and Andrea Bischoff1
(1)
Pediatric Surgery, Colorectal Center for Children Cincinnati Children’s Hospital, Cincinnati, OH, USA
23.1 Introduction
23.2 Neonatal Approach
23.5 Bifid Scrotum
23.6 Hypospadias
23.9 Ectopic Vas Deferens
23.10 Ectopic Verumontanum
23.11 Megalourethra
23.13 Neurogenic Bladder
23.14 Postoperative Problems
23.16 Sexual Problems
23.17 Tethered Cord
Electronic supplementary material
Supplementary material is available in the online version of this chapter at 10.1007/978-3-319-14989-9_23.
23.1 Introduction
Urogenital problems in patients with anorectal malformation represent a very important source of morbidity; in fact, it is more likely for a child with an anorectal malformation to die from a urologic problem, rather than from gastrointestinal problems or any other associated defect. Pediatric surgeons must keep in mind that the patient with an anorectal malformation represents a potential life-threatening problem originated in the urogenital tract, mainly kidney damage and eventually kidney failure.
The association of urogenital defects with anorectal malformations has been recognized for a long time. The incidence has been estimated to vary from 25 to 85 % [1–17]. The majority of reports inform of an incidence around 30–50 %. We believe that the difference found between different authors is a consequence of the thoroughness of the search. A high index of suspicion, in an institution with high technologic support, mostly likely will find more urologic problems.
Table 23.1 shows the frequency of associated urologic defects for each type of anorectal malformation. The anorectal defects are listed according to the degree of complexity, being the simplest the perineal fistulas and in the opposite extreme the cloacal exstrophy and other complex malformations. It can be very easily seen that the frequency of association increases directly proportional to the height of the defect and the complexity of the malformation. This information is very valuable to increase the index of suspicion of the clinician and allow an early detection of the urologic problems, particularly those that may produce renal damage and eventually kidney failure.
Table 23.1
Frequency of association of urologic defects for each type of anorectal malformation
Females | |||
|---|---|---|---|
Absent kidney (%) | Vesicoureteral reflux (%) | Hydronephrosis (%) | |
Cloaca CC >3 cm | 26 | 40 | 45 |
Cloacal exstrophy | 26 | 21 | 9 |
Cloaca CC <3 cm | 17 | 21 | 22 |
Vestibular | 10 | 13 | 6 |
Perineal | 4 | 5 | 5 |
Males | |||||
|---|---|---|---|---|---|
Absent kidney (%) | Vesicoureteral reflux (%) | Hydronephrosis (%) | Hypospadias (%) | UDT (%) | |
Bladder neck fistula | 31 | 29 | 24 | 15 | 18 |
Prostatic | 16 | 6 | 6 | 6 | 7 |
Bulbar | 11 | 13 | 9 | 10 | 5 |
No fistula | 7 | 7 | 4 | 3 | 10 |
Perineal | 2 | 3 | 6 | 1 | 2 |
It is a relatively common experience to see a patient that was born with an anorectal malformation that was repaired early in life, and the parents were not alerted about the fact that the patient had a urologic abnormality, and years later, the parents have the very unpleasant experience of learning that the child needs a kidney transplant. In some cases, the patient is born with the kidneys already severely damaged, and the role of the physician should be directed to avoid worsening of that problem. On the other hand, some patients are born with normal kidneys, but the lack of care of a problem such as vesicoureteral reflux and a poor functional bladder eventually may damage the kidneys, and the patient may require a kidney transplant or even die. This last situation is mostly preventable and should not occur. All this means that it is very important to suspect, diagnose, and treat adequately all those urologic problems associated with anorectal malformations. In addition, patients with anorectal malformations should be followed on a long-term basis, monitoring their kidney function and the anatomy of the urinary tract, because we know that many of them have anatomic or functional problems with a tendency to deteriorate.
23.2 Neonatal Approach
If a pediatric surgeon has no experience in the diagnosis and management of urologic problems, he should work together with a pediatric urologist in the initial evaluation of a patient with anorectal malformation.
All newborn babies with anorectal malformations should have a kidney ultrasound, within the first 24 h of life and before any surgical intervention. Female babies, particularly those with cloaca (single perineal orifice), must have, in addition, a pelvic ultrasound to rule out the presence of a hydrocolpos.
We believe that a male baby with a normal kidney ultrasound, that is passing urine satisfactorily, has a normal sacrum, and no evidence of tethered cord can be operated on without any further urologic tests. On the other hand, if the baby has hydronephrosis, he will require further urologic studies, including a voiding cystourethrogram. We do not believe that the voiding cystourethrogram should be done routinely, in all male babies with anorectal malformations, because in our experience, it is not a good study to diagnose the location of the recto-urinary fistula. In addition, the urethral catheterization in babies with rectourethral fistulas sometimes is difficult, because the patients frequently have kinks or narrowing of the urethra, particularly at the junction between the rectum and the urethra.
