Small Intestine
3.1 Crushed Brunner glands vs. Whipple disease
3.2 Crushed Brunner glands vs. Mesenchymal lesions in small bowel mucosa
3.3 Chronic duodenitis vs. Intracytoplasmic lipid
3.4 Chronic duodenitis vs. Abetalipoproteinemia
3.5 Atypical stromal cells in ulcers vs. Small bowel sarcomas
3.6 Whipple disease vs. Mycobacterium avium complex
3.7 Common variable immunodeficiency vs. Normal small intestine
3.8 Autoimmune enteropathy vs. Normal small bowel
3.9 Autoimmune enteropathy vs. Common variable immunodeficiency
3.10 Celiac disease vs. Small bowel bacterial overgrowth of enteric contents
3.11 Celiac disease vs. Common variable immunodeficiency
3.12 Collagenous sprue/enteritis vs. Radiation change
3.13 Behcet syndrome affecting small bowel vs. Crohn disease
3.14 Microvillus inclusion disease vs. Intracytoplasmic lipid
3.15 Microvillus inclusion disease vs. Tufting enteropathy
3.17 Infectious enteritis vs. Crohn disease
3.18 Pyloric gland adenoma vs. Tubular adenoma
3.19 Nodular duodenitis vs. Tubular/tubulovillous adenoma
3.20 Peutz-Jeghers polyp vs. Juvenile polyp
3.21 Cronkhite-Canada Polyps vs. Juvenile polyps
3.22 Primary small bowel cancer vs. Metastases to small bowel
3.23 Small bowel follicular lymphoma vs. Reactive lymphoid hyperplasia
3.24 Small bowel follicular lymphoma vs. Mucosa-associated lymphoid tissue (MALT) lymphoma
3.25 Small bowel follicular lymphoma vs. Mantle cell lymphoma
3.26 Burkitt lymphoma vs. Diffuse large B-cell lymphoma
3.27 Enteropathy-associated T-cell lymphoma vs. Enteropathy/celiac disease
3.28 Systemic mastocytosis vs. Langerhans cell histiocytosis
3.29 Small bowel gastrointestinal stromal tumor vs. Inflammatory fibroid polyp
3.30 Epithelioid small bowel gastrointestinal stromal tumor vs. Carcinoma
3.31 Small bowel gastrointestinal stromal tumor vs. Leiomyosarcoma
3.33 Small bowel gastrointestinal stromal tumor vs. Melanoma
3.34 Clear cell sarcoma–like tumor (malignant gastrointestinal neuroectodermal tumor) vs. Melanoma
3.35 Small bowel gastrointestinal stromal tumor vs. Mesenteric fibromatosis
3.36 Mesenteric Fibromatosis vs. Sclerosing mesenteritis
3.37 Mesenteric Fibromatosis vs. Calcifying fibrous tumors
3.38 Duodenal well-differentiated neuroendocrine carcinoid tumor vs. Duodenitis
3.1 Crushed Brunner glands vs. Whipple disease
| Crushed Brunner Glands | Whipple Disease | |
|---|---|---|
| Age/Gender | Can be encountered in any duodenal biopsy (Brunner glands are specific to the duodenum) | Adults; male predominance |
| Location | Any part of duodenum | Anywhere in the small bowel or anywhere in the body, for that matter |
| Symptoms | None. This is an artifact of biopsy | Depends on presentation—classically malabsorption-associated symptoms but patients can present with central nervous system manifestations, adenopathy, or rheumatologic symptoms |
| Signs | None | Site dependent, classically with diarrhea and wasting but patients with central nervous system disease can present with seizures or other central nervous system signs |
| Etiology | Damage to the duodenal mucosa as a result of the act of performing the biopsy and very commonly encountered | Infection with Tropheryma whipplei |
| Histology | ||
| 1. Nodules of crushed Brunner glands, often in continuity with well-preserved Brunner glands (Fig. 3.1.1). Lacteals are not dilated 2. PAS/AB strongly positive in “foamy” pattern (Fig. 3.1.2) 3. Negative Whipple bacillus immunostain but not necessary to perform this stain (Fig. 3.1.3) | 1. Small bowel samples show expansion of the lamina propria with foamy macrophages and multiple dilated spaces (Fig. 3.1.4) 2. PAS/AB staining shows granular cytoplasm in the distended macrophages (Fig. 3.1.5) 3. Whipple bacillus immunolabeling is reactive (Fig. 3.1.6) | |
| Special studies |
|
|
| Treatment | None | Antibiotics: Doxycycline and hydroxychloroquine for 12 months followed by lifelong treatment with doxycycline, as potentially fatal relapses can occur. T. whipplei seems to be an opportunistic bacterium that causes chronic infections in susceptible patients with as yet unknown predisposing factors |
| Prognosis | Not applicable. This is an incidental finding | The infection is difficult to eradicate, and, as above, lifelong antibiotic treatment is needed |
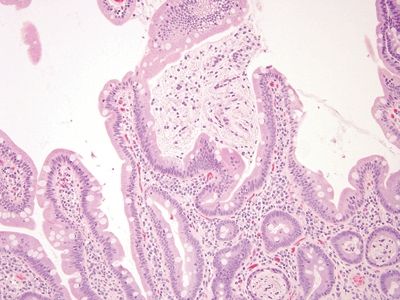
Figure 3.1.1 Duodenum, crushed Brunner glands. Note the area in the center of the field. There is a crushed Brunner gland in the lumen in close association with surface mucosa. Material from Bruner glands is also in the lumina of some of the crypts at the lower right. Note also the cellularity of duodenal lamina propria.
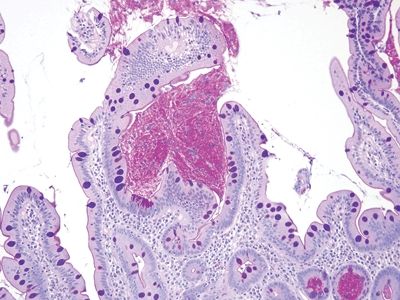
Figure 3.1.2 Duodenum, crushed Brunner glands, PAS/AB stain. This is a PAS/AB of the same are seen in Figure 3.1.1. Brunner glands stain magenta. There are small compact nuclei and abundant neutral mucin with a slightly fibrillary appearance.
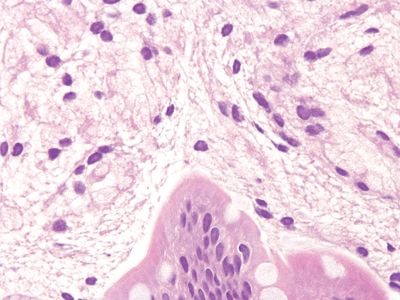
Figure 3.1.3 Duodenum, crushed Brunner glands. At very high magnification, the slightly fibrillary crushed cytoplasm appears vacuolated as well.
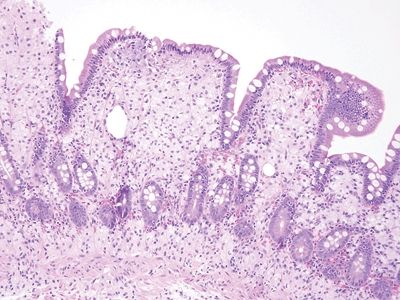
Figure 3.1.4 Whipple disease. The lamina propria is expanded with foamy macrophages, but the lumina of the crypts are empty. Note the dilated spaces, a characteristic feature of Whipple disease.
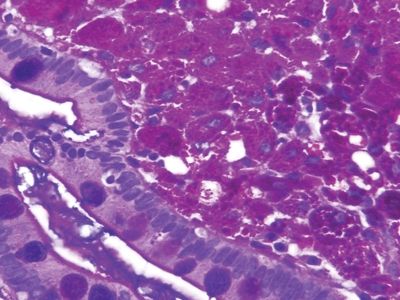
Figure 3.1.5 Whipple disease, PAS/AB stain. The macrophages contain coarse granular deposits in contrast to the delicate fibrillary material in crushed Brunner glands.
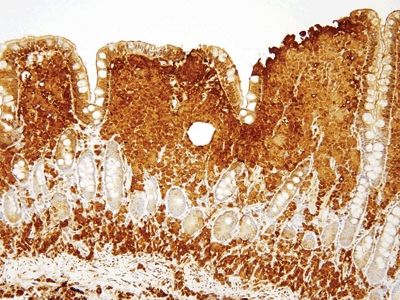
Figure 3.1.6 Whipple disease, immunolabeling. There is strong coarsely granular staining in untreated cases, which becomes less prominent after treatment.
