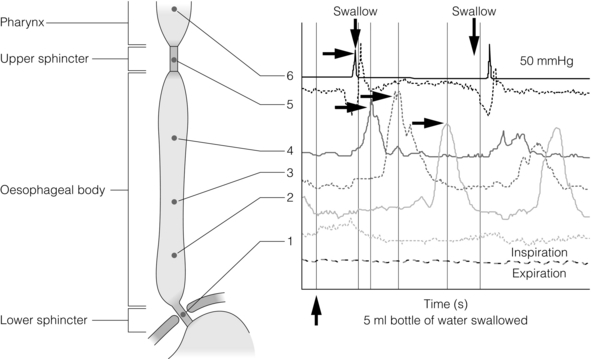
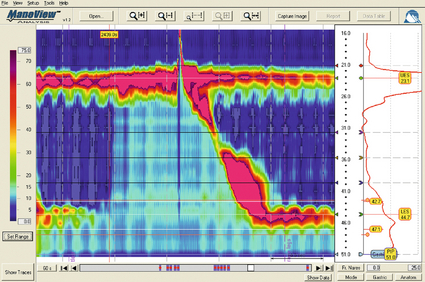
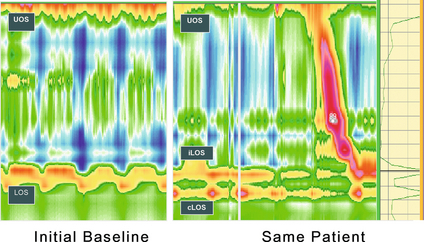
12 Gastro-oesophageal reflux disease (GORD) describes symptoms or mucosal damage caused by reflux of gastric contents into the oesophagus.1 Symptoms are variable and along with damage are associated with many permutations of motility, endoscopic and physiological abnormalities. GORD is the major factor leading to the increasing incidence of oesophageal adenocarcinoma,2 and is one of the commonest health problems in the developed world. Gastro-oesophageal reflux is universal, and as such can be viewed as a normal physiological process. Such physiological episodes are asymptomatic and rapidly cleared. They occur mainly after meals, in the upright position and when awake.3,4 By contrast, pathological reflux results in chronic symptoms or mucosal damage. However, the subjectivity of symptoms is such that the boundary between the two is blurred, with many people viewing occasional reflux symptoms as normal, without seeking medical attention. For example, the reflux of air (belching) due to gastric distension is a universal experience, yet depending upon frequency and patient perception may be either normal or or a symptom of GORD. Unsurprisingly, the epidemiology of GORD is difficult to determine. A 2011 population-based cohort study of 45 000 people from Norway5 found the prevalence of any symptoms to be 41%, weekly symptoms to be 17% and severe symptoms to be 6.7% The figures had increased by 30%, 24% and 47%, respectively, over the previous decade. Incidence of any symptoms was 3%, with that of severe symptoms 0.2%, with spontaneous resolution of these symptoms occurring in 2% and 1%, respectively. A systematic review of 15 studies estimated a similar prevalence of weekly symptoms in 10–20% in the Western world (5% in Asia).6 These findings were similar to a smaller Finnish study of 1562 consecutive patients referred for endoscopy. With the caveat of this selection bias, the overall incidence was estimated as 3%, of which 2% had endoscopic mucosal damage.7 Two additional studies have reached similar conclusions, with 32–38% of those with symptoms having normal endoscopies.8,9 Importantly, the converse may be true, with up to 20% of those with oesophagitis or Barrett’s being asymptomatic.10 Traditionally, GORD has been viewed as a spectrum of a single disease, with endoscopy-negative symptoms at the mild end, increasing grades of oesophagitis representing progressively severe disease, culminating in Barrett’s metaplasia.1 However, this approach is undermined by a number of clinical, endoscopic and physiological findings, suggesting that the three may in fact be distinct groups and disease processes in themselves (Box 12.1).11,12 Most importantly, endoscopic progression from one end of the spectrum to the other is rare.12 Symptoms typically vary between the three GORD groups discussed above. Those with symptomatic endoscopy-negative disease (or non-erosive reflux) tend to have severe, often atypical symptoms with variable response to acid suppression (presumably representing oesophageal hypersensitivity to acid and non-acid reflux).12 By contrast, those with erosive reflux and oesophagitis tend towards more typical symptoms responding well to acid suppression.12 Finally, those with Barrett’s often have minimal symptoms, possibly due to the relative insensitivity of metaplastic epithelium to acid.13 On swallowing the upper and lower oesophageal sphincters relax, and the food or liquid bolus is propelled distally by peristalsis to the stomach. The responsible mechanisms involve complicated coordination between central and local neuromuscular mechanisms. Peristalsis is the sequential contraction of the oesophageal body. Animal models demonstrate progression aborally in both longitudinal and circular layers, without torque.14 Primary peristalsis is instigated centrally in the swallowing centre by swallowing, with the wave arising in the pharynx. A persistent bolus distends the oesophagus, triggering local neural mechanisms and a secondary peristaltic wave.15 Tertiary