If the baby has tethered cord and/or a very abnormal sacrum (sacral ratio less than 0.4), he is at risk of suffering from deterioration of the bladder function, which eventually may affect the kidneys and therefore is considered a high-risk patient from the urologic point of view, even if the initial kidney ultrasound is normal.
The female baby with a cloaca represents a particularly difficult challenge (see Chap. 16). Thirty percent of patients born with a cloaca have a hydrocolpos that may produce an extrinsic compression of the ureterovesical junction, which results in megaureter and hydronephrosis. As we mentioned in the Chap. 16, before considering drainage of the urinary tract with a nephrostomy or ureterostomy, the surgeon must drain the hydrocolpos and reevaluate the megaureter and hydronephrosis. After the hydrocolpos has been drained, the megaureter will improve or disappear, as well as the hydronephrosis. If the hydronephrosis or the megaureter persists, the baby must be subjected to further evaluation to consider the possibility of making a ureterostomy, nephrostomy, or vesicostomy. We have seen many babies with cloacas, hydronephrosis, and megaureter, subjected to unnecessary ureterostomies, nephrostomies, and/or vesicostomies, in a futile attempt to improve the hydronephrosis, without draining the hydrocolpos (see Chap. 16).
There is a small group of patients with a cloaca that are born with a very narrow (almost atresia) common channel which interferes with the emptying of the bladder. After draining the hydrocolpos, an ultrasound may show that the bladder is still full and does not empty well. An attempt to catheterize the bladder may be extremely difficult; in those cases it is justified to do a cystostomy or a vesicostomy.
Another possible indication for a vesicostomy is the case of a baby with demonstrated massive vesicoureteral reflux, megaureter, hydronephrosis, and important risk factors for poor bladder function, such as poor sacrum and tethered cord.
The newborn baby should not be taken to the operating room until the surgeon has ruled out and treated important associated urologic problems. After this, the baby is taken to the operating room to perform either a colostomy or a primary repair of the anorectal malformation.
23.3 The Importance of the Colostomy Type from the Urologic Point of View
The location and type of colostomy may have important urologic implications. It is important for the neonatologist as well as the urologist to remember this (see Chap. 5).
The colostomy that we recommend is one created at the end of the descending colon and proximal to the sigmoid loop. The purpose of that specific location is dual, first, to avoid prolapse of the proximal stoma, since the descending colon is fixed, and, second, to leave enough distal bowel (loop of the sigmoid) to allow a successful pull-through. In addition, we specifically recommend to make the distal stoma (mucous fistula) very narrow in order to avoid prolapse, but still, we believe that is important to keep it open to be able to irrigate the distal bowel and to perform radiologic studies, most specifically, a high-pressure distal colostogram. We strongly recommend separating both stomas in order to be able to put a colostomy bag, including only the proximal stoma and not the distal one. When both stomas are too close together, the mother is forced to use a single bag to cover both stomas, which may produce fecal contamination of the distal bowel and consequently of the urinary tract. Loop colostomies, therefore, from our point of view, are formally contraindicated in anorectal malformations, because they represent a source of fecal contamination of the urinary tract.
During the creation of the colostomy, we specifically recommend the surgeons to wash the bowel distal to the mucous fistula until it is completely collapsed and cleaned of meconium. If a baby receives a colostomy and after the procedure is not doing well and suffers from frequent urinary tract infections and from episodes of acidosis, it is extremely important for the physician to look at the type of colostomy that the baby has, looking for an explanation for that behavior. A loop colostomy producing frequent urinary tract infections would require an operation to separate the stomas. If the surgeon did not wash the distal bowel, sometimes the accumulation of meconium, plus mucus passing through the fistula, produces urinary tract infections. Finally, when the colostomy was opened in the transverse colon (which we consider contraindicated), the meconium may pass into the urinary tract, but also urine passes from the urinary tract into the colon where it is absorbed, producing hyperchloremic acidosis that may interfere with the growth and development of the newborn baby. When a pediatric urologist is called to see a baby with this kind of problem, he should look at the type of colostomy that the baby has.
23.4 Most Common Urologic Abnormalities in Male Patients with Anorectal Malformations
23.4.1 Absent Kidney
The most common congenital urologic defect associated to anorectal malformations is an absent, dysplastic, multicystic, or nonfunctional kidney. In our series, 106 out of our 909 male patients and 165 out of 1,123 females were born with this problem.
Figure 23.1 shows the images of a patient with an absent kidney. Figure 23.1a shows the ultrasound and Fig. 23.1b shows a renal scan. This is by far the most common type of anatomic abnormality seen in anorectal malformations. The kidney may be totally absent or rather represented by a nonfunctional multicystic and/or dysplastic kidney remnant.
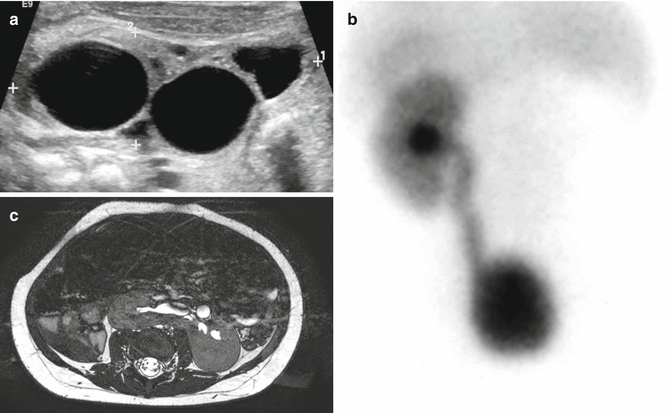

Fig. 23.1
Absent kidney (multicystic, dysplastic). (a) Ultrasound showing an image of a multicystic kidney. (b) Renal scan showing an absent kidney. (c) MRI showing a horseshoe kidney
Figure 23.2a shows the images of an ultrasound in a patient with hydronephrosis, and Fig. 23.2b shows a case with hydronephrosis and megaureter in a patient with anorectal malformation. This problem was present in 198 out of 1,123 female patients and 70 out of 909 male patients. The presence of a megaureter in patients with anorectal malformations, most of the time, represents the existence of severe vesicoureteral reflux, since the cases of ureterovesical obstruction are very unusual. Of course, we must remember that cloaca patients may have an extrinsic compression of the ureterovesical junction that is released when a dilated vagina is drained.
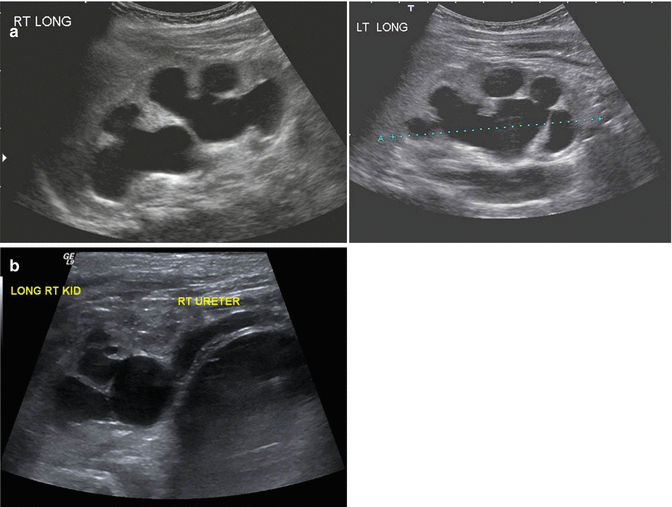

Fig. 23.2
Hydronephrosis (ultrasound image). (a) Bilateral hydronephrosis. (b) Hydronephrosis and megaureter
Figure 23.3 shows the images of a voiding cystourethrogram of a baby suffering from vesicoureteral reflux (Fig. 23.3a unilateral and Fig. 23.3b bilateral). In our series, this problem was present in 207 out of 1,123 females and 133 out of 909 males. The incidence of reflux in cases of anorectal malformations has been estimated to vary from 30 to 60 % [18–21]. In our series it occurred between 14 and 18 % of the cases.
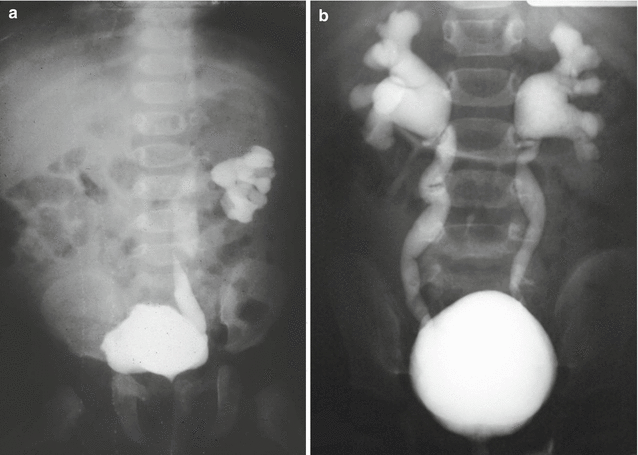

Fig. 23.3
Vesicoureteral reflux (VCUG). (a) Unilateral. (b) Bilateral
The presence of severe megaureter and vesicoureteral reflux in a newborn baby is an ominous sign. That baby will require special care to protect the kidneys, particularly in cases with real or potential neurogenic bladder, such as patients with severe sacral deformity and/or tethered cord. The best way to protect the kidneys in those babies is with a temporary vesicostomy, since trying to perform a ureteral reimplantation, with ureteral tapering, in an infant with a well-known poor functional bladder most likely will not succeed and may put the marginal renal function of the patient at risk.
There is one particular group of male patients born with a recto-bladder neck fistula. Twenty-five patients of a group of 111 patients born with a bladder neck fistula had an absent kidney, megaureter, and hydronephrosis in the opposite site (Fig. 23.4). These patients are at high risk to end up with a kidney transplant. The fact that a baby is born with hydronephrosis means that those kidneys had been suffering in utero and at birth already have a significant degree of damage. We have seen patients with hydronephrosis at birth and with a normal creatinine; they maintain the normal level of serum creatinine during the first years of life, but when they have a growth spurt, sometimes the creatinine suddenly elevates, indicating that most likely the patient was born with a limited kidney function, which became obvious when the patient grows significantly. Those cases with bladder neck fistula, single kidney, megaureter, and hydronephrosis should be considered high-risk patients.
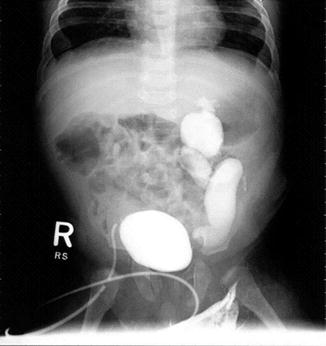

Fig. 23.4
VCUG in a patient with a single kidney with hydronephrosis and reflux
The management of vesicoureteral reflux in patients with anorectal malformations deserves a special comment. The general principles of management of this disorder, in patients without anorectal malformations, should not be extrapolated and applied for cases with anorectal malformation for the following reasons:
A.
Patients with anorectal malformations frequently suffer from a spectrum of sacral abnormalities that have a demonstrated effect on bladder function.
B.
At least 25 % of patients with ARM suffer from tethered cord, which also has a significant negative effect on bladder function.
C.
The repair of an anorectal malformation, when done inappropriately, produces different degrees of neurogenic bladder.
D.
The repair of complex cloacas includes a significant dissection between the vagina(s), the bladder neck, the trigone, and the urethra. All these have a very significant effect on the bladder function. In addition, the scar produced by that dissection may represent a serious challenge to perform a reimplantation.
It is becoming obvious that the bladder function is a fundamental factor to take into consideration when making a decision about a ureteral reimplantation. A ureteral reimplantation, done in a patient with neurogenic bladder and incapacity to empty, most likely will fail. The decision to reimplant a ureter must not be taken without a previous urodynamic evaluation. This is particularly true in patients with megaureter and hydronephrosis. The ureteral reimplantation must be performed being sure that the patient is able to empty the bladder spontaneously or with intermittent catheterization through the urethra [22] or through an appendicovesicostomy tract (Mitrofanoff principle) [23, 24].
Because of all these reasons, in general, we try to avoid a reimplantation in a newborn with reflux, megaureter, and hydronephrosis. A vesicostomy is a better option, under those circumstances; it protects the upper tracts and gives us time to reevaluate the case and make an appropriate decision.
23.4.2 Urethral Problems
Figure 23.5 shows an image of male urethrogram of a patient born with an anorectal malformation and a recto-urinary fistula. The kink shown in the study most likely corresponds to the location of the fistula. However, we believe that the VCUG is not the ideal study to determine the location of the fistula. Some patients suffer from a urethral stricture or a kink of the urethra at the same location of the junction of the rectum (fistula) (Fig. 23.6). The posterior sagittal approach represents an ideal way to repair urethral strictures. This can be done at the same time of the repair of the anorectal malformation. We have operated on our patients with this kind of problem and found relatively simple the urethral repair. The results have been good. Figure 23.7 shows operative pictures of a repair of a urethral stricture located at the same location of the fistula. A urethroplasty was performed using a flap of rectal tissue (Figs. 23.7a–d).
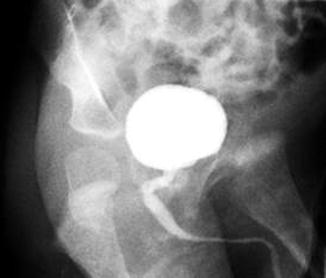
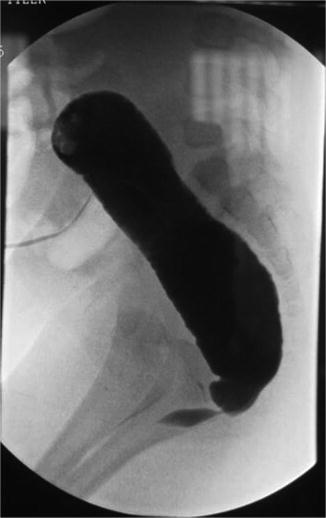
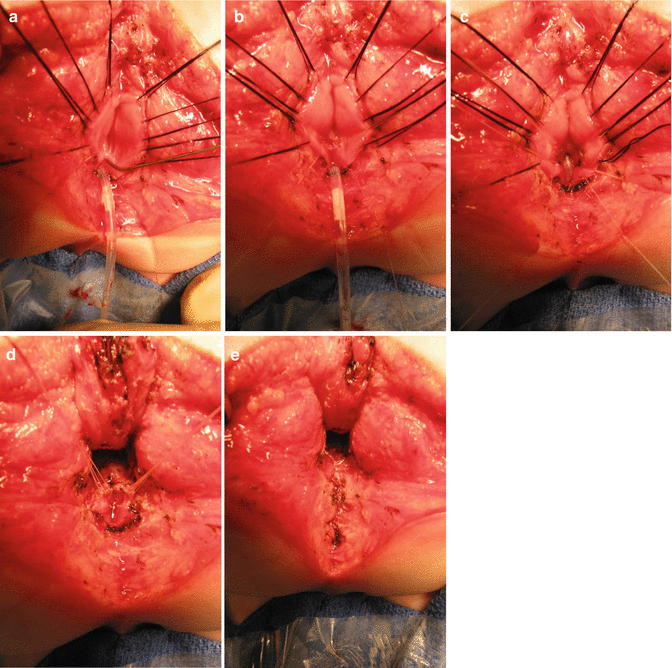

Fig. 23.5
Radiologic image of a kinked urethra. The narrowing and/or the kink is located at the junction of the rectum and the urethra (fistula site)

Fig. 23.6
Distal colostogram showing a urethral stricture at the junction of the rectum with the urethra

Fig. 23.7
Operative findings in a patient with urethral stricture. The catheter was redirected into the bladder under direct vision, and the urethral stricture was repaired. (a) The rectum open, lacrimal probe into upper the urethra, catheter emerging from the bulbar urethra. (b) The upper portion of the urethra has been enlarged with a plasty. (c) The urethral catheter was passed toward the bladder. (d) The area of stricture is expanded using rectal tissue. (e) Reconstruction finished
It is relatively common to see cases that have a very acute kink of the urethra at the same location where the rectum joins the urinary tract. When the doctors, radiologists, or surgeons try to pass a catheter, the catheter stops at that place or rather goes into the rectum. To try to overcome this obstacle, it is recommended to use a coude type of catheter (curved tip) to get access to the bladder. Sometimes, however, even with the use of this catheter, it is not possible to catheterize the bladder. In such cases it is necessary to pass the cystoscope using a wire in order to catheterize the bladder. There is a maneuver that proved to be useful to pass a Foley catheter under those circumstances. A lacrimal probe is inserted in the hole of the catheter, and the lacrimal probe is curved at its tip and can be externally directed (see Chap. 9). If the patient, in addition, suffers from a urethral stricture, then there is no way to pass the catheter into the bladder. Under those circumstances we allow the catheter to go into the rectum and approach the patient posterior sagittally; when the rectum is opened, one can see the catheter inside, and then under direct vision, one can redirect the catheter into the bladder. If the surgeon finds that there is a urethral narrowing, we have been using a little piece of the local rectal tissue to create a small flap to enlarge the area of the stricture of the urethra with good results (Fig. 23.7).
23.5 Bifid Scrotum
In our series of 909 cases of male patients with anorectal malformation, we found 102 patients suffering from a bifid scrotum (Fig. 23.8). Most of the cases of bifid scrotum occur in babies who have anorectal malformations that belong to the “bad” side of the spectrum, in other words, patients that have bladder neck or high prostatic fistulas. Therefore, a bifid scrotum should be considered as a sign of a potential complex malformation.
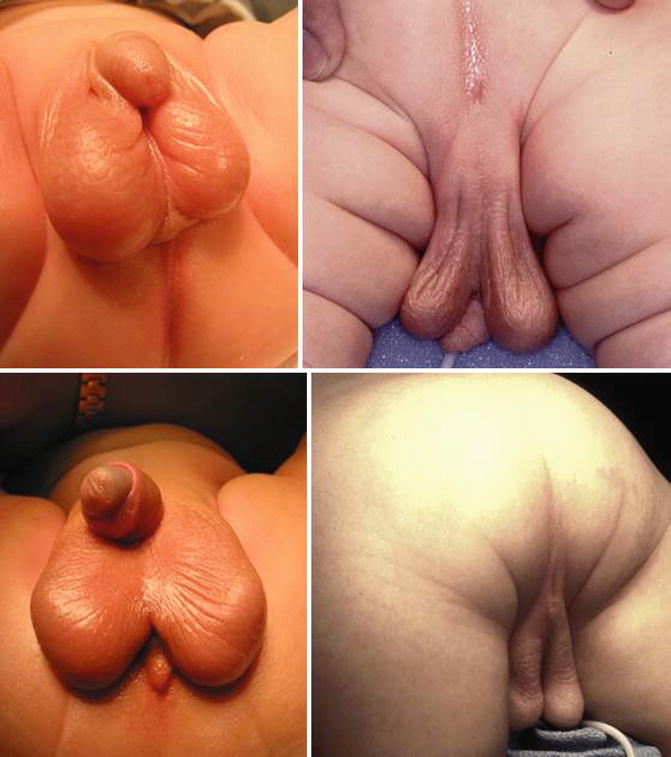

Fig. 23.8
Different types of bifid scrotum
Through the years, we learned to repair the bifid scrotum in a rather simple way, at the same time that we repair the anorectal malformation. It is important to recognize that in cases of bifid scrotum, there is a midline portion of nonelastic skin that separates both hemiscrotums (Fig. 23.9a–e). The skin of the scrotum is recognized because it has rugae and is elastic. The skin in between the hemiscrotum lacks that kind of rugae, is rather smooth, and is nonelastic. The technique that we use consists of resecting the smooth, nonelastic portion of the skin. Figure 23.9a shows how we have marked the limits that separate the smooth, nonelastic skin from the normal rugae scrotal skin. Figure 23.9b shows the incision that is made with the needle-tip cautery. Figure 23.9c shows that the smooth skin has been resected and the beginning of a two layer suture of the skin edges (Fig. 23.9d). Both layers sutured with multiple, interrupted, 6-0 long-term absorbable sutures. Figure 23.9e shows the final aspect of the repair. Later in life, it is very difficult to know that this patient had a repair of a bifid scrotum and the parents have expressed their satisfaction.
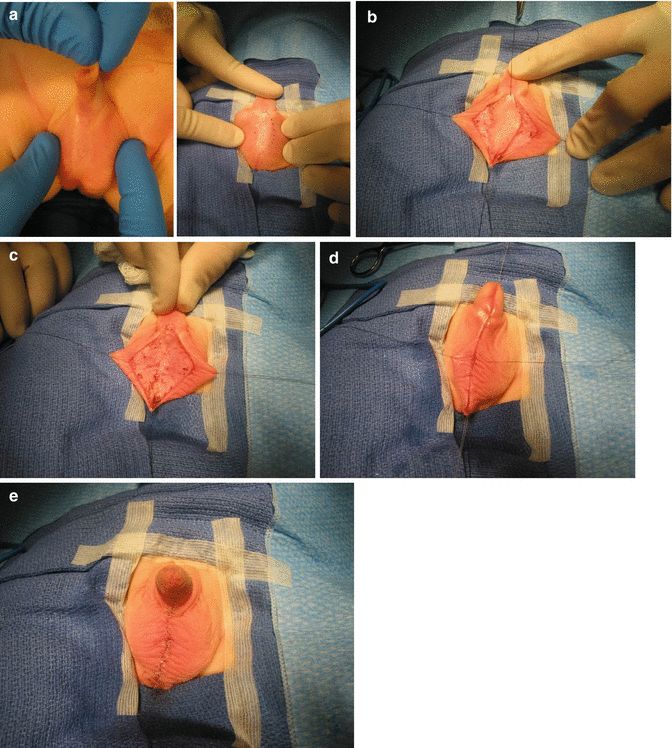

Fig. 23.9
Repair of a bifid scrotum. (a) Marking the limits of the abnormal skin. (b) Incision. (c) Resected non elastic skin. (d) Two-layer suture. (e) Final aspect
23.6 Hypospadias
Sixty of our male patients suffered from different types and severity of hypospadias. It may go from a subcoronal type (Fig. 23.10a) to an extreme type in which the urethral orifice is located immediately anterior to the anal opening (Fig. 23.10c). Extreme, severe types of hypospadias are frequently associated to penoscrotal inversion.