3.2 Crushed Brunner glands vs. Mesenchymal lesions in small bowel mucosa
| Crushed Brunner Glands | Mesenchymal Lesions in Small Bowel Mucosa | |
|---|---|---|
| Age/Gender | Can be encountered in any duodenal biopsy (Brunner glands are specific to the duodenum) | Male predominance; usually middle-aged males |
| Location | Any part of duodenum | Throughout the small bowel |
| Symptoms | None. This is an artifact of biopsy | Related to the size and location. For example, inflammatory fibroid polyp presents with intussusception and therefore with symptoms of obstruction. Leiomyomas are associated with the muscularis mucosae in the small bowel. Clear cell sarcoma–like tumors arise in the ileum in young patients. Nerve sheath tumors are the most likely to appear myxoid and resemble Brunner glands. Gastrointestinal stromal tumors (GISTs) are usually not seen on mucosal biopsies, but when they are seen, they are more cellular than crushed Brunner glands |
| Signs | None | Those of obstruction in patients whose tumors grow large or cause intussusception |
| Etiology | Damage to the duodenal mucosa as a result of the act of performing the biopsy and very commonly encountered | Various tumor types |
| Histology | ||
| 1. Nodules of crushed Brunner glands, often in continuity with well-preserved Brunner glands (Fig. 3.2.1). They involve either the lamina propria or the submucosa. These are very common in biopsies 2. PAS/AB strongly positive in “foamy” pattern (Fig. 3.2.2) 3. Negative S100 protein immunostain but not necessary to perform this stain (Fig. 3.2.3) | 1. This is a leiomyoma arising in association with muscularis mucosae (Fig. 3.2.4) 2. This nerve sheath tumor lacks the bubbly cytoplasm present in crushed Brunner glands (Fig. 3.2.5) 3. This nerve sheath tumor expresses S100 protein (Fig. 3.2.6) | |
| Special studies |
|
|
| Treatment | None | Depends on tumor type. Most mesenchymal lesions seen on mucosal biopsies of the small bowel (and thus in the differential diagnosis of Brunner glands) are clearly different from Brunner glands |
| Prognosis | Not applicable. This is an incidental finding | Depends on tumor type. Inflammatory fibroid polyp and most nerve sheath tumors and leiomyoma are all benign, whereas some GISTs are malignant |
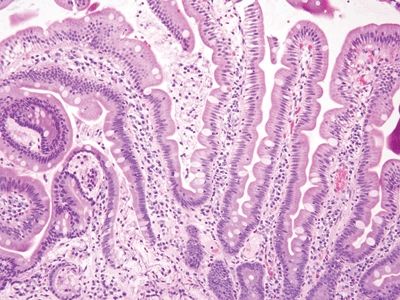
Figure 3.2.1 Duodenum, crushed Brunner glands. The material from the crushed Brunner glands is in the lumina of the crypts extending as sloughed material onto the surface but also in contiguity with more well-formed Brunner glands at the lower left.
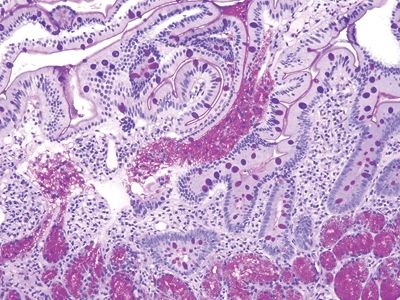
Figure 3.2.2 Duodenum, crushed Brunner glands, PAS/AB. There are well-preserved Brunner glands in the deeper part of the sample, and they have extruded themselves into the lumen.
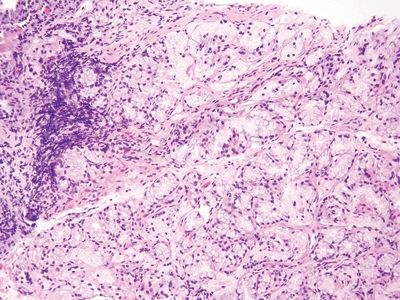
Figure 3.2.3 Duodenum, crushed Brunner glands. These glands are less crushed than are those in other images. The Brunner glands become more prominent after cycles of damage and repair to the duodenal mucosa.
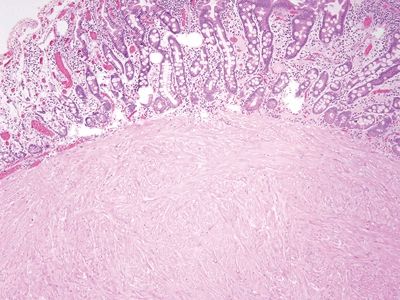
Figure 3.2.4 Leiomyoma. This brightly eosinophilic lesion has arisen in association with the muscularis mucosae.
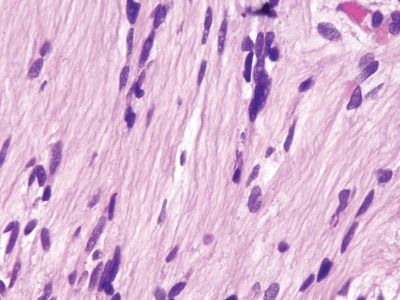
Figure 3.2.5 Nerve sheath tumor. Note that the nuclei differ dramatically from those in the crushed Brunner glands and the fibrillary cytoplasm appears less vacuolated.
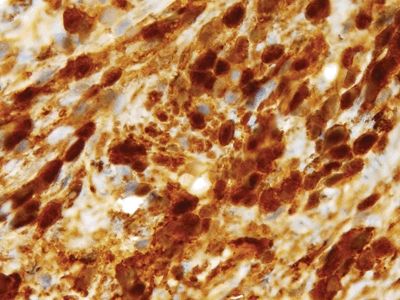
Figure 3.2.6 Nerve sheath tumor. The strong S100 protein staining is characteristic.
3.3 Chronic duodenitis vs. Intracytoplasmic lipid
| Chronic Duodenitis | Intracytoplasmic Lipid in Duodenal Biopsies | |
|---|---|---|
| Age/Gender | Classically encountered in associated with H. pylori gastritis but can be encountered for any of a variety of reasons. As such there is a wide age range and no real gender predilection | Typically adults; no gender predilection |
| Location | Anywhere in duodenum but typically in the bulb (the first part receiving the gastric contents) | No specific location in duodenum |
| Symptoms | None from the chronic duodenitis itself but from the cause of the duodenitis. For example, persons with H. pylori gastritis may have pain from gastric ulcers and have duodenitis on biopsies. If there is chronic duodenitis from Crohn disease, the patient may have diarrhea, abdominal pain, and small bowel obstruction. If the patient has chronic duodenitis from taking nonsteroidal anti-inflammatory drugs, the patient may have vague abdominal pain | Usually none except when associated with abetalipoproteinemia |
| Signs | No specific signs. The duodenum may appear nodular at endoscopy | Usually none unless in case of abetalipoproteinemia |
| Etiology | Cycles of damage and repair to the duodenal mucosa, which results in gastric mucin cell metaplasia | In most cases, dietary indiscretion. Patients are advised to refrain from eating prior to upper endoscopy, but some are noncompliant so absorbed lipid may be seen on endoscopic samples |
| Histology | ||
| 1. The key finding is gastric mucin cell metaplasia of the surface epithelium (Fig. 3.3.1). Often, there are reparative epithelial changes as well 2. At higher magnification, there is a surface cap of neutral mucin in the epithelium. This contrasts to the normal brush border seen in the duodenum (Fig. 3.3.2) 3. PAS/AB staining highlights neutral mucin in the metaplastic surface epithelium (Fig. 3.3.3) | 1. The brush border is intact, but bubbly material can be seen in enterocytes. It is not well demarcated like mucin droplets in mucin cell metaplasia
(Fig. 3.3.4) 2. At higher magnification, the adjoining cells lack cytoplasmic lipid in most patients and the finding is focal (Fig. 3.3.5) 3. PAS/AB staining is negative (Fig. 3.3.6) | |
| Special studies |
|
|
| Treatment | None for the metaplasia itself. Any treatment is directed at the underlying etiology of the metaplasia—for example, treatment of H. pylori gastritis | None |
| Prognosis | Excellent for the metaplasia. The underlying condition responsible for the metaplasia can be addressed if it is known | Incidental finding in most cases |
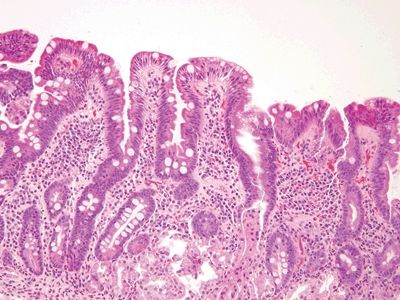
Figure 3.3.1 Chronic duodenitis. Regardless of the type of injury, when the duodenal mucosa is injured, it responds with gastric-type metaplasia. This can be recognized by noticing the gastric-type surface mucin in the area just to the right of center.
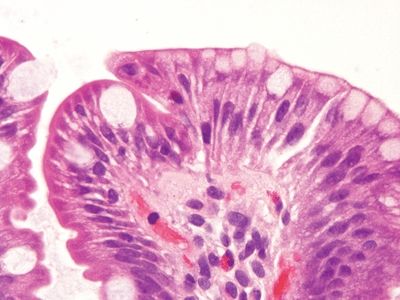
Figure 3.3.2 Chronic duodenitis, gastric mucin cell metaplasia. Compare the smaller caps of apical gastric-type metaplastic mucin at the right to the appearance of the mucin in the goblet cells to the left.
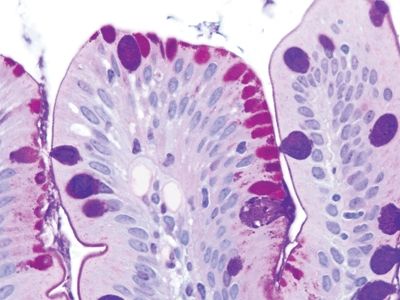
Figure 3.3.3 Chronic duodenitis, gastric mucin cell metaplasia, PAS/AB. The apical caps of neutral mucin are magenta and on the right, and the quantity of mucin is less than that in the blue/purple goblet cells toward the left side of the field.
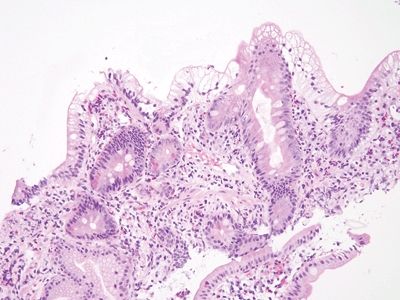
Figure 3.3.4 Intracytoplasmic lipid in duodenal biopsies (“lipid hang-up”). Usually this is a reflection of “cheating” on the NPO requirement prior to upper endoscopy. The surface absorptive cells are stuffed with lipid (not neutral mucin) that has been absorbed, but the brush border is intact.
