacterial infections are only in the range of 60–70%, in part because the presence of neutrophils in stools is variable in patients with diarrhea caused by Salmonella or Yersinia infection. Fecal leukocytes are usually absent from the watery stools caused by viral infections and toxin-producing bacteria with exceptions being C difficile- and cytomegalovirus-induced enterocolitis, in which fecal neutrophils may be present (see Table 5–3).
| Usually Present | Usually Absent | Variable |
|---|---|---|
Shigellosis Campylobacter Invasive Escherichia coli | Noroviruses Rotavirus Vibrio cholerae Enterotoxic E coli Enterohemorrhagic E coli Staphylococcus or Bacillus cereus food poisoning Giardiasis | Salmonellosis Noncholera vibrios Clostridium difficile Yersinia Amoebiasis Cytomegalovirus |
Despite its limitations, fecal leukocyte testing remains worthwhile and, if positive, helps select patients who merit more extensive diagnostic evaluation and possibly empiric antibiotic therapy. Fecal lactoferrin and fecal calprotectin, surrogate markers for fecal neutrophils, can be measured using latex agglutination and enzyme-linked immunosorbent assay (ELISA). The tests have been used most widely in assessing patients with IBD. They appear to be more sensitive, less observer dependent, but more costly than the conventional fecal leukocyte assay. Falsely positive results for fecal lactoferrin may occur in breast-fed infants.
The indications for and cost-effectiveness of obtaining stool cultures from patients with acute diarrhea are still being debated. However, there is general consensus that too many are obtained. Most surveys report an incidence of positive stool cultures in only 1.5–3% of submitted stools with an estimated cost of more than $1000 per positive culture. As indicated earlier, most episodes of acute diarrhea are self-limited and resolve within 48–72 hours, often before the results of a stool culture are available. Stool cultures are rarely informative in nonimmunosuppressed patients who develop diarrhea 3 or more days after hospitalization.
Stool cultures should be obtained from patients with severe dehydration, fever exceeding 102°F (38.9°C), dysenteric stools, and stools that contain neutrophils, and from those in whom the culture may ultimately guide therapy. Stool cultures should also be obtained from all immunosuppressed patients who develop diarrhea or patients with other severe comorbidities, including acute IBD flares. Stool cultures should be obtained for epidemiologic purposes from food service workers or if an outbreak of diarrheal disease is suspected.
Once obtained, the stool specimen should be sent to the laboratory as soon as possible to minimize false-negative results. The clinician should also be cognizant of which organisms are identified by the particular laboratory performing the culture. In some laboratories, only Salmonella, Shigella, and Campylobacter are identified during routine processing. Hence, the clinician should specify other organisms if suspected, such as Yersinia, enterohemorrhagic E coli (EHEC), Aeromonas, or noncholera Vibrio, so that proper media and culture conditions are used.
Blood cultures should be obtained from patients with high fever, those with shaking chills, and those who are immunosuppressed.
The yield of positive examinations for ova and parasites is low among patients with acute diarrhea as most parasitic infestations that cause diarrhea cause symptoms lasting several weeks or more. Indications for assessment include diarrhea that persists for more than 2 weeks, a history of travel to areas endemic for parasitic disease, exposure of children to day care centers or summer camps, a concomitant history of immunosuppression (HIV, chemotherapy, immunosuppressive drugs), known idiopathic IBD, gay lifestyle, and unexplained peripheral eosinophilia. As ova and parasites can be passed intermittently, optimal testing requires prompt examination of each of three specimens collected on separate days. If giardiasis or cryptosporidiosis is suspected, ELISAs or immunochromatographic assays that detect Giardia and/or Cryptosporidium antigens are superior to conventional microscopic examination of the stool, with sensitivity of 80–90% and >90% specificity.