contractions are aberrant, synchronous contractions of oesophageal segments, which play no role in peristalsis. During peristalsis, the oesophageal body contracts segmentally, with the greatest pressure at the mid-point of the contracted segment, a few centimetres behind the bolus16 (Fig. 12.1). Figure 12.1 A standard six-sensor manometry trace of normal oesophageal peristalsis. Adapted from Anggiansah A, Marshal R. Use of the oesophageal laboratory, 1st edn. Oxford: Isis Medical Media, 2000. With permission from Isis Medical Media. Whether the longitudinal and circular layers contract synchronously or not is disputed. A number of studies have suggested that the circular layer initially hyperpolarises, to contract two seconds after the longitudinal layer.17–19 However, this has been disputed, and it has been suggested that any delay is a function of the manometric techniques used.20 In any case, the involvement of both layers confers mechanical advantages, with longitudinal contractions shortening the circular layer, allowing greater contractile force. In addition, contraction of both reduces wall tension at the site of contraction.21 Primary peristalsis is initiated centrally (via the vagus) and modified peripherally (via local myogenic and neuronal mechanisms). Vagal efferents from the nucleus ambiguous initiate skeletal muscle contraction, and those from the dorsomotor nucleus innervate smooth muscle. These do so via intrinsic neurons of the myenteric plexus (between the muscle layers). Modification within the oesophageal body occurs in response to volume, temperature and acid receptors, with central feedback via vagal afferents. Warm boluses initiate an exaggerated peristaltic wave, whereas cold boluses (e.g. ice cream) may not provoke distal peristalsis.22 This is true for wet and dry swallows, respectively. In addition, oesophageal acid receptors are believed to allow protective clearance of refluxate. Crucial to successful peristalsis is the oesophageal latency period. Following initial stimulation of the circular muscle layers, a variable period of membrane hyperpolarisation occurs, primarily mediated by intrinsic nitric oxide inhibition.23 This period increases progressively, moving distally along the oesophageal body, with greater inhibitory inhibition.24 Initial (or deglutitive) inhibition describes a refractory period of the oesophageal body due to myogenic and inhibitory properties. Swallowing for a second time within seconds causes the first peristaltic wave to be aborted.25 This allows rapid sequences of swallows (used primarily for drinking). A positive pressure gradient of approximately 10 mmHg exists between the stomach and oesophagus. The stomach and abdominal oesophagus lie within 5 mmHg of positive intra-abdominal pressure, with the thoracic oesophagus exposed to 5 mmHg of negative pressure. That gastro-oesophageal reflux is the exception rather than the rule is due to several factors: the lower oesophageal sphincter (LOS), the diaphragmatic sphincter or ‘pinch-cock’, distal oesophageal compression, and other mechanical barriers such as the cardio-oesophageal angle (of His) and the mucosal rosette (Box 12.2). The LOS is the primary antireflux mechanism. Although not an anatomically discrete sphincter, it is a resting high-pressure zone in the distal oesophagus, which relaxes appropriately to allow swallowing, belching and vomiting.26 It is composed of specialised smooth muscle, arranged in either clasp or sling formation, running in the distal 1–4 cm of the oesophagus and blending with the cardia.27 The basal tone of the LOS is both intrinisic (inherent muscular properties) and extrinsic (excitatory cholinergic vagal input). Clasp and sling fibres vary in resting tone and responsiveness to stimulation; sling fibres have lower tone but greater responsiveness, probably due to differing proportions of contractile proteins. These fibres are also asymmetrically arranged, potentially accounting for the longitudinal and radial asymmetry of basal pressure (being greatest posteriorly and on the right).28 LOS muscle cells are tonic, and as such distinct from the phasic cells of the