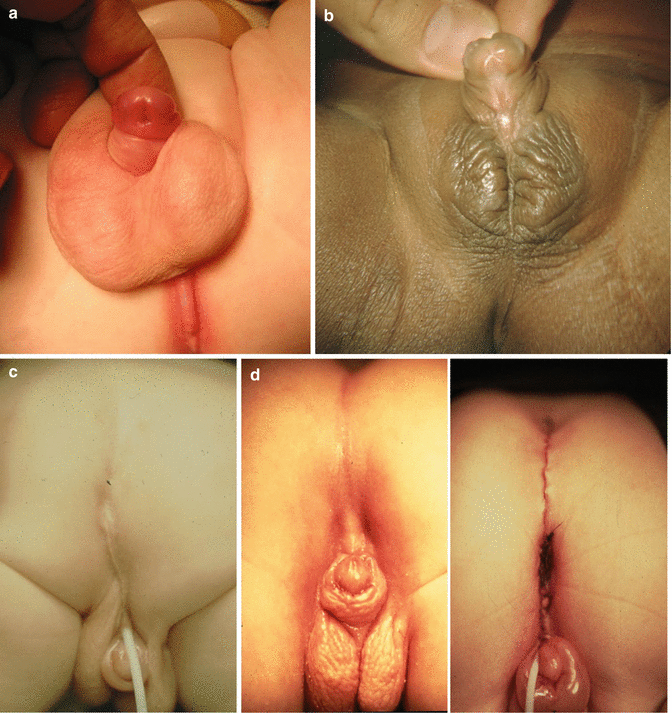

Fig. 23.10
Hypospadias. (a) Subcoronal. (b) Mid-shaft. (c) Perineal. (d) Repaired anorectal malformation with severe hypospadias. The urethra was originally located next to the anal opening. The urethral opening was moved forward, and the anus was moved back
The timing for the repair of hypospadias is something rather controversial. Some pediatric urologists like to repair the hypospadias before the colostomy is closed, to avoid fecal contamination during the postoperative period. Some others prefer to wait until the patient is older to be able to deal with better tissues and have more chances of success. From the point of view of the pediatric surgeon, it is important to identify the most severe type of hypospadias (Fig. 23.10c, d) in which the urethral orifice is located immediately anterior to the anal opening. The surgeon must keep in mind that the rectum and urethra share a common wall, usually in the area of the spongiosum tissue. Those babies have a strictured anal opening located anterior to the center of the sphincter, and therefore, the operation will consist in moving the anus back and simultaneously to correct the severe chordee. In order to do that, the surgeons have to work on the common wall between the rectum and the urethra and move, if possible, the urethra a little forward. The idea is to create a reasonable space between the anal opening and the urethral opening and move the penis forward, away from the anal opening. After this repair, the urologist will take care of the problem of repairing the rest of the perineal urethra, as well as the penile urethra. Figure 23.10d shows the external aspect of a patient that underwent a repair of an anorectal malformation and separation of the rectum from the urethra in a severe perineal hypospadias.
23.7 Ectopic Ureters in Males
This malformation has been reported in cases without ARM [25]. It seems to be relatively rare. We believe that it occurs more often in patients with anorectal malformations.
Twenty-one of our male patients suffer from some form of ureteral ectopia. In patients with anorectal malformations, we have seen that the ureters have a tendency to open ectopically, in the line that goes from the normal location of the ureter in the trigone toward the bladder neck. It is relatively common to see patients with a ureteral orifice located closer to the bladder neck. There are however cases in whom, definitely, we see the ureter opening in the bladder neck (Fig. 23.11a). The opening of the ureter in the bladder neck frequently is associated with some degree of ureteral obstruction and megaureter. In addition, the presence of the ureter in that abnormal location may interfere with the closure of the bladder neck, producing a certain degree of bladder neck incapacity to hold the urine. We have relocated the ureters out of the bladder neck with success, recovering urinary control in patients that were leaking urine.