The recently reported rising incidence and increased morbidity and mortality of C difficile colitis (see later discussion) underscores the importance of C difficile toxin testing of the stool for timely diagnosis and implementation of therapy for this important disease. Groups at risk and, hence, indications for the test include patients who have received recent or even distant antibiotic therapy, patients who develop diarrhea in a hospital or other institutional setting, immunosuppressed patients, and patients with idiopathic IBD. Community-acquired cases even in the absence of known recent antibiotic use are being increasingly recognized.
The cytotoxicity assay using cultured fibroblasts has been the gold standard for testing for C difficile toxin, with sensitivity of approximately 70–90% and almost absolute specificity. However, the test is costly and requires 2–3 days for completion. Also available are enzyme immunoassays (EIAs) for C difficile toxins A and B which lack high sensitivity, EIA for clostridial glutamate dehydrogenase (GDH) which screen for clostridia but not its toxins, polymerase chain reaction (PCR) for toxin genes which have high sensitivity but have been associated with false positives and direct culture of C difficile, the latter of use for epidemiologic studies. A stepwise approach is now often used in clinical laboratories, first screening for clostridial GDH and/or toxins with EIA and then, if either is positive, following with PCR for toxin genes.
Sigmoidoscopy and biopsy can be very useful in selected patients with acute diarrhea. Sigmoidoscopy is indicated in patients with bloody, dysenteric stools and tenesmus lasting more than 3–4 days. The endoscopic appearance of the mucosa is often indistinguishable in patients with acute infectious diarrhea caused by invasive organisms compared with patients with idiopathic IBD, but in such patients, it provides some information as to extent and severity of disease. Additionally, rectosigmoid biopsies may help distinguish acute infectious diarrhea from diarrhea caused by an acute flare-up of IBD (Plates 1 and 2). Sigmoidoscopy can also help exclude ischemic colitis, which may present with abdominal pain and bloody diarrhea. Sigmoidoscopy can provide a rapid, tentative diagnosis of C difficile colitis if the characteristic pseudomembranous lesions are present. Additionally, sigmoidoscopy can provide rapid diagnosis of amoebiasis if secretions from the edge of ulcers are examined microscopically and the organism is seen. Mucosal biopsy from immunosuppressed patients may detect cytomegalovirus or herpetic proctocolitis. Upper endoscopy is indicated in patients with persistent diarrhea and malabsorption and, through examination of duodenal aspirates and mucosal biopsies, may detect parasitic infections including giardiasis, cryptosporidiosis, Cystoisospora infection, and occasionally strongyloidiasis.
[PubMed: 20346452]
Imaging studies are usually not required in patients with uncomplicated acute diarrhea. An abdominal flat and upright film may be useful if there is worrisome abdominal distention in order to screen for toxic megacolon, which can occur in C difficile colitis and colonic amoebiasis. On occasion, computed tomography (CT) or ultrasound scan of the abdomen and pelvis may be indicated, if there is associated severe abdominal pain, tenderness, or evidence of peritoneal irritation as may occur in, for example, Yersinia enterocolitis, to exclude a focal intra-abdominal or pelvic process.
Some general principles of treatment of acute diarrhea are summarized next. When specific therapy of a defined cause of acute diarrhea is indicated as, for example, for C difficile colitis, such treatment is described later, in the section titled “Acute Diarrheal Disease Caused by Specific Infections.”
Oral rehydration therapy will suffice in most patients who develop acute diarrhea. In milder cases, juices, broth, and water along with salted crackers usually suffice. If dehydration is more severe, oral rehydration fluids should be utilized. The World Health Organization (WHO) recommends the following formula per liter of water:
2.6 g sodium chloride
2.5 g sodium bicarbonate or 2.9 g trisodium citrate
1.5 g potassium chloride
13.5 g glucose or 27 g sucrose
Several oral rehydration solutions in addition to that recommended by the WHO are commercially available. Rehydrate closely mimics WHO oral rehydration solutions, whereas others (Pedialyte, Enfalyte) contain less sodium and may require intake of a larger volume. Maintaining a 1:1 molar ratio of sodium to carbohydrate facilitates sodium transport in diarrheal diseases in which the enteric sodium–glucose cotransport mechanism remains intact as, for example, in cholera or enterotoxigenic E coli (ETEC) infection. Some clinicians favor preparations formulated with complex carbohydrates such as rice-based solutions (CeraLyte). The WHO recommends zinc supplementation of oral rehydration solutions for children (10 mg/day for infants <6 months old; 20 mg/day for children >6 months old), but there are no recommendations for adults due to lack of data.