oesophageal body (which have no intrinsic tone). Neurological control of the LOS is complicated. Central control is mediated by vagal efferents originating in the dorsal motor nucleus (with excitatory cell bodies cephalad and inhibitory cells caudally).29 Sensory feedback from the LOS is relayed via the tractus solitarius. Central neurotransmitters include glutamate, adrenaline, dopamine, acetylcholine and nitric oxide. Peripherally, vagal fibres synapse in the myenteric plexus via acetylcholine.30 Both atropine and vagotomy reduce resting LOS pressure significantly.31 Nitric oxide is the primary inhibitory neurotransmitter of the LOS: in murine models nitric oxide knockout increases resting LOS pressure and prevents the transient LOS relaxations (TLOSRs) required for swallowing32 (Fig. 12.2). This inhibition may be further modified by the interstitial cells of Cajal.33 Figure 12.2 High-resolution manometry demonstrating transient lower oesophageal sphincter relaxation (TLOSR). The upper oesophageal sphincter is demonstrated by the upper red and yellow zone, and the LOS by the lower red and yellow zone. On relaxation of the lower oesophageal sphincter a common cavity is created between the stomach and oesophagus – demonstrated by the light blue zone on the spatiotemporal plot (centre). These events are also observed on the axial pressure plot (right). The event is terminated and oesophagus cleared by primary peristalsis with intra-oesophageal pressure returning to baseline levels. Images acquired by 36-channel SSI Manoscan 360. Reproduced from Fox MR, Bredenoord AJ. Oesophageal high-resolution manometry: moving from research into clinical practice. Gut 2008; 57(3):405–23. With permission from BMJ Publishing Group Ltd. LOS resting pressure varies physiologically, in particular reducing with meals.34 It also reduces in response to pharyngeal tactile stimulation (in the absence of swallowing or peristalsis).35 In control subjects, transient increases in intra-abdominal pressure (e.g. induced by straight leg raising, coughing and straining) are met with reflex increase in LOS pressure that is both greater and faster than the increase in gastric pressure.36 This response is due in part to neuromuscular reflexes (vagally mediated) and transmitted pressure from the crus of the diaphragm. Similarly, protective increases in pressure occur in response to drops in intrathoracic pressure.37 TLOSRs are appropriate short-lived (usually less than 10 seconds) relaxations instigated by primary or secondary peristalsis, gastric distension and vomiting.38,39 In asymptomatic controls, TLOSRs account for 94% of reflux episodes,40 primarily occurring after meals and when upright rather than supine34,41 (correlating with the distribution of asymptomatic physiological reflux episodes discussed above). Fifty per cent of TLOSRs result in reflux episodes.40,42 Their genesis is incompletely understood, but seems to be a vagally mediated response to proximal gastric distension sensed by mechanoreceptors.41,43 Fundoplication reduces the frequency of TLOSRs, potentially via reducing cardiac distensibility.44,45 Manometrically, TLOSRs are similar to the belch reflex, and many reflux episodes follow belching,46,47 suggesting that the former are aberrations of the latter. Pathological and physiological (swallow-induced) TLOSRs appear to be distinct, with pathological lasting longer (up to 45 seconds).48 In a small controlled study of patients with oesophagitis, pathological reflux was found to be due to three primary mechanisms: TLOSRs (65%), spontaneous reflux due to low resting LOS pressure (18%) and transient increased intra-abdominal pressure (17%).40 However, the commonest mechanisms vary between individuals. The slings of the right crus constitute a ‘pinch-cock’ mechanism. In the absence of a hiatus hernia, it is difficult to separate the relative contributions of the diaphragmatic sphincter and LOS. In those with a surgically resected LOS, however, a basal high-pressure zone is detectable (oscillating in line with ventilation), suggesting that the diaphragmatic sphincter has a tonic component.50 Such increases