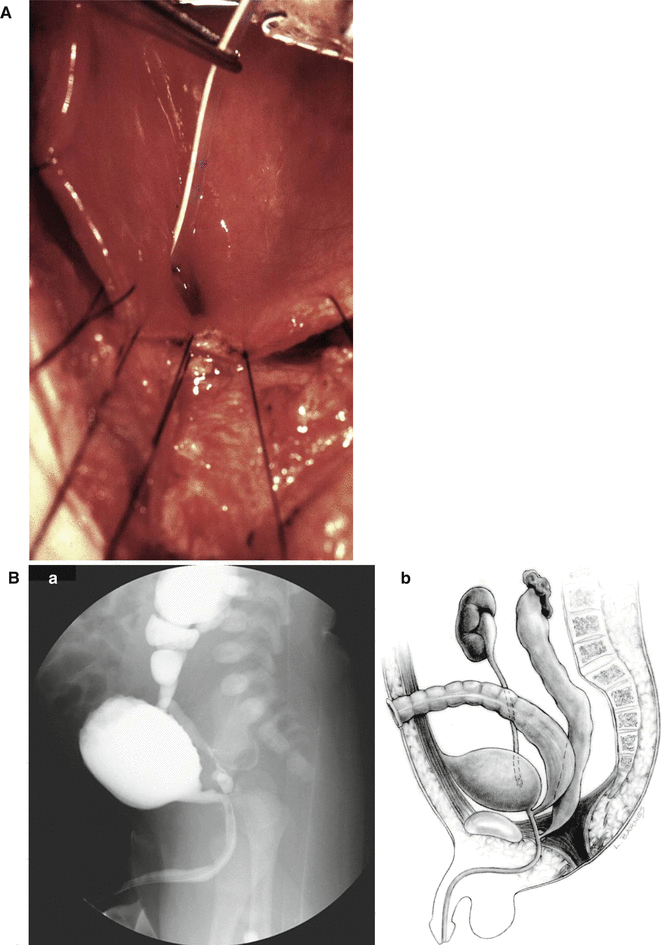

Fig. 23.11
Diagram showing ectopic ureters. (A) Connected to the bladder neck. (B) Connected to the posterior urethra. (a) Voiding cystourethrogram. (b) Diagram
The most severe type of ureteral ectopia in males occurs when the ureters are connected to the posterior urethra (Fig. 23.11b). In these cases, the patient suffers from severe megaureter, and they frequently have severe damage to the kidney that usually ends up with a nephrectomy. The surgeon should be alerted about the fact that the megaureter connected to the posterior urethra may give the false intraoperative impression of the rectum. In one specific case, the patient was operated in another city without a distal colostogram, and the surgeon entered posterior sagittally looking for the rectum, found a “tubular structure,” separated it from the posterior urethra, and pulled it down, believing it was the rectum, and it turned out to be a megaureter. The patient had actually a recto-bladder neck fistula.
As expected, the frequency of ectopic ureter is higher in patients with higher and more complex malformations. If one already made the diagnosis of an ectopic ureter located in the posterior urethra, the posterior sagittal approach represents a golden opportunity to identify the megaureter, separate it from the posterior urethra, close the posterior urethra, and push the megaureter up into the pelvis, to be recovered through a small incision in the groin, where it can be connected to the skin as a ureterostomy. It will take several months for us to determine the degree of renal damage that the kidney has and whether or not the patient will benefit from a nephrectomy or a ureteral reimplantation.
23.8 Ectopic Ureters in Females
All that we said about the ectopic ureters in the trigone and bladder neck is true for female patients. The ureters also may be ectopically connected to the urethra in a female. Figure 23.12 shows a picture taken during a transpubic approach of a female patient with a complex malformation. It would be very difficult to separate the ureter with a different type of approach. We use the transpubic approach in cases that we believe would be very difficult to approach in a different way.
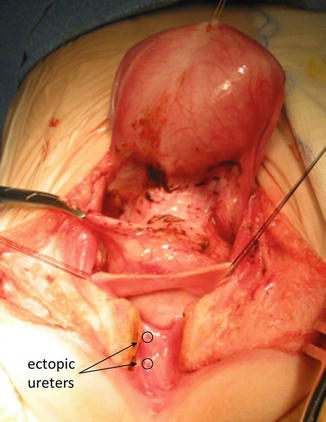

Fig. 23.12
Ectopic ureters in the urethra of a female patient with a complex malformation. Intraoperative picture taken during a transpubic approach
In patients with cloaca, it is also relatively common to find ureters connected ectopically to the vagina. Again, most of the time these ureters belong to severely damaged kidneys.
23.9 Ectopic Vas Deferens
This rare anomaly has been reported in patients without anorectal malformation [26–28]. The most common clinical manifestation has been orchiepididymitis. However, we found more publications related to this malformation in cases of anorectal malformations [29–38].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