Intravenous hydration is often overutilized in developed countries such as the United States. However, if diarrhea is massive and dehydration severe, especially in infants and young children, the elderly, and patients unable to rehydrate orally due to nausea, vomiting, or other comorbidities, intravenous rehydration is indicated. Serum electrolyte levels should be closely monitored and imbalances corrected.
[PubMed: 19550407]
[PubMed: 15245700]
The antimotility agent loperamide can be used with caution to provide symptomatic relief in patients without high fever, colonic distention, or dysenteric stools. The usual dose is 4 mg initially followed by 2 mg every 4–6 hours, as required for persistent diarrhea, not to exceed 16 mg/24 h. Absorption may be increased by prolonging contact time of luminal contents with mucosa, although oral fluid intake should be continued. Fluid may pool in the gut lumen as antimotility agents do not decrease intestinal secretion. Diphenoxylate has also been used but is less desirable as it crosses the blood-brain barrier and has central opiate actions. Antimotility drugs should generally be avoided in patients with dysentery, and no antimotility drug should ever be used if the possibility of toxic megacolon is a concern. Moreover, there is some evidence that suggests that the use of antimotility agents, while reducing the number of stools, may prolong the course of some enteric infections, including those caused by Shigella and ETEC, perhaps by prolonging the time required for clearance of causative organisms and toxins from the gut. Bismuth subsalicylate reduces stool number, but there is no evidence that fecal fluid losses are decreased significantly.
Although a controversial topic, antibiotics are overutilized in acute infectious diarrhea. Most episodes are self-limited and clear within 2–4 days or less without specific therapy; hence, most patients would derive little or no benefit and risk potential harm if antibiotics were indiscriminately prescribed. Their use predisposes to the development of resistant organisms. They provide no benefit in viral diarrhea caused by rotaviruses or noroviruses and may be harmful in EHEC infections, prolong fecal excretion of nontyphoidal Salmonella, and precipitate C difficile colitis. Their benefit in Yersinia, Campylobacter, and Aeromonas infections is controversial.
Generally accepted indications for the use of an empiric antibiotic include fever greater than 102°F (38.9°C) and chills, febrile dysentery, traveler’s diarrhea that is severe or affects an individual in whom a shortened duration of illness is urgent, individuals with severe dehydration and prolonged, severe diarrhea (1 week or more), and the immunocompromised host. Quinolones are widely used for empiric treatment; macrolides such as azithromycin or erythromycin and, for travelers, rifaximin are alternatives.
Antibiotics should be avoided in patients with bloody stools, abdominal pain, but little or no fever until EHEC has been excluded from consideration.
Recommendations regarding antibiotic therapy for specific pathogens that cause acute infections are discussed in the next section.
[PubMed: 19457416]
ACUTE DIARRHEAL DISEASE CAUSED BY SPECIFIC INFECTIONS
As indicated previously, most acute diarrheal disease is caused by transmissible infectious viruses, bacteria, or parasites. Infectious organisms cause disease by a number of mechanisms (Table 5–4). Some actually invade and proliferate in the intestinal epithelium, underlying mucosa, and lymphoid follicles. Others produce tissue-damaging cytotoxins while in the lumen and in contact with the epithelium. Still others elaborate enterotoxins that produce profound functional alterations but no detectable histologic lesion. Some adhere to the epithelial surface and induce epithelial damage and mucosal inflammation without invasion. Finally, still others produce toxins in vitro that, when ingested, induce symptomatic gastrointestinal illness.