in high-pressure zone pressure with inspiration are abolished with curare in feline models, so as with the LOS the diaphragmatic sphincter seems to have both basal and reactive tonicity.51 These increases in pressure are related to depth of inspiration49 and are maximal during sleep (when the gastro-oesophageal pressure gradient is greatest).52 Functionally, in the absence of a hiatus hernia, both LOS and diaphragmatic sphincter contribute to the high-pressure zone.53 However, the mechanism of diaphragmatic sphincter relaxation differs neurochemically and functionally. Oesophageal distension (due to swallowing) triggers incomplete diaphragmatic sphincter relaxation, whereas TLOSRs cause complete relaxation.54 As with the LOS, this relaxation is both central and local. Central control is mediated via vagal afferents modifying control of the diaphragm,55 although this may be distal to the medulla in site.56 Local control may be due to stretching of diaphragmatic sphincter fibres by contraction of longitudinal oesophageal fibres.57 Variation in the distensibility of the diaphragmatic sphincter affects reflux; greater distensibility predisposes to more reflux episodes.58 The abdominal oesophagus is exposed to positive pressure within the abdomen. This has a compressive effect on the distal oesophagus/LOS, with a shorter length correlating with more frequent reflux.59 This is particularly important with increasing age and a recumbent position. The phreno-oesophageal ligament is a prolongation of abdominal fascia originating from the abdominal surface of the diaphragm, which anchors the oesophagus. As it approaches the oesophagus, it decussates into upper and lower leaves. The former inserts just above the squamocolumnar junction, with the latter inserting a few centimetres below, but is less well defined and often absent. The upper leaf inserts into the submucosa and intramuscular septae. This fibromuscular anchor is not absolute, allowing the oesophagus to slide 2 cm cranially when swallowing and, crucially, up to 4 cm during TLOSRs.60,61 Whether the oesophagus herniates into the thorax under physiological conditions is unlikely, however, as the diaphragmatic hiatus moves almost as far cranially. This arrangement serves to keep the distal oesophagus within this positive pressure environment, ensuring that any increases in intra-abdominal pressure are transmitted to the LOS.62 Anatomical variations in strength and height of insertion of the phreno-oesophageal ligament presumably influence the length of oesophagus to which this applies. Disruption of the ligament predisposes to sliding hiatus hernias, although the hernia sac will still envelop the LOS within the physiological (but not anatomical) abdomen. The acute angle of the gastro-oesophageal junction may represent a partial barrier to reflux, functioning as a ‘flap valve’ (the cardio-oesophageal angle of His). This angle disappears after death, suggesting that the angle is maintained by tonic contraction of the oblique sling fibres of the cardia.63 This ‘valve’ lies below the physiological high-pressure zone determined with radiological contrast, and so its contribution is therefore limited. The mucosal rosette of the gastro-oesophageal junction is also reported to act as a partial barrier. The main defence mechanisms comprise oesophageal clearance and tissue resistance. Reflex peristalsis is induced by oesophageal acid receptors and aided by gravity, both of which are impaired, particularly during sleep.64 The oesophagus has a number of further protective mechanisms, both intrinsic and extrinsic, termed tissue resistance. These are such that continuous in vivo exposure of oesophageal epithelium to hydrochloric acid and pepsin does not cause damage for 1 hour.65 Mechanisms are classed as pre-epithelial, epithelial and post-epithelial. Pre-epithelial resistance is conferred by the oesophageal buffer layer, augmented by saliva. In contrast to the gastroduodenal buffer layer, the (non-Barrett’s) oesophageal epithelium lacks a mucous gel barrier and the ability to secrete bicarbonate. However, the buffer needs only to raise pH above 3 to prevent pepsin-induced damage.66 This buffer is augmented by the neutralising properties of saliva. Swallowing saliva contributes to approximately 5% of acid clearance by neutralising acid remaining after peristaltic clearance.67 It has a pH of 7.02,68 but the solubility of its mucins means that it cannot contribute permanently to the oesophageal buffer.69 Consequently, when salivary production and primary oesophageal peristalsis cease at night, the oesophagus is particularly vulnerable.70 The epithelium itself possesses a combination of properties: a protective transmural electrochemical gradient, tight cell junctions, pH-dependent cation channels and intracellular buffers. Post-epithelial characteristics include adaptive perfusion and epithelial repair. For refluxed acid to cause epithelial cell death, hydrogen ions must enter the epithelial cytosol in sufficient quantities and for long enough to disrupt cytosol homeostasis and induce apoptosis/necrosis. One mechanism is transit of acid into the intercellular space, disrupting cell junctions and increasing permeability into the space, resulting in greater levels of acid. However, acid levels below that required to cause cellular damage may still be symptomatic. Nociceptors underlying heartburn can be found within the intercellular space within three cell layers of the oesophageal lumen,71 thus helping to explain the existence of heartburn without frank oesophagitis. Quite why pain and oesophagitis may be independent is not clear. Potentially this represents visceral hypersensitivity,72 or a reduction in acid sensitivity due to chronic reflux acid.73 The complexity of the body’s control of swallowing and reflux means that there are numerous opportunities for disruption (and therefore pharmacological and surgical intervention). Pathological reflux is caused by excessive frequency of TLOSRs, inadequate high-pressure zone resting pressure, and inability of the LOS and diaphragmatic sphincter to compensate for increases in abdominal pressure. A number of factors have been implicated in these mechanisms (Table 12.1). A small number of studies have demonstrated the role of a positive family history. A parental history of GORD conferred an odds ratio (OR) of 1.5 in one study,74 with an OR of 2.6 when symptoms are extended to that of an immediate relative.75 In the latter study, no association was found with GORD prevalence between spouses, implying genetic effects to be greater than shared environmental effects (although this has been disputed).76 Greater concordance has also been demonstrated between monozygotic than dizygotic twins.74 No association between sex and GORD has been demonstrated. Findings assessing age have been equivocal; both European and US studies assessing symptomatology have found contradictory results. Overall, a marginally increased risk with increasing age seems likely (OR 1.1)77 up to the age of 55, and possibly beyond.78 Longitudinal studies show that smoking increases the risk of GORD (OR 1.1–2.6)77,78 due to a chronic reduction in LOS pressure, combined with acute provocation of reflux (via coughing/deep inspiration). Particular foods and drinks are often felt to predispose to reflux episodes, although a Swedish case–control study of over 1000 subjects found no association between an extensive range of foodstuffs (including chocolate, coffee, onions and acidic fruits) and chronic symptoms of reflux (although the authors admit this may be due to avoidance in sufferers).79 The study also found no correlation between portion size and eating late in the evening (albeit with the same caveat). Coffee is consistently cited as a factor, although one not demonstrated by additional studies.74 Any underlying mechanism is believed to be due to caffeine-mediated inhibition of phosphodiesterase, inducing LOS relaxation.80 Alcohol is generally agreed to lead to reflux – the US study above78 found an OR of 1.8 between alcohol consumption and GORD. Again, this is thought to be via a reduction in LOS pressure, combined with direct irritation.80 Obesity has been repeatedy shown to correlate with GORD, with ORs between 1.378 and 2.8.77 A