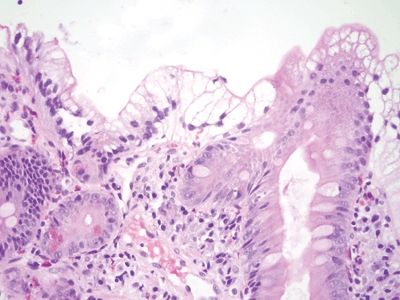
Figure 3.3.5 Intracytoplasmic lipid in duodenal biopsies (“lipid hang-up”). Higher magnification of the area shown in Figure 3.3.4.
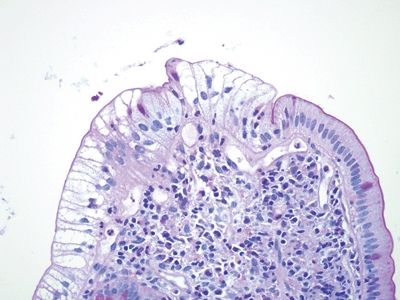
Figure 3.3.6 Intracytoplasmic lipid in duodenal biopsies (“lipid hang-up”), PAS/AB stain. There is no mucin in the cytoplasm, only nonstaining lipids, but the brush border is intact.
3.4 Chronic duodenitis vs. Abetalipoproteinemia
| Chronic Duodenitis | Abetalipoproteinemia | |
|---|---|---|
| Age/Gender | Classically encountered in associated with H. pylori gastritis but can be encountered for any of a variety of reasons. As such, there is a wide age range and no real gender predilection | Abetalipoproteinemia (ABL) and homozygous hypobetalipoproteinemia (HHBL) present in infancy with chronic diarrhea and failure to thrive |
| Location | Anywhere in the duodenum but typically in the bulb (the first part receiving the gastric contents) | No specific location in duodenum but most pronounced there since it is the site of absorption |
| Symptoms | None from the chronic duodenitis itself but from the cause of the duodenitis. For example, persons with H. pylori gastritis may have pain from gastric ulcers and have duodenitis on biopsies. If there is chronic duodenitis from Crohn disease, the patient may have diarrhea, abdominal pain, and small bowel obstruction. If the patient has chronic duodenitis from taking nonsteroidal anti-inflammatory drugs, the patient may have vague abdominal pain | Diarrhea initially. The disease leads to hypocholesterolemia and malabsorption of fat-soluble vitamins and thus to retinal degeneration, neuropathy, coagulopathy, and hepatic steatosis |
| Signs | No specific signs. The duodenum may appear nodular at endoscopy | Retinal degeneration, neuropathy, and coagulopathy. Acanthocytes are seen in peripheral blood, and the patients have nearly absent LDL-C, TG, and apo B |
| Etiology | Cycles of damage and repair to the duodenal mucosa, which results in gastric mucin cell metaplasia | Patients have improper packaging and secretion of apolipoprotein (apo) B–containing lipoprotein particles due to mutations either in both alleles of the MTP (alias MTTP) gene encoding microsomal triglyceride transfer protein (MTP) or both alleles of the APOB gene itself in the case of ABL and HHBL. Definitive diagnosis involves sequencing the MTP and APOB genes, for which >30 and >60 mutations have been described for ABL and HHBL, respectively |
| Histology | ||
| 1. The key finding is gastric mucin cell metaplasia of the surface epithelium
(Fig. 3.4.1). Often, there are reparative epithelial changes as well 2. At higher magnification, there is a surface cap of neutral mucin in the epithelium. This contrasts to the normal brush border seen in the duodenum (Fig. 3.4.2) 3. PAS/AB staining highlights neutral mucin in the metaplastic surface epithelium (Fig. 3.4.3) | 1. The brush border is intact, but bubbly material can be seen in enterocytes in a diffuse pattern with no area spared. There can be villous blunting, and all enterocytes are stuffed with lipid (Fig. 3.4.4) 2. At higher magnification, the adjoining all the cells contain the lipid (Fig. 3.4.5) 3. PAS/AB staining is negative (Fig. 3.4.6) | |
| Special studies |
| |
| Treatment | None for the metaplasia itself. Any treatment is directed at the underlying etiology of the metaplasia—for example, treatment of H. pylori gastritis | Low-fat diet and supplementation with essential fatty acids and oral fat-soluble vitamins |
| Prognosis | Excellent for the metaplasia. The underlying condition responsible for the metaplasia can be addressed if it is known | Strict adherence to diet can reduce neurologic damage and halt progression, but overall life expectancy is reduced. Some patients have successful pregnancies. Many patients live until the sixth decade |
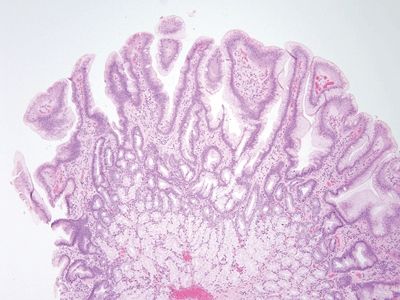
Figure 3.4.1 Chronic duodenitis. This lesion appeared nodular. The eye-catching feature is that there is extensive gastric mucin cell metaplasia on the surface.
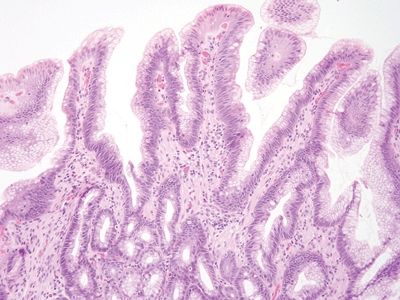
Figure 3.4.2 Chronic duodenitis. The surface mucin is very different from intracytoplasmic lipid.
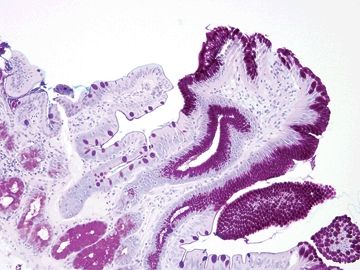
Figure 3.4.3 Chronic duodenitis, PAS/AB. This stain shows a striking contrast between the absorptive epithelium with a crisply delineated brush border on the left and the metaplastic gastric-type surface showing crisply demarcated apical mucin caps on the right.
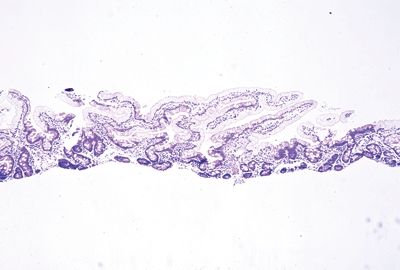
Figure 3.4.4 Abetalipoproteinemia. This is essentially completely dramatic “lipid hang-up” in which essentially the entire sample shows the intracytoplasmic lipid in the surface absorptive. It appears similar to the findings in Figures 3.3.4–3.3.6 but is diffusely present in the sample.
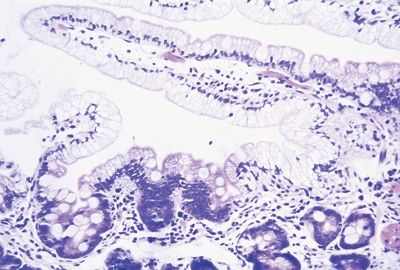
Figure 3.4.5 Abetalipoproteinemia. Higher magnification of the case seen in Figure 3.4.4. Note that the surface brush border is intact, but the absorptive cells are stuffed with lipid.
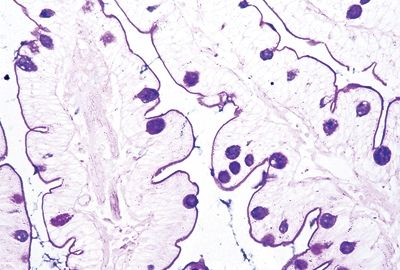
Figure 3.4.6 Abetalipoproteinemia, PAS/AB stain. The brush border and goblet cells are highlighted, but the material in the absorptive cells is not mucin but lipid, so it shows no staining.
Figure 3.4.7 Abetalipoproteinemia. This peripheral blood smear shows striking acanthocytosis.
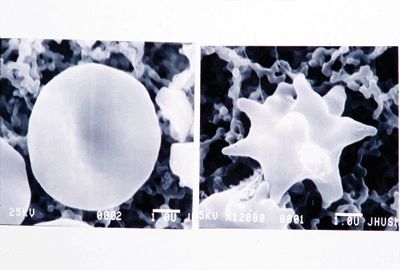
Figure 3.4.8 Abetalipoproteinemia. Ultrastructure of an acanthocyte (right) compared to a normal erythrocyte (left).
3.5 Atypical stromal cells in ulcers vs. Small bowel sarcomas
| Atypical Stromal Cells in Ulcers | Small Bowel Sarcomas | |
|---|---|---|
| Age/Gender | Typically adult | Predominance in older adults; male predominance overall for most lesions (leiomyosarcoma and malignant gastrointestinal stromal tumor/GIST). Clear cell sarcoma–like tumor predominates in young adults |
| Location | Anywhere in the small bowel | GISTS and leiomyosarcomas seem to be more common in the jejunum and ileum than duodenum, presumably since the jejunum and ileum are longer. GISTs are far more common than leiomyosarcomas overall. Clear cell sarcoma–like tumors predominate in the ileum and are extremely rare. Occasionally, other sarcomas (such as dedifferentiated liposarcomas) can extend into the small intestines and erode into the mucosa |
| Symptoms | None attributable to the atypical stromal cells per se. Whatever has resulted in the ulcer (including inflammatory bowel disease or ulcers associated with nonsteroidal anti-inflammatory drugs) | Typically, patients present with abdominal pain or symptoms of obstruction |
| Signs | Endoscopic ulcers may be seen and biopsied | A mass is identified |
| Etiology | Atypical stromal cells in ulcers are attributed to hypoxia | Small intestinal GISTs are related to KIT mutations and found sporadically but also without KIT mutations in patients with neurofibromatosis, type 1 (the GISTS found in children and patients with Carney syndrome or Carney-Stratakis syndrome are in the stomach). Leiomyomas arise sporadically, as do clear cell sarcoma–like tumors; the latter are associated with EWS gene fusions |
| Histology | ||
| 1. At low magnification, atypical stromal cells are seen at the interface of the nonviable exudate and underlying granulation tissue (Fig. 3.5.1) 2. Atypical stromal cells have smudged nuclei and a low nuclear-to-cytoplasmic ratio (Figs. 3.5.2–3.5.4) | 1. In general, in contrast to atypical stromal cells, the lesional cells in sarcomas do not show zonation and form a bulky mass (Figs. 3.5.5–3.5.8) | |
| Special studies |
|
|
| Treatment | None | Resection and targeted therapy or chemotherapy, depending on sarcoma type |
| Prognosis | These reactive cells are related to the etiology of the ulcer and measures to address the ulcer result in their disappearance | The overall prognosis depends on tumor size, type, and grade as well as the availability of targeted therapy |
Figure 3.5.1 Atypical stromal cells in ulcers. This is a resection from a patient with Crohn disease. There is an ulcer at the top of the image. Even at this low magnification, enlarged nucleus can be seen in the bed of the ulcer at the interface between the hypoxic ulcer and the viable granulation tissue below. This architectural pattern would be unusual for a sarcoma.
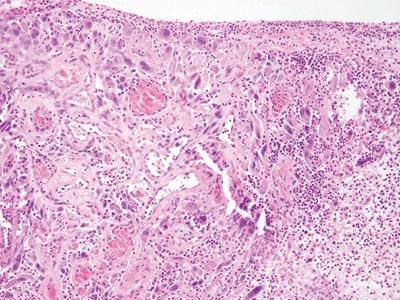
Figure 3.5.2 Atypical stromal cells in ulcers. The atypical cells proliferate at the interface of viable and nonviable tissue.
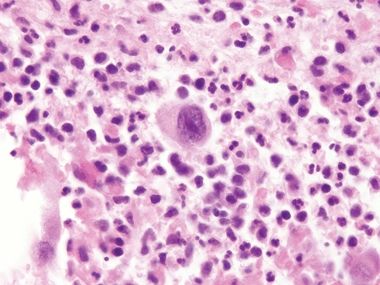
Figure 3.5.3 Atypical stromal cells in ulcers. At high magnification, the cells are large and atypical with “smudged” nuclei and a relatively low nuclear-to-cytoplasmic ratio. Note the necroinflammatory debris of the ulcer around the atypical cell.
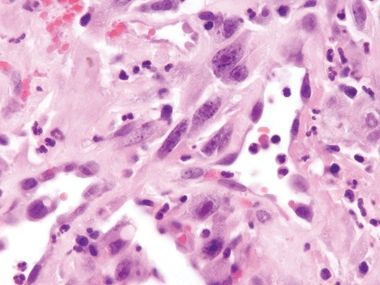
Figure 3.5.4 Atypical stromal cells in ulcers. These cells manifest macronucleoli but otherwise degenerative chromatin.
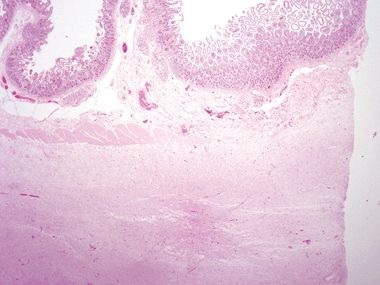
Figure 3.5.5 Dedifferentiated liposarcoma involving small bowel. This lesion has extended from the retroperitoneum into the muscularis propria but not into the mucosa (sarcomas typically do not involve the small bowel mucosa). Note that it is a large confluent mass rather than an atypical rind of enlarged spindle cells.
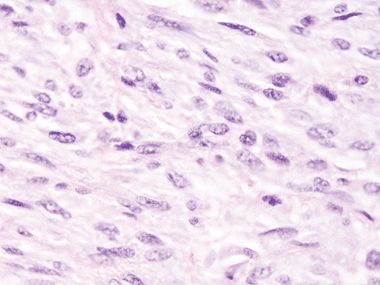
Figure 3.5.6 Dedifferentiated liposarcoma involving small bowel. The malignant cells feature nuclear heterochromatin.
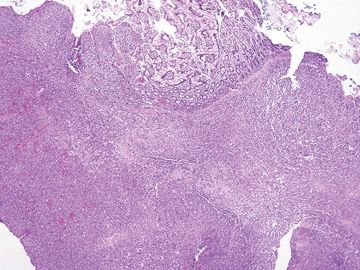
Figure 3.5.7 Angiosarcoma involving small bowel. This lesion proliferates in a confluent fashion, extending from the submucosa into the lamina propria on this mucosal biopsy.
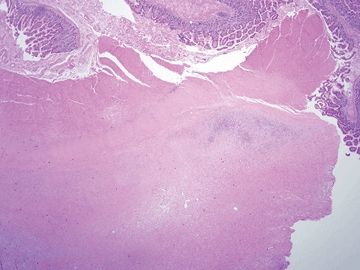
Figure 3.5.8 Low-grade fibromyxoid sarcoma involving small bowel. This tumor has extended into the muscularis. It is difficult to diagnose specifically on this low-power view, but the principle is that sarcomas for confluent masses rather than an atypical layer as in the pseudosarcomatous cells seen in Figures 3.5.1–3.5.4.
3.6 Whipple disease vs. Mycobacterium avium complex
| Whipple Disease | Mycobacterium Avium Infection | |
|---|---|---|
| Age/Gender | Adults; male predominance | Typically adults; strong male predominance since most common in patients infected with human immunodeficiency virus (HIV). May be encountered in any immunosuppressed person, including patients with inflammatory bowel disease taking immune modulation treatments. Patients commonly manifest mycobacterium infection in the duodenum. Prophylaxis against this infection is suggested in persons with HIV infection with CD4 counts of <50 cells/μL. The incidence of such immunosuppression has declined with the advent of combination antiretroviral therapy |
| Location | Anywhere in the small bowel or anywhere in the body, for that matter | Duodenum |
| Symptoms | Depends on presentation—classically malabsorption-associated symptoms but patients can present with central nervous system manifestations, adenopathy, or rheumatologic symptoms | Diarrhea and wasting |
| Signs | Site dependent, classically with diarrhea and wasting but patients with central nervous system disease can present with seizures or other central nervous system signs | EGD discloses white nodules and plaques in the duodenum |
| Etiology | Infection with Tropheryma whipplei | Mycobacterium avium spp. |
| Histology | ||
| 1. Small bowel samples show expansion of the lamina propria with foamy macrophages and multiple dilated spaces (Fig. 3.6.1) 2. PAS/AB staining shows granular cytoplasm in the distended macrophages (Fig. 3.6.2) 3. Whipple bacillus immunolabeling is reactive (Fig. 3.6.3) | 1. Villi are distended with foamy macrophages, but there are no cystic spaces/dilated lacteals (Fig. 3.6.4) 2. PAS/AB staining shows a delicate material within the macrophages and “negative” outlines of the slender bacterial forms (Fig. 3.6.5) 3. Whipple stain is negative, but acid-fast staining highlights organisms (Fig. 3.6.6) | |
| Special studies |
|
|
| Treatment | Antibiotics: Doxycycline and hydroxychloroquine for 12 months followed by lifelong treatment with doxycycline, as potentially fatal relapses can occur. T. whipplei seems to be an opportunistic bacterium that causes chronic infections in susceptible patients with as yet unknown predisposing factors | Azithromycin or clarithromycin as prophylaxis, which can be discontinued once CD4 counts are >100 cells/μL for greater than or equal to 3 months. For treatment, clarithromycin is recommended as primary therapy |
| Prognosis | The infection is difficult to eradicate, and, as above, lifelong antibiotic treatment is needed | Excellent if combined with a goal or immune restoration |
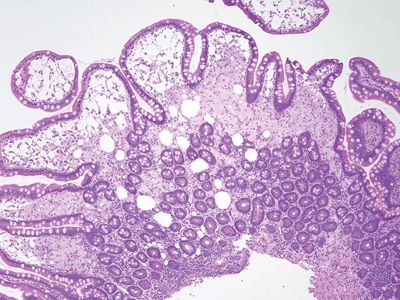
Figure 3.6.1 Whipple disease. The lamina propria is stuffed with macrophages, and there are numerous dilated spaces as well.
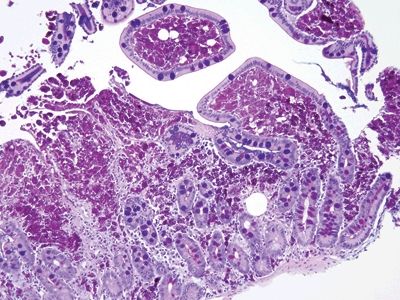
Figure 3.6.2 Whipple disease, PAS/AB. Note the “chunky,” coarse granular appearance of the contents of the macrophages.
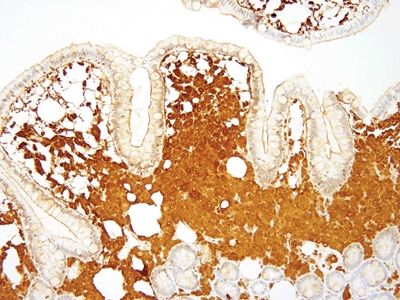
Figure 3.6.3 Whipple disease, Whipple bacillus immunolabeling. The immunostain also shows a coarse granular appearance.
Figure 3.6.4 Mycobacterium avium complex enteritis. The appearance overlaps that of Whipple disease, but the quality of the macrophages in the lamina propria is different, even on the H&E stain. In addition, there are no cystic spaces.
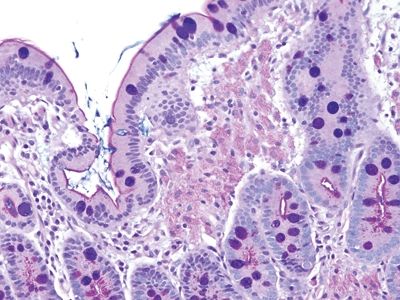
Figure 3.6.5 Mycobacterium avium complex enteritis, PAS/AB. Note the delicate appearance of the PAS reactive mycobacteria in the macrophages compared to that of Whipple disease. In this case, a negative image imparted by the waxy coats of the organisms can be appreciated.
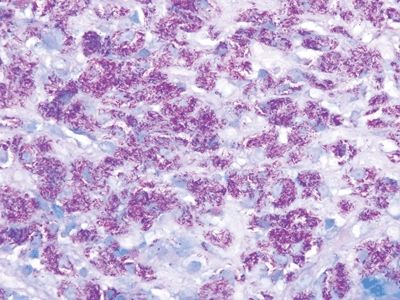
Figure 3.6.6 Mycobacterium avium complex enteritis, acid-fast stain. Lamina propria macrophages are bursting with acid-fast organisms.
3.7 Common variable immunodeficiency vs. Normal small intestine
| Common Variable Immunodeficiency | Normal Small Intestine | |
|---|---|---|
| Age/Gender | Bimodal distribution in children and then in middle-aged adults for first presentation. Probably, more than one disorder is represented | Not applicable |
| Location | Systemic manifestations but findings attributable to the small bowel are common | Images here are of duodenum since it is most commonly biopsied |
| Symptoms | Diarrhea, abdominal pain. Sometimes, the diarrhea is attributable to infections, classically giardiasis | Not applicable |
| Signs | Nothing specific. Patients have manifestations of hypogammaglobulinemia or agammaglobulinemia including multiple infections. At endoscopy, prominent small lymphoid nodules are common | Not applicable |
| Etiology | Probably, many mechanisms, but patients have reduced immunoglobulin. Frequently, the immunoglobulins are autoantibodies, and patients may have multiple immune alterations | Not applicable |
| Histology | ||
| 1. The key finding is loss of lamina propria plasma cells, which is seen in about 70% of patients. In the other 30% of patients, the diagnosis is impossible on histologic grounds in isolation (Figs. 3.7.1–3.7.3) 2. Some examples show celiac disease–like findings (Fig. 3.7.4) | 1. Normal duodenum shows a brush border, slender long villi, and short crypts. The lamina propria contains lymphocytes, plasma cells, eosinophils, and mast cells. There are intraepithelial lymphocytes in the crypts but fewer at the tips of the villi (Figs. 3.7.5–3.7.8) | |
| Special studies |
|
|
| Treatment | Management of the ensuing infections. Administration of immunoglobulin | Not applicable |
| Prognosis | Patients are at risk of infections and neoplasms. Lifelong management is required | Not applicable |
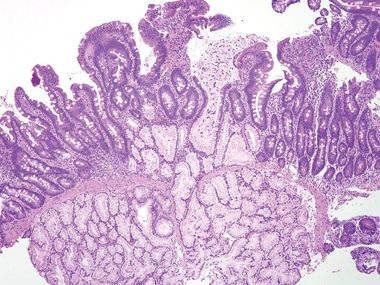
Figure 3.7.1 Common variable immunodeficiency. Diagnosis of this condition usually requires clinical correlation, but the majority of cases have a key finding—namely, the lack of plasma cells in the lamina propria, which will be overlooked if not specifically assessed. At low magnification, this case appears wholly nonspecific (although there is a crushed Brunner gland to enjoy).
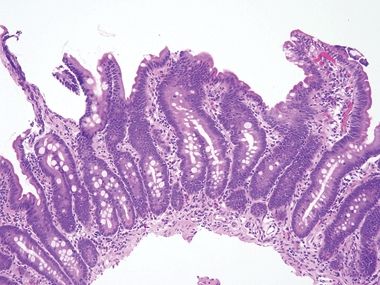
Figure 3.7.2 Common variable immunodeficiency. Slightly higher power than that seen in Figure 3.7.1 offers a hint—the lamina propria seems to have rescued numbers of cells. Normally, it is chock-full of plasma cells.
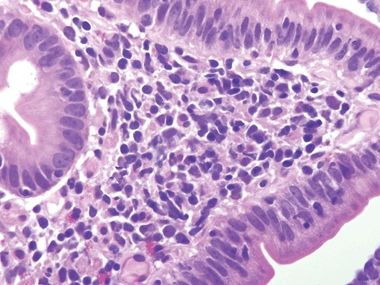
Figure 3.7.3 Common variable immunodeficiency. At high magnification, plasma cells are absent from the lamina propria, a finding that supports the diagnosis of common variable immunodeficiency, but one that is present in about two-thirds of cases rather than in all cases.
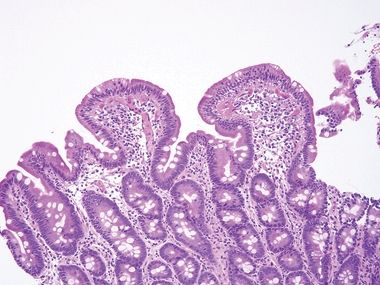
Figure 3.7.4 Common variable immunodeficiency. There is prominent surface lymphocytosis in this example, a feature that can lead to an incorrect interpretation of celiac disease. Unfortunately, small bowel biopsies from a host of conditions display overlapping features such that correlation with clinical and laboratory features is always key to diagnosis.
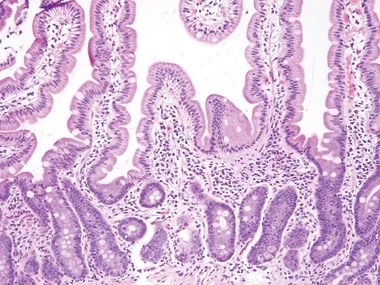
Figure 3.7.5 Normal small intestinal mucosa. Tall slender villi are present. Even at this low magnification, plasma cells are apparent, populating the lamina propria along with lymphocytes, eosinophils, and mast cells. Neutrophils should be absent.
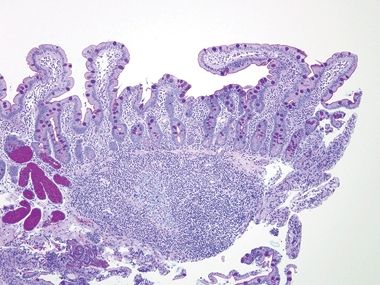
Figure 3.7.6 Normal small intestinal mucosa, PAS/AB stain. This image offers a good view of the anticipated density of lymphoplasmacytic cells in the small bowel lamina propria. Note the lymphoid aggregate.
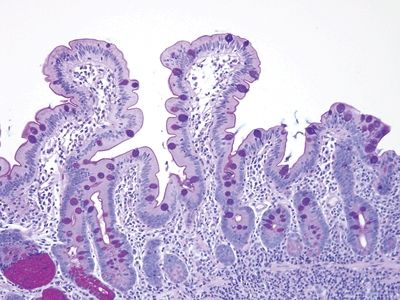
Figure 3.7.7 Normal small intestinal mucosa, PAS/AB stain. Note the slender brush border, goblet cells, and density of lamina propria contents.
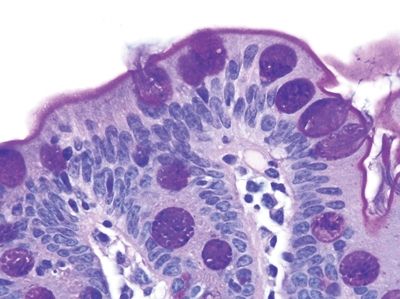
Figure 3.7.8 Normal small intestinal mucosa, PAS/AB stain. Minimal lamina propria is present, but it shows plasma cells.
3.8 Autoimmune enteropathy vs. Normal small bowel
| Autoimmune Enteropathy | Normal Small Intestine | |
|---|---|---|
| Age/gender | The term “autoimmune enteropathy” can be used to encompass several conditions. So-called immune dysregulation, polyendocrinopathy, enteropathy, X-linked (IPEX) syndrome is included and manifests in boys, whereas common variable immunodeficiency can be also associated with autoantibodies against enterocytes. All forms are associated with immune dysregulation | Not applicable |
| Location | Affects the small bowel and colon, primarily the small bowel | Images here are of the duodenum since it is most commonly biopsied |
| Symptoms | Diarrhea, wasting | Not applicable |
| Signs | Hypoalbuminemia | Not applicable |
| Etiology | Variable but some examples in individuals with FOXP3 mutations (this molecule governs regulatory T cells (Tregs)) | Not applicable |
| Histology | ||
| 1. Small bowel samples may appear celiac disease–like with prominent intraepithelial lymphocytosis and can be indistinguishable. The key in some cases is to recognize the absence of goblet cells and/or Paneth cells and prominent crypt apoptosis (Figs. 3.8.1–3.8.3) 2. Some patients taking olmesartan (Benicar) or similar angiotensin II receptor blockers show celiac-like findings as well, with prominent apoptosis (Fig. 3.8.4) | 1. Normal duodenum shows a brush border, slender long villi, and short crypts. The lamina propria contains lymphocytes, plasma cells, eosinophils, and mast cells. There are intraepithelial lymphocytes in the crypts but fewer at the tips of the villi. Lastly, nearly every crypt has several Paneth cells (Figs. 3.8.5–3.8.8). In fact, these biopsies were taken from the same patient as that seen in Figures 3.8.1–3.8.3 after intensive treatment | |
| Special studies |
|
|
| Treatment | Steroids | Not applicable |
| Prognosis | Reflective of the etiology for the autoimmunity. Patients can be managed but are often at risk for life-threatening infections based on associated conditions | Not applicable |
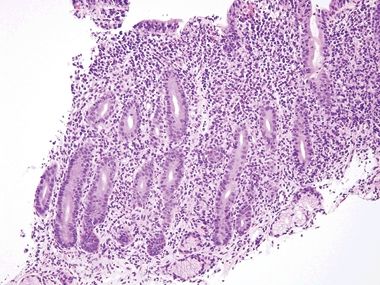
Figure 3.8.1 Autoimmune enteropathy. This biopsy shows a packed lamina propria, but what is missing? There is not a single goblet cell or Paneth cell, a clue to autoimmune enteropathy. Sometimes, severe mucosal injury results in foci lacking these constituents as well, so a clinicopathologic dialogue is always indicated when evaluating such patients, who are typically quite ill.
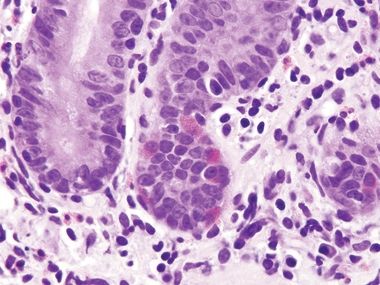
Figure 3.8.2 Autoimmune enteropathy. Lots of endocrine cells are present (their delicate cytoplasmic granules are oriented away from the lumen to reach the capillaries) but no Paneth cells, which have coarser granules than endocrine cells. Normally, the majority of small bowel crypts show Paneth cells at their bases.
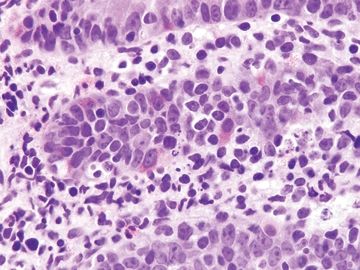
Figure 3.8.3 Autoimmune enteropathy. This example shows striking crypt apoptosis as well as absence of goblet cells and Paneth cells.
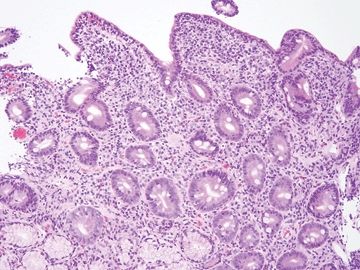
Figure 3.8.4 Enteropathy associated with olmesartan (Benicar, a blood pressure medication) can display features difficult to separate from celiac disease, collagenous sprue, and autoimmune diseases. This example shows prominent surface lymphocytosis akin to celiac disease and also featured prominent crypt apoptosis.
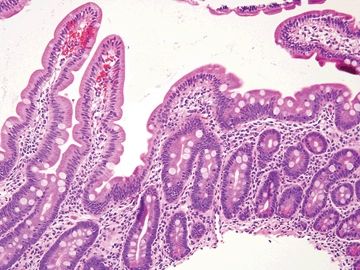
Figure 3.8.5 Normal small bowel. The brush border, long villi, lamina propria plasma cells, goblet cells, and Paneth cells are all present and normal.
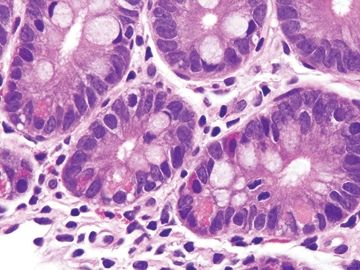
Figure 3.8.6 Normal small bowel. The crypt at the right crypt shows a few Paneth cells in the lower part with large luminally facing granules and endocrine cells in the upper part f the crypt with smaller, redder granules that point towards the lamina propria.
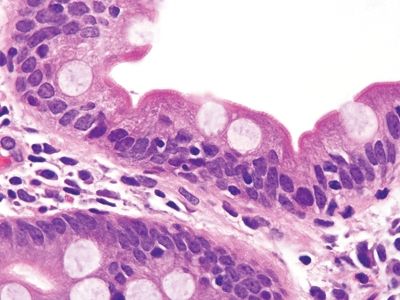
Figure 3.8.7 Normal small bowel. This field is from the base of a villus and shows a few intraepithelial lymphocytes, a normal finding. There should be fewer lymphocytes in the tips of the villi.
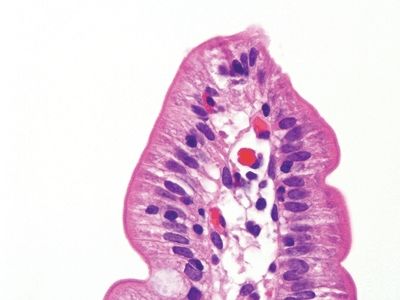
Figure 3.8.8 Normal small bowel. This villous tip only shows a few intraepithelial lymphocytes and certainly fewer than the base. In fact, all these images are from the same patient as that seen in Figures 3.8.1–3.8.3 after an intensive course of immunomodulatory therapy. A success!
3.9 Autoimmune enteropathy vs. Common variable immunodeficiency
| Autoimmune Enteropathy | Common Variable Immunodeficiency | |
|---|---|---|
| Age/gender | The term “autoimmune enteropathy” can be used to encompass several conditions. So-called immune dysregulation, polyendocrinopathy, enteropathy, X-linked (IPEX) syndrome is included and manifests in boys. All forms are associated with immune dysregulation. Patients with common variable immunodeficiency can have autoimmune enteropathy as a component so the two overlap, but the key feature of autoimmune enteropathy is immune damage to enterocytes and Paneth cells | Bimodal distribution in children and then in middle-aged adults for first presentation. Probably, more than one disorder is represented |
| Location | Affects the small bowel and colon, primarily the small bowel | Systemic manifestations but findings attributable to the small bowel are common |
| Symptoms | Diarrhea, wasting | Diarrhea, abdominal pain. Sometimes the diarrhea is attributable to infections, classically giardiasis |
| Signs | Hypoalbuminemia | Nothing specific. Patients have manifestations of hypogammaglobulinemia or agammaglobulinemia including multiple infections. At endoscopy, prominent small lymphoid nodules are common |
| Etiology | Variable but some examples in individuals with FOXP3 mutations (this molecule governs regulatory T cells) | Probably, many mechanisms, but patients have reduced immunoglobulin. Frequently, the immunoglobulins are autoantibodies, and patients may have multiple immune alterations |
| Histology | 1. Small bowel samples may appear celiac disease–like with prominent intraepithelial lymphocytosis and can be indistinguishable. The key in some cases is to recognize the absence of goblet cells and/or Paneth cells and prominent crypt apoptosis (Figs. 3.9.1–3.9.3) | 1. The key finding is loss of lamina propria plasma cells, which is seen in about 70% of patients. In the other 30% of patients, the diagnosis is impossible on histologic grounds in isolation (Figs. 3.9.4–3.9.6) |
| Special studies |
|
|
| Treatment | Steroids | Management of the ensuing infections. Administration of immunoglobulin |
| Prognosis | Reflective of the etiology for the autoimmunity. Patients can be managed but are often at risk for life-threatening infections based on associated conditions | Patients are at risk of infections and neoplasms. Some patients acquire an autoimmune enteropathy component so the conditions are overlapping. Lifelong management is required |
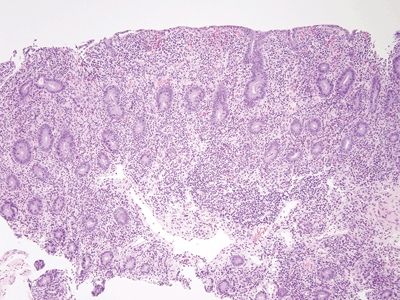
Figure 3.9.1 Autoimmune enteropathy. This process appears very similar to celiac disease, but if one pays attention to checking each compartment in a small bowel biopsy, it becomes apparent that this biopsy lacks goblet cells and Paneth cells.
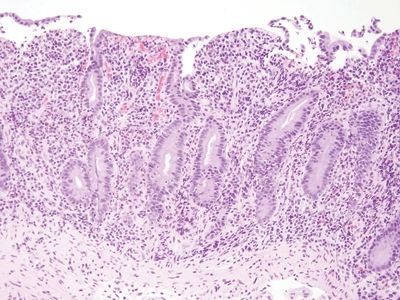
Figure 3.9.2 Autoimmune enteropathy. Higher magnification of the case seen in Figure 3.9.1. There is intraepithelial lymphocytosis in addition to loss of goblet cells and Paneth cells.
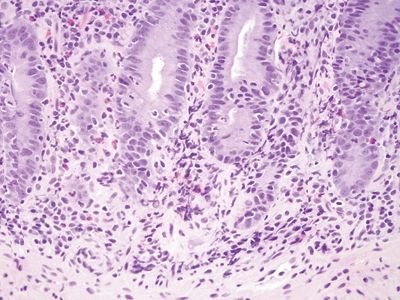
Figure 3.9.3 Autoimmune enteropathy. This case also shows some acute inflammation, so correlation with studies for organisms is also important.
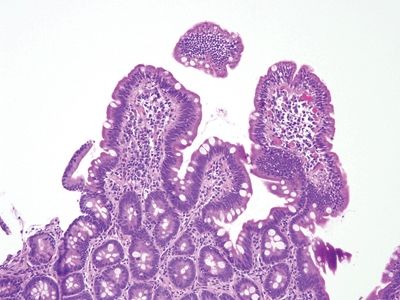
Figure 3.9.4 Common variable immunodeficiency. This biopsy appears unremarkable other than a bit of intraepithelial lymphocytosis in the tips of the villi, which should raise the possibility of “latent” celiac disease. However, the lamina propria appears slightly empty.
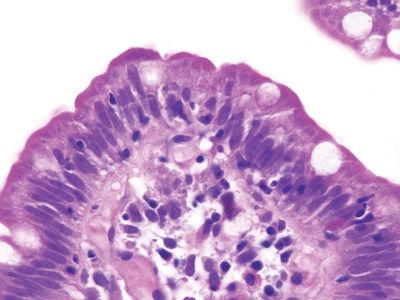
Figure 3.9.5 Common variable immunodeficiency. This intraepithelial lymphocytosis should prompt a concern for celiac disease but look in the lamina propria. Plasma cells are absent.
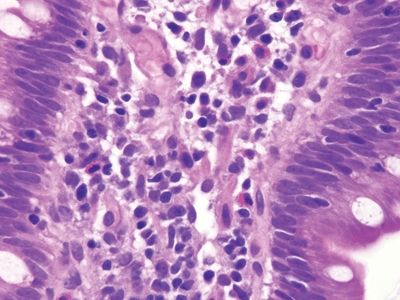
Figure 3.9.6 Common variable immunodeficiency. Plasma cells are absent. Unfortunately, some cases have plasma cells. In such cases, antibody testing for celiac disease is typically negative, and the patients are prone to many infections, clues that might prompt testing.
3.10 Celiac disease vs. Small bowel bacterial overgrowth of enteric contents
| Celiac Disease | Small Bowel Bacterial Overgrowth from Stasis of Enteric Contents | |
|---|---|---|
| Age/Gender | Presents at any age and race but classically in white children with female predilection (1.5–2X) and associated with certain HLA types (HLA-DQ2 and HLA-DQ8). More common in developing countries than in the past with introduction of wheat into the diet. Once vanishingly rare in Southeast Asia, now emerging there | Adults |
| Location | Affects entire body, typically assessed on duodenal biopsies | Anywhere in the small bowel |
| Symptoms | Chronic diarrhea, weight loss, abdominal distension | Abdominal pain, flatulence, bloating, loose stools |
| Signs | Abdominal distension, aphthous stomatitis, short stature, iron deficiency, dermatitis herpetiformis, reduced bone density | Defined as the presence of bacteria in excess of 105 colony forming units per millimeter on culture of upper intestinal (duodenal) aspirates. Alternate noninvasive test include lactulose and glucose hydrogen breath tests |
| Etiology | Systemic immune-mediated response to dietary gluten in genetically susceptible hosts. Diagnosis is confirmed by serologic testing. IgA anti–tissue transglutaminase detection is the preferred initial screen. Cannot be used in persons with IgA deficiency | Classically attributed to changes in small bowel flora dues to stasis, usually in patients with obstruction from strictures and adhesions but more recently found in patients lacking anatomic abnormalities. There is controversial overlap with irritable bowel syndrome, especially since office breath testing can be lucrative |
| Histology | ||
| 1. In classic case, villous blunting, crypt hyperplasia, prominent intraepithelial lymphocytosis, and expansion of lamina propria, usually not prominent acute inflammation (Figs. 3.10.1–3.10.3) 2. Some examples show preserved villous architecture but intraepithelial lymphocytosis concentrated in the tips of the villi (Fig. 3.10.4) | 1. Can be essentially identical to celiac disease with villous blunting, intraepithelial lymphocytosis, and crypt hyperplasia. Neutrophils can be a component, but there are no histologic features that allow perfect distinction (Figs. 3.10.5–3.10.7) 2. Intraepithelial lymphocytosis without villous blunting can be present, as per celiac disease (Fig. 3.10.8) 3. Essentially the histologic features in such cases must be correlated with laboratory findings | |
| Special studies |
|
|
| Treatment | Lifelong gluten-free diet | Antibiotics, ideally based on culture sensitivities |
| Prognosis | Overall excellent with adherence to diet. Patients at slightly increased risk for lymphomas and small bowel adenocarcinoma but these risks are still small. Life expectancy normal to slightly shortened | Depends on the etiology of the enteric stasis |
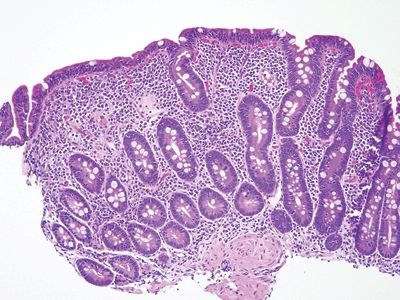
Figure 3.10.1 Celiac disease. This classic lesion shows complete villous atrophy and striking surface lymphocytosis. The lamina propria is expanded with plasma cell–rich contents.
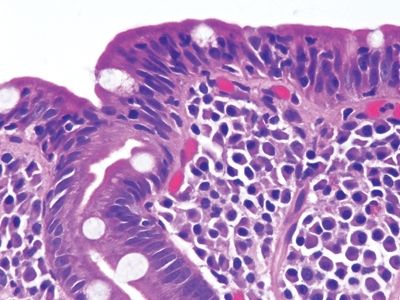
Figure 3.10.2 Celiac disease. Note the lamina plasmacytosis and surface lymphocytosis.
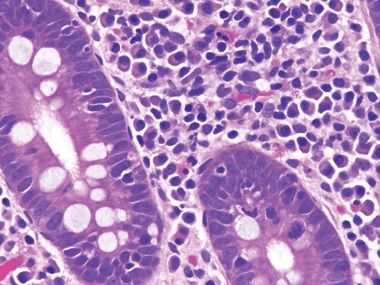
Figure 3.10.3 Celiac disease. The crypts also display lymphocytosis.
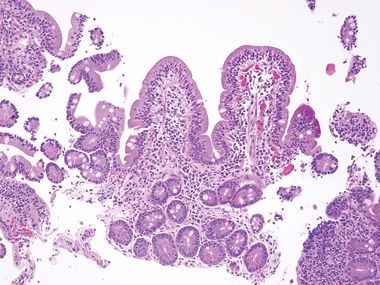
Figure 3.10.4 Celiac disease. This proven example shows incomplete villous atrophy but prominent intraepithelial lymphocytosis, which would be regarded as “Marsh 2 pattern” in one of the classification schemes. However, with this pattern of injury, a diagnosis of celiac disease can be confirmed only about 20% of the time. As such, “Marsh grading” is meaningless unless the pathologist already knows that the patient has celiac disease.
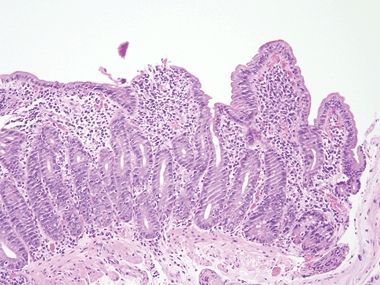
Figure 3.10.5 Small bowel bacterial overgrowth (often attributed to stasis of enteric contents). Unfortunately, the findings are nonspecific, but this example appears very “busy” at lower power. Goblet cells are reduced, presumably as a result of rapid mucosal regeneration, but Paneth cells are present.
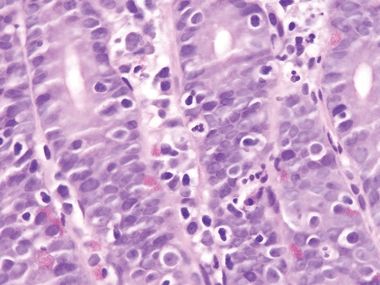
Figure 3.10.6 Small-bowel bacterial overgrowth (often attributed to stasis of enteric contents). Note the neutrophils in the epithelium.
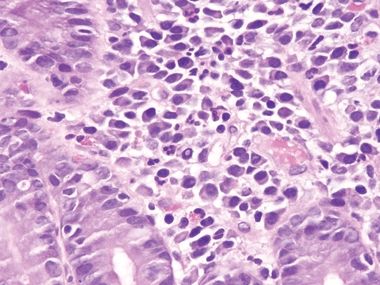
Figure 3.10.7 Small bowel bacterial overgrowth (often attributed to stasis of enteric contents). This field shows prominent lamina propria neutrophils. Plasma cells and damaged goblet cells are also seen.
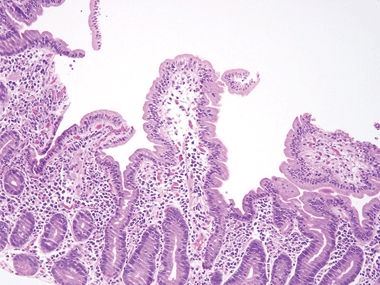
Figure 3.10.8 Small bowel bacterial overgrowth (often attributed to stasis of enteric contents). This example is difficult to separate from latent celiac disease in the absence of correlation with laboratory testing and clinical history.
3.11 Celiac disease vs. Common variable immunodeficiency
| Celiac Disease | Common Variable Immunodeficiency | |
|---|---|---|
| Age/Gender | Presents at any age and race but classically in white children with female predilection (1.5–2X) and associated with certain HLA types (HLA-DQ2 and HLA-DQ8). More common in developing countries than in the past with introduction of wheat into the diet. Once vanishingly rare in Southeast Asia, now emerging there, presumably as a result of dietary changes from rice to wheat | Bimodal distribution in children and then in middle-aged adults for first presentation. Probably, more than one disorder is represented |
| Location | Affects the entire body, typically assessed on duodenal biopsies | Systemic manifestations but findings attributable to the small bowel are common |
| Symptoms | Protean but classic cases with chronic diarrhea, weight loss, and abdominal distension | Diarrhea, abdominal pain. Sometimes the diarrhea is attributable to infections, classically giardiasis |
| Signs | Abdominal distension, aphthous stomatitis, short stature, iron deficiency, dermatitis herpetiformis, reduced bone density | Nothing specific. Patients have manifestations of hypogammaglobulinemia or agammaglobulinemia including multiple infections. At endoscopy, prominent small lymphoid nodules are common |
| Etiology | Systemic immune-mediated response to dietary gluten in genetically susceptible hosts. Diagnosis is confirmed by serologic testing. IgA anti–tissue transglutaminase detection is the preferred initial screen. Cannot be used in persons with IgA deficiency | Probably, many mechanisms, but patients have reduced immunoglobulin. Frequently, the immunoglobulins are autoantibodies, and patients may have multiple immune alterations |
| Histology | ||
| 1. In classic case, villous blunting, crypt hyperplasia, prominent intraepithelial lymphocytosis, and expansion of lamina propria, usually not prominent acute inflammation (Figs. 3.11.1–3.11.3) 2. There are ample plasma cells in the lamina propria (Fig. 3.11.3) | 1. The key finding is loss of lamina propria plasma cells, which is seen in about 70% of patients. In the other 30% of patients, the diagnosis is impossible on histologic grounds in isolation but the presence of infectious agents such as Giardia or cytomegalovirus (Figs. 3.11.4–3.11.6) | |
| Special studies |
|
|
| Treatment | Lifelong gluten-free diet | Management of the ensuing infections. Administration of immunoglobulin |
| Prognosis | Overall excellent with adherence to diet. Patients at slightly increased risk for lymphomas and small bowel adenocarcinoma but these risks are still small. Life expectancy normal to slightly shortened | Patients are at risk of infections and neoplasms. Some patients acquire an autoimmune enteropathy component so the conditions are overlapping. Lifelong management is required |
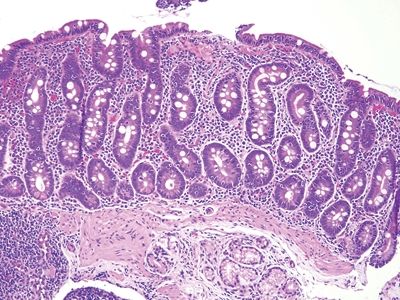
Figure 3.11.1 Celiac disease. This classic example shows striking intraepithelial lymphocytosis and a “stuffed” lamina propria with numerous plasma cells. Goblet and Paneth cells are plentiful.
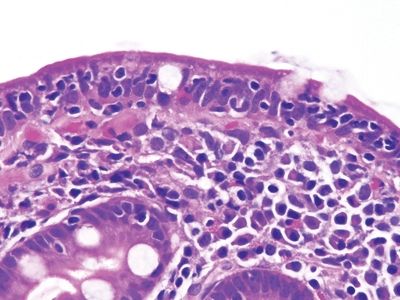
Figure 3.11.2 Celiac disease. Numerous lymphocytes are present in the surface epithelium, and there are many lamina propria plasma cells.
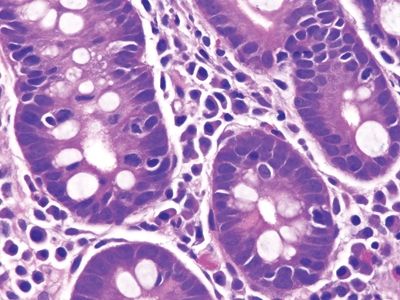
Figure 3.11.3 Celiac disease. Intraepithelial lymphocytes are present in the crypts. Plasma cells are the primary lamina propria constituent.
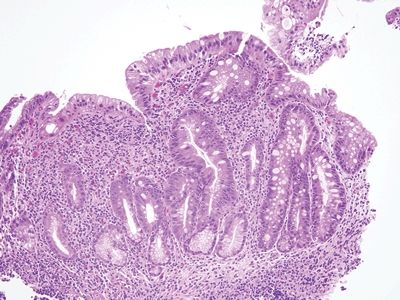
Figure 3.11.4 Common variable immunodeficiency. The lamina propria is expanded but, in this case, not by plasma cells. Patients with common variable immunodeficiency are prone to infections, and this appearance may reflect this.
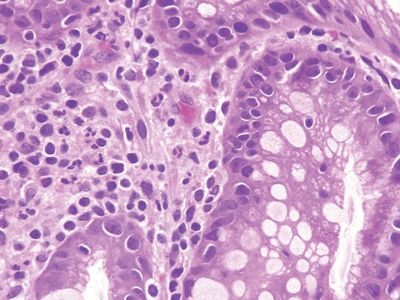
Figure 3.11.5 Common variable immunodeficiency. At high magnification in this case, plasma cells are absent from the lamina propria but neutrophils are present, probably a reflection of infection.
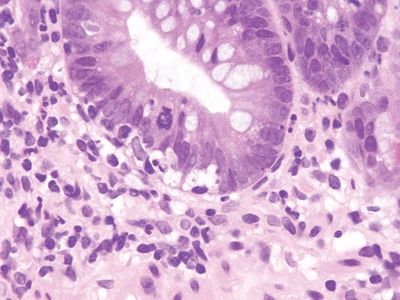
Figure 3.11.6 Common variable immunodeficiency. This example shows prominent crypt apoptosis. Note the lack of lamina propria plasma cells.
3.12 Collagenous sprue/enteritis vs. Radiation change
| Collagenous Sprue | Radiation Injury to Small Bowel | |
|---|---|---|
| Age/Gender | Late 50s to older with female predominance | Usually adults; male predominance secondary to overall male predominance in cancer |
| Location | Typically in duodenum but similar collagen deposition can be found in the stomach (collagenous gastritis) and colon (collagenous colitis) in the same patients. Similar findings can also complicate the use of olmesartan for hypertension | Anywhere in the small bowel near radiation sites but typically detected in duodenal samples in patients with pancreatobiliary or gastric cancers |
| Symptoms | Prominent chronic diarrhea and weight loss | Typically those of obstruction—abdominal pain, vomiting, and constipation |
| Signs | At endoscopy, similar appearances to those of celiac disease with evidence of atrophy. Patients may have laboratory profiles that include hypoalbuminemia. Celiac disease serology is negative or is initially positive, and then, the patient stops responding to gluten withdrawal, and celiac serology is negative. HLA-DQ2/HLA-DQ8 panels negative in many cases as well | Abdominal tenderness on palpation. Atrophic appearance or ulcers on endoscopy |
| Etiology | Some cases are associated with celiac disease that becomes refractory to gluten, and some are idiopathic. Medications can trigger this pattern of injury (e.g., olmesartan, a blood pressure medication) | Tissue damage from therapeutic radiation |
| Histology | ||
| 1. The key finding is increased subepithelial collagen that encircles capillaries and extends into the lamina propria. The quality rather than the quantity is key, so measuring the collagen is not necessary. Intraepithelial lymphocytosis should be present, and some examples feature prominent crypt apoptosis (Figs. 3.12.1–3.12.4) | 1. Sclerosis of lamina propria (can be transmural but on mucosal biopsies the lamina propria is the damaged component that is seen) generally without accompanying lymphocytosis. Vascular ectasia and vascular wall sclerosis can be present. However, the stasis from transmural fibrosis can result in some lymphocytosis (Figs. 3.12.5 and 3.12.6) 2. Differs from amyloidosis by appearing more eosinophilic collagen. An example of amyloidosis is shown, which also tends to affect the vessels but with a different appearance on H&E (Fig. 3.12.7) 3. Occasionally, the pathologist is fortunate enough to encounter selective internal radiation microspheres (SIRS) used to administer yttrium therapy, usually to liver metastases (Fig. 3.12.8) | |
| Special studies |
|
|
| Treatment | Steroids are the first line of therapy, and some patients have complete recovery, whereas others require additional immunosuppression. A small subset progresses to T-cell lymphomas | Generally none but some patients may require resections to relieve obstruction |
| Prognosis | Overall favorable following immune modulation but some patients have persistent nutritional impairment or progression to lymphoma | That of the cancer that was treated by radiation |
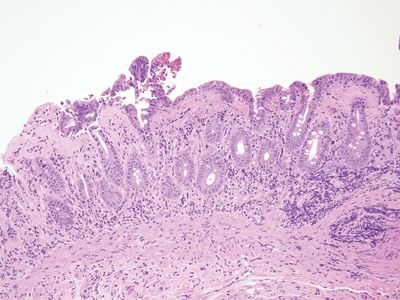
Figure 3.12.1 Collagenous sprue/collagenous enteritis. The subepithelial collagen is thickened and encircles fibroblasts, inflammatory cells, and capillaries. There is mild intraepithelial lymphocytosis. The deep lamina propria is expanded.
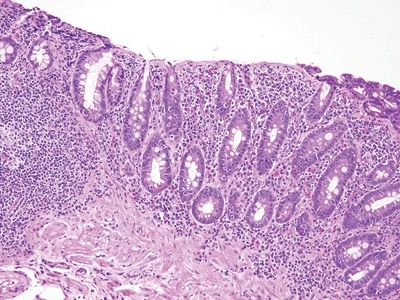
Figure 3.12.2 Collagenous sprue/collagenous enteritis. In this example, the friable surface epithelium has “chipped off,” but intraepithelial lymphocytosis is apparent. Note the lamina propria plasma cells, lymphocytes, and eosinophils.