| Mechanisms | Examples |
|---|---|
| Invasive | Rotavirus Campylobacter Salmonella Shigella |
| Cytotoxic | Clostridium difficile Enterohemorrhagic Escherichia coli |
| Enterotoxic | Vibrio cholerae Enterotoxigenic E coli |
| Enteroadherent | Enteropathogenic E coli Giardiasis |
| Toxin produced in vitro | Bacillus cereus Staphylococcus aureus |
The human host has defenses that resist enteric infections. Gastric acid secretion reduces bacterial colonization of the proximal small intestine, and there is evidence that individuals who have had acid-reducing gastric surgery are more susceptible to nontyphoidal salmonellosis and selected other enteric infections. Normal gastric motility is a defense factor; stasis predisposes to intestinal intraluminal bacterial proliferation, and there is evidence that the use of antimotility agents prolongs diarrhea caused by some enteric pathogens. The normal intestinal flora itself is protective. Its alteration by antibiotic therapy predisposes to C difficile colitis and reduces the number of organisms required to produce nontyphoidal salmonellosis. Antibodies secreted into the intestinal lumen by the intestine and via the biliary tract appear to play a protective role in reducing colonization of pathogens in the gut.
[PubMed: 15575058]
Noroviruses are caliciviruses—small, single-stranded RNA viruses that, despite extensive efforts, have not been cultured (Table 5–5). It is estimated that these viruses produce approximately 19–21 million episodes of illness, 56,000–71,000 hospitalizations, and 570–800 deaths annually in the United States. Noroviruses are the leading cause of foodborne disease in the United States. With advent of effective rotavirus vaccines, norovirus disease is now the most common cause of viral gastroenteritis in children as well as adults. Epidemic outbreaks in hospitals, nursing homes, in schools from catered food, in restaurants, and on cruise ships have been widely publicized, but endemic cases also occur. Transmission is via the fecal-oral route through contaminated food and water, from virus-contaminated environmental surfaces, and direct person-to-person contact. After ingestion, noroviruses bind to histo-blood group antigens on the enterocyte surface. The histo-blood group antigens are a major determinant of host susceptibility; nonsecretors are resistant to infection by a majority of strains of norovirus. Histologic studies of volunteers have shown that after ingestion, noroviruses induce a mild to moderate mucosal enteritis in susceptible individuals that peaks in severity approximately 48 hours after viral ingestion and resolves completely within 4–6 weeks or earlier. Lesions of the gastric mucosa were not detected after ingestion of Norwalk agent.
| Feature | Noroviruses | Rotavirus |
|---|---|---|
| Size | 27 nm | 70 nm |
| Nucleic acid | SS RNA | DS RNA |
| Age | >2 years | Infants, young children |
| Transmission | Fecal-oral: food, water | Fecal-oral |
| Pathologic findings | Mild enteritis | Mild to severe enteritis |
| Incubation | 1–2 days | 1–3 days |
| Duration | 1–3 days | 5–7 days |
| Diagnostics | RT-PCR and immunoassays are available; used largely to identify outbreaks | Commercial ELISA, latex agglutination, and RT-PCR assays useful for diagnosis |
| Treatment | Rehydration as needed | Vigorous rehydration |
| Vaccine | Several investigational vaccines are being tested | RotaTeq, Rotarix |
The incubation period is usually 12–48 hours, and clinical symptoms may include nausea, vomiting, watery diarrhea, and abdominal cramps. Fever, if present, is usually mild (<101.5°F [38.6°C]). The duration of clinical symptoms is short, usually ranging from a few hours to 3 days. The characteristic histologic lesion has been observed in asymptomatic volunteers after norovirus ingestion, providing evidence that some infections are subclinical. Routine laboratory studies remain normal unless severe dehydration develops, in which case elevations of blood urea nitrogen and creatinine and leukocytosis may be seen.
The diagnosis depends on the epidemiology and clinical features. A reverse transcription PCR assay and diagnostic immunoassays are available and are used largely to identify outbreaks but are not generally used to diagnose individual patients.