meta-analysis of nine studies found an OR of 1.4 for a body mass index (BMI) of 25–30, and 1.9 for a BMI greater than 30.81 Multiple mechanisms have been proposed: impaired LOS pressure, increased intra-abdominal pressure and delayed gastric emptying.81 A combination of GORD and obesity is a potent risk factor for oesophageal adenocarcinoma.82 The effect of weight loss on symptoms varies between studies, having been shown to improve symptoms83 or have no effect.84 Pregnancy induces progesterone-mediated LOS relaxation, in addition to increasing abdominal pressure. GORD has been associated with a number of gastrointestinal and extragastrointestinal conditions, including irritable bowel syndrome, peptic ulceration, angina,78 psychosomatic symptoms, anxiety and depression.75,85 Anticholinergic medications increase risk (OR 1.5),74 but non-steroidal anti- inflammatory drugs do not. Gastric function can influence GORD via a number of mechanisms. Overall, Helicobacter pylori seems to protect against GORD and its complications, via atrophic gastritis.86 A Japanese study found H. pylori to be present in 71% of controls, 30% of those with oesophagitis and almost 0% of those with Barrett’s.87 However, this relationship is not simple, and is dependent upon site of infection, extent and strain type. Indeed, antral infection induces acid hypersecretion and therefore may increase GORD. Helicobacter pylori eradication might therefore worsen GORD rates – however, associations with gastric carcinoma and ulceration are strong, and in practice eradication does not hamper treatment of oesophagitis with proton-pump inhibitors.88 The hypersecretion of acid seen in Zollinger–Ellison syndrome is unsurprisingly associated with higher rates of GORD and oesophagitis;89 however, supraphysiological levels of acid secretion in those with ‘normal’ GORD has not been demonstrated.90 Delayed gastric emptying seems a plausible factor in GORD, and a recent study reported a prevalence of 26% in those with GORD.91 The consequent distension hypothetically may induce more TLOSRs and acid secretion, although this is unproven. For the reasons outlined above, sliding hiatus hernia is a strong risk factor for GORD. A multicentric study found the endoscopic prevalence of hiatus hernia overall to be 5.8%, rising to 32% in those with oesophagitis. One radiological study of those with oesophagitis found a much higher rate of 90%.92 Two-thirds of those with a hiatus hernia have GORD, and oesophagitis is more common.93 The underlying mechanisms involve both an increase in duration and frequency of reflux episodes94 (Fig. 12.3). Figure 12.3 High-resolution manometry showing a single high-pressure zone in the lower oesophagus crossing the diaphragm on the left (LOS). In this spatiotemporal plot higher pressures are presented in the yellow–red spectrum and lower pressures in the green–blue spectrum. On the right there is separation of the lower oesophageal high-pressure zone (iLOS) and the high-pressure zone created by the diaphragmatic crura (cLOS) suggestive of a transient hiatus hernia. The longitudinal red and yellow zone on the right demonstrates the propagation of a peristaltic wave down the oesophagus. LOS, lower oesophageal sphincter; cLOS, crural LOS; iLOS, intrinsic LOS; UOS, upper oesophageal sphincter. Reproduced from Fox MR, Bredenoord AJ. Oesophageal high-resolution manometry: moving from research into clinical practice. Gut 2008; 57(3):405–23. With permission from BMJ Publishing Group Ltd. As discussed above, peristaltic acid clearance is an integral component of the body’s antireflux mechanism. Surrogate radiological and manometric simulation using barium fluoroscopy in one study demonstrated complete clearance with a single effective peristaltic wave greater than 20 mmHg.94 In 1968, Booth et al.95 described their standard acid clearance test, having demonstrated that those with GORD required more swallows to clear acid than controls. However, whilst subsequent studies have confirmed this, the utility of this test is very limited by both poor sensitivity and specificity96 for assessing GORD. The potential role of reduced or delayed acid clearance in GORD as a function of dysmotility is plausible and supported by a number of studies. Pathological reflux episodes last longer than physiological episodes, and this has been suggested to be due to impaired acid clearance. Corroborating evidence by DeMeester et al.97 expanded upon the different patterns of reflux seen between cases and controls. The former experienced longer nocturnal supine episodes, compared with the more physiological transient upright episodes in the latter. These nocturnal episodes are due to reduced peristaltic acid clearance,97 both in terms of reduced frequency and probably quality of peristaltic waves,98,99 compounded by a lack of gravitational clearance.100 Elevating the head of the bed compensates partially, and improves both acid clearance and microscopic oesophagitis.101 In one of the first such studies, Olsen and Schlegel102 assessed motility in 50 subjects with oesophagitis, finding normal activity in 28%, incoordinated peristalsis in 32%, hypotensive peristalsis in 37% and complete motor failure in 8%. Progressively worse motility was seen with worsening degrees of oesophagitis. In a separate study, 48% of those with severe oesophagitis displayed peristaltic dysfunction.103 A later study supported this, finding non-specific dysmotility or aperistalsis in 64% of those with benign peptic stricutres, compared to 32% of those with non-stricturing GORD.104 It is highly relevant, of course, that oesophageal sensitivity to acid is often reduced,1 and this combination of motor and sensory dysfunction may lead to particularly severe acid exposure, with resultant significant mucosal damage.105 Abnormal motility has been found in 46% of those with Barrett’s oesophagus, with a longer segment correlating with worse motility.106 These findings have recently been corroborated in a 1000-patient (with proven GORD) study by Diener et al.107 Peristalsis was normal in 56%, ineffective (hypotensive or incoordinated) in 21% and non-specifically abnormal in 23%. Those with ineffective peristalsis had worse symptoms, slower acid clearance and greater mucosal injury. Whether this dysmotility with consequent delayed acid clearance is a primary phenomenon or occurs secondary to reflux-induced damage is unclear. Eriksen et al.,108 using the solid bolus oesophageal egg transit test, compared the transit times of those with GORD and controls. Whilst delayed transit times correlated with the frequency of prolonged reflux episodes, no correlation was demonstrated between symptoms and oesophagitis and motility. The study authors argued that this dysmotility may be a primary phenomenon. Contradictory evidence exists in the form of improvements in oesophageal motility seen after antireflux surgery.109 Other studies have found no such improvements, despite objective improvements in reflux, suggesting that either reflux-induced dysmotility may be permanent or it is a primary phenomenon.110,111 Perhaps both may be true, with reflux-induced dysmotility perpetuating a vicious cycle of impaired peristalsis and LOS function. Certainly, molecular mechanisms exist for this. Inflammation has been shown to impair both oesophageal motility and LOS function. Proinflammatory cytokines such as interleukin (IL)-1B, IL-6 and IL-8 have been shown to inhibit in vitro motility.112 IL-1B induces prostaglandin E2 (PGE2)-mediated relaxation of the LOS,113 and IL-6 induces PGE2- and platelet activating factor-mediated relaxation of the LOS.114 Inflammation also increases nitric oxide and consequent inhibition of motility.115
Pathophysiology and investigation of gastro-oesophageal reflux disease
Introduction
Epidemiology
Symptoms
Normal oesophageal physiology
Antireflux mechanisms
Lower oesophageal sphincter
Diaphragmatic sphincter
Distal oesophageal compression
Other mechanical barriers
Oesophageal mucosal acid defence mechanisms
Risk factors for reflux
Inherited factors
Demographic factors
Lifestyle factors
Medical factors
Hiatus hernia
Oesophageal dysmotility and GORD: cause or effect?
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Abdominal Key
Fastest Abdominal Insight Engine