Treatment is supportive and consists of oral rehydration with carbohydrate–electrolyte solutions (see earlier discussion) and, in a small minority of patients with severe dehydration, by intravenous hydration. Symptomatic treatments such as bismuth subsalicylate and loperamide can be used but are usually not needed, and there is little evidence that they significantly influence fecal fluid losses. There is no effective antiviral agent. Attempts to develop an effective vaccine are ongoing and some early phase 2 studies are encouraging.
The prognosis is excellent for this generally self-limited disease, although deaths may occur in association with norovirus gastroenteritis, largely in debilitated, elderly patients with significant comorbidities or in very young children.
[PubMed: 19864676]
[PubMed: 23876403]
Rotaviruses are a major cause of infectious diarrhea worldwide, accounting for an estimated 500,000 or more deaths each year. In the United States, prior to the widespread use of rotavirus vaccines, approximately 2.7 million children developed rotavirus gastroenteritis yearly, resulting in over 50,000 hospitalizations and an estimated 30 deaths. However, there is increasing evidence that with the introduction of effective vaccines in 2006, the incidence of rotavirus infection has decreased dramatically by over 80%.
Rotaviruses are double-stranded RNA viruses (see Table 5–5) whose classification is complex. There are several groups, which, in turn, may contain subgroups and multiple serotypes. Most but not all disease in humans is caused by group A. The virus invades, proliferates in, and destroys enterocytes, resulting in the passage of abundant virions in the stool of infected individuals for 7–10 days; however, virions can be shed for up to 3–4 weeks. Significant enteritis develops, with patchy shortening or even complete loss of villi, crypt hyperplasia, and inflammation of the lamina propria.
Symptomatic illness is more common in winter and occurs largely in infants and young children. Watery diarrhea, which can be profuse, and vomiting are the major symptoms, accompanied by fever in 50% or more of cases. Reinfections in older children and adults are usually asymptomatic or mild but can occasionally cause severe symptoms. Dehydration is a hazard—especially in infants, if diarrhea is accompanied by vomiting that precludes adequate oral fluid intake—and is the major cause of morbidity and death. The illness generally lasts from 4 to 7 days, but more protracted diarrhea has been reported. The diagnosis can be established by demonstrating rotavirus antigen in the stool by specific ELISA, latex agglutination, or PCR assays.
Hydration is the cornerstone of therapy and can often be achieved with oral rehydration regimens containing sugar, salt, and water or commercial oral rehydration formulations unless diarrhea is profuse and accompanied by vomiting, in which case intravenous rehydration may be lifesaving. The American Academy of Pediatrics does not recommend symptomatic therapy, including antimotility drugs, bismuth subsalicylate, and probiotics, for children younger than 5 years.
The overall prognosis is excellent, with the vast majority of patients recovering completely within 7–10 days, except as noted earlier.
A pentavalent human-bovine reassortant rotavirus vaccine (RotaTeq) and a monovalent attenuated human rotavirus vaccine (Rotarix) are licensed in the United States. Vaccination is recommended for all infants who have no contraindications. There appears to be a very small increase in the risk of intussusception during the first week after administration of the first vaccine dose but experts agree that the benefits of vaccination greatly outweigh this risk.
[PubMed: 24422677]
[PubMed: 19907323]
[PubMed: 23487388]
Enteric adenovirus causes a rotavirus-like endemic gastroenteritis primarily in children younger than 2 years. Its incubation period is longer than that of rotavirus, but it appears to be less contagious. Astrovirus also causes endemic gastroenteritis primarily in children but also among elderly and immunocompromised adults; occasional multicase outbreaks have been described. Clinically, gastroenteritis caused by adenovirus and astrovirus resembles that caused by rotavirus and norovirus, and the principles of therapy are the same. Diarrhea may also accompany enterovirus and coxsackievirus infections, although manifestations involving other systems often overshadow any accompanying diarrhea.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree