10
Palliative treatments of carcinoma of the oesophagus and stomach
Epidemiology and survival
Accurate information about the proportion of patients with oesophageal and gastric cancer who are treated with palliative intent is difficult to obtain. This largely reflects variations in the selection of patients for treatment. National audit data from England and Wales show that about 64% of new patients with oesophageal or gastric cancer undergo primary palliative treatment, although this varies between cancer networks and worldwide.1–3 Trials continue to examine the role of palliative chemotherapy or radiotherapy in oesophageal cancer, but currently there is no evidence to show a survival benefit when compared to best supportive care.4 The median survival is less than 8 months and few survive beyond 1 year. It is recommended, therefore, that treatment is tailored to the general status of the patient in order to reduce symptoms with minimal risks and side-effects.5
The resection rate for patients with gastric cancer is greater than that for oesophageal cancer (25%), probably because distal gastrectomy is still widely employed to overcome gastric outlet obstruction even in patients with advanced disease.1 Gastrojejunostomy (laparoscopic or open) or endoscopic stenting are also commonly performed if the tumour is very advanced or the patient frail. There is a lack of well-designed and conducted trials comparing distal gastrectomy with bypass or duodenal stenting, but systematic reviews comparing gastrojejunostomy with endoscopic stenting suggest that stent placement may be more favourable in patients with a very short life expectancy, although bypass is preferable in patients with a prolonged prognosis.6,7 The randomised trials examining these issues are often small, however, and well-designed studies are still needed. Palliative chemotherapy, either alone or in combination with biological therapies, may lead to improved survival in gastric cancer. A Cochrane systematic review shows that chemotherapy significantly increases survival in comparison with best supportive care and that combination chemotherapy improves survival compared to single-agent 5-fluorouracil (5-FU).8 It is also recommended that patients are tested for human epidermal growth factor receptor 2 (HER-2) status and a monoclonal antibody (trastuzumab) be added in patients with HER-2-positive tumours.8,9 Despite these improvements in the treatment of advanced gastric cancer overall outcomes remain poor, with a median survival of less than 14 months.
Disease stage, age and general performance status influence outcomes and survival, although the effect of age may be largely due to more comorbidity in older patients.10 Another predictor of mortality is the length of the oesophageal tumour, mainly because this increases the likelihood of nodal involvement with large tumours.11 All these factors need to be taken into consideration when planning treatment.
Patient selection and multidisciplinary teams
Since the introduction of the National Health Service Cancer Plan in the UK in 2000, treatment decisions for patients with cancer are mandated to be made within the context of a multidisciplinary team.12 Guidelines for the constitution and processes for upper gastrointestinal multidisciplinary teams have been published and national peer review processes audit team working.12,13 Teams consist of core members, specialist nurses, gastroenterologists, oncologists, pathologists, radiologists, administrators, palliative medicine experts and surgeons. Additional members may include cytologists, dieticians and researchers from clinical trials units. The aim of the team is to review available evidence for each new patient and make optimal treatment decisions. Evidence includes information about the cell type, disease stage, patient comorbidity and choice, and expert discussion of best available treatments. Although team working has been widely implemented across the UK and is recommended by some continental European centres, in North America a similar role of ‘tumour boards’ is not mandatory within cancer care.14 Currently, evidence to support team working is sparse, based upon longitudinal or retrospective case series. It is also uncertain how to best evaluate the quality of multidisciplinary teams because outcomes are dependent upon so many variables. It has been suggested that monitoring implementation of team decisions further evaluates team working. In one centre it has been shown that 15% (95% confidence interval (CI) 10–20%) of team decisions change after the meeting.15 The most common reason cited for changing team decisions was lack of available information about patient choice and comorbidity. There is also uncertainty whether upper gastrointestinal multidisciplinary teams should routinely discuss patients who develop disease recurrence following radical treatment.16 If this becomes mandatory in the UK the workload of teams would increase; however, patients with recurrence should be offered the full range of palliative treatments and so this requires further consideration. Team working is an area that is likely to develop over the next decade; professionals may need training in team-working skills and the infrastructure to support these processes is required.17
After establishing a diagnosis, new patients require careful assessment to decide whether treatment should be directed towards attempting a cure, or if palliation of symptoms is more appropriate. Careful patient selection has been shown to significantly influence results. Principal factors to consider are the type and stage of the tumour, physical and psychological well-being of the patient, and knowledge of patient preferences. Decisions should be considered in the knowledge of treatment outcomes, including impact on patients’ health-related quality of life. Figs 10.1 and 10.2 illustrate pathways that can be used to select patients for palliative treatment.
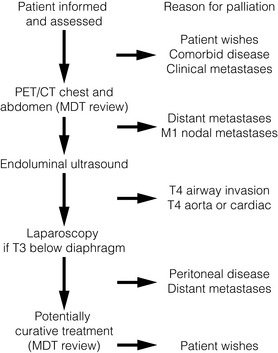
Figure 10.1 Algorithm for selection for palliative or curative treatment of oesophageal and junctional tumours. CT, computed tomography; MDT, multidisciplinary team; PET, positron emission tomography.
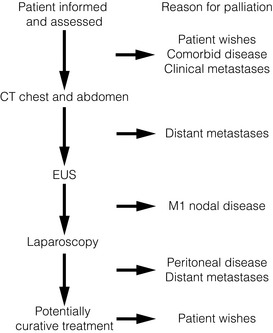
Figure 10.2 Algorithm for selection for palliative or curative treatment for cancers of the gastric body or antrum. CT, computed tomography; EUS, endoscopic ultrasonography.
Fitness for treatment
The place of oesophagectomy in many older patients is often easily settled because of general debilitation or multiple coexistent medical problems. Age in itself does not preclude octogenarians from surgery, but in most series older patients are carefully selected. In general, patients who are not fit enough for oesophagectomy are also unable to tolerate a radical course of radiotherapy or definitive chemoradiation. On the whole, surgery for gastric tumours is better tolerated than oesophageal surgery by the elderly population, but patients still require careful preoperative assessment before undergoing major resection. Anaesthetic assessment for surgery is considered in more detail in Chapter 4.
Patient preferences and information provision
Information about the diagnosis and prognosis of oesophageal and gastric cancer should be offered to all patients and it is essential that a nurse specialist is involved in this process whenever possible. The volume and type of information required will vary between individuals, although evidence from studies of patients’ information needs performed in other disease sites generally shows that patients wish to have as much information as possible and prefer the information to be provided by a health professional, as well as in other forms such as a booklet or CD-ROM.17 It is necessary to inform patients of the potential treatments and alternatives together with treatment-related benefits and risks, both in the short and long term. Surveys of patients’ information needs also show that information about impact on health-related quality of life is considered important to patients during treatment decision-making.18 Ensuring that consultations provide this information in a way that is understood is difficult and requires that professionals are trained. In the UK it is recommended that all specialist cancer teams undergo training in advanced communication skills.19 All clinicians will be faced with patients who demand every small chance of cure, despite its risks, and others who wish to receive minimal, dignified intervention. Communicating outcomes, providing adequate information and listening to patients’ views is necessary so that patients and their families have access to as much information and support as required.
Symptoms and signs of advanced oesophageal and gastric cancer
Tumours of the gastric body and antrum
Gastric cancer commonly has an insidious presentation and some patients have few symptoms. Slow blood loss may eventually result in symptoms of anaemia. Haematemesis is a rare first presentation. Vague upper gastrointestinal problems, such as epigastric discomfort, early satiety and gastro-oesophageal reflux, are common. Tumours of the distal stomach cause outlet obstruction and patients describe epigastric fullness, reflux and nausea, finally leading to effortless vomiting. The presence of an epigastric mass, supraclavicular lymphadenopathy, jaundice, ascites or pleural effusions all reflect advanced disease. Less commonly, bony pain and symptoms of increased intracranial pressure are seen related to metastatic spread. Symptoms of oesophageal and gastric cancer are listed in Box 10.1.
Palliative treatments for cancer of the oesophagus and gastric cardia
A variety of approaches are available for the palliation of advanced tumours of the oesophagus and gastric cardia. These include rigid plastic tube insertion, self-expanding metal stents (SEMS), brachytherapy, external beam radiotherapy, chemotherapy, chemical and thermal ablation, palliative resection (for gastric cancer) and bypass surgery either as single or combination treatment modalities. These different treatment modalities can treat and palliate the cancers and therefore there is no longer a role for palliative surgery for oesophageal cancer, which has a major detrimental impact on patients’ quality of life.20 Generally, patients undergoing palliative surgery do not have sufficient time to recover from the operation before they experience symptoms of metastatic disease. Historical data also show that palliative resection is associated with high perioperative mortality and morbidity rates. It is possible that the improved surgical techniques and perioperative care may mean that minimal access surgical resection may in certain situations be suitable for the palliation of oesophageal cancer; however, well-designed studies are needed to corroborate this. Evidence for the effectiveness of non-surgical interventions for the palliation of malignant dysphagia in the treatment of primary oesophageal cancer is summarised in a recent Cochrane review that includes 40 studies.21 Overall, it concluded that SEMS insertion is safe, quick and effective in palliating malignant dysphagia compared to other modalities. High-dose intraluminal brachytherapy may be a suitable alternative and provide additional survival benefit and quality of life. The individual studies examining endoscopic methods of relieving luminal obstruction are considered below, with other sections concentrating on treatments for palliation of other common problems in oesophageal or oesophagogastric junctional cancer:
1. Endoscopic methods of relieving luminal obstruction.
2. Chemotherapy, radiotherapy, chemoradiotherapy and monoclonal antibody treatments.
3. Management of aero-digestive fistulas.
4. Management of recurrent laryngeal nerve palsy.
The endoscopic relief of luminal obstruction
Malignant dysphagia may be relieved by stent insertion, brachytherapy, external beam radiotherapy, chemotherapy or tumour ablation with photothermal, photodynamic therapy, or by the injection of cytotoxic substances.20 Many modalities are complementary and SEMS insertion is safe, effective and quicker in palliating dysphagia compared to other modalities. No one method or combination is greatly superior to the rest in terms of relief of dysphagia, although some evidence is emerging to show better long-term relief of dysphagia with high-dose intraluminal brachytherapy. This might provide a suitable alternative and may provide additional survival with a better quality of life compared to metal stent placement.22 Historically, dilatation was advocated for the palliation of malignant dysphagia and rigid plastic tubes were inserted following this. Because of the short-lived benefits of dilatation alone and the associated risks of perforation, its use nowadays is reduced to that of a preliminary measure before definitive management of dysphagia. Minimal oesophageal dilatation may be performed to allow insertion of an SEMS or to place a brachytherapy bougie. Guidelines on the use of dilatation in clinical practice recommend careful preparation, polyvinyl wire-guided bougies or hydrostatic balloons.23 Strictures with severe narrowing and angulation are best negotiated under X-ray screening.
The majority of randomised trials evaluating palliative treatments for dysphagia have been small and single-centred, and may therefore have lacked power to detect differences between treatment arms. Table 10.1 summarises the randomised controlled trials published before the end of 2011, evaluating interventions of the palliation of malignant dysphagia. The table includes only trials that randomised more than a total of 50 patients, excluding smaller studies because they are less likely to influence practice. Even within the included studies, eight (50%) may be at risk of selection bias because methods used to conceal the allocation sequence were unclear (i.e. intervention allocations could have been foreseen before or during enrolment).
Table 10.1
Prospective randomised controlled trials of endoscopic palliation of malignant dysphagia (n > 50)
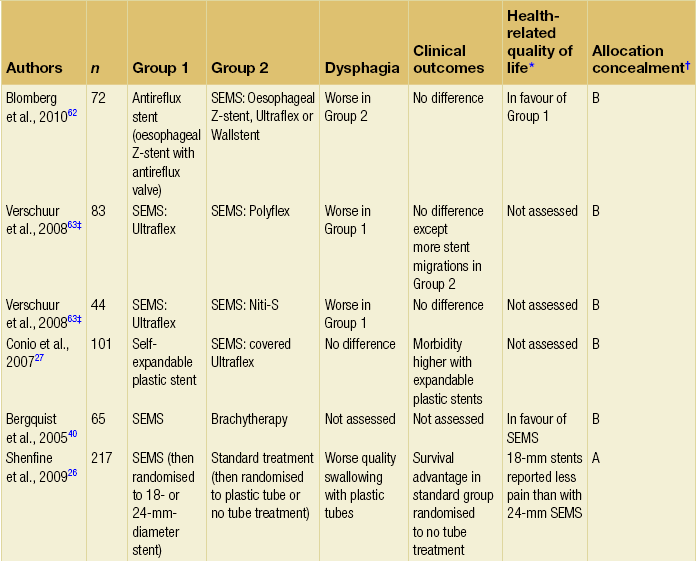
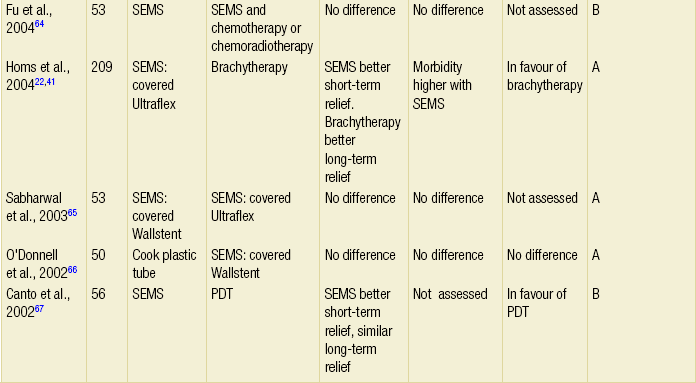
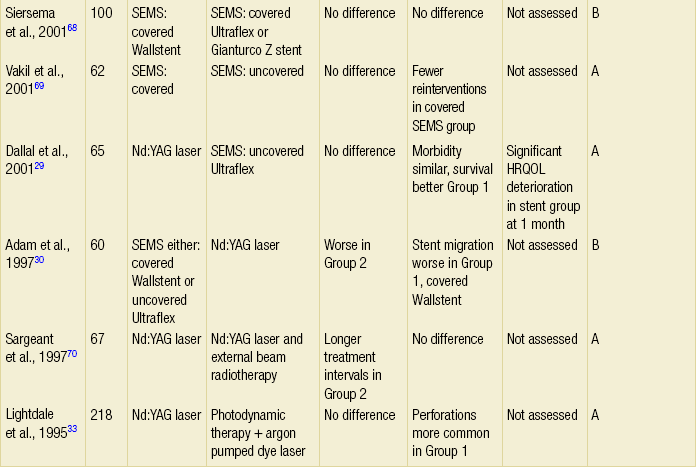
Note: 30-day mortality rates were similar in all of the above trials.
*Health-related quality-of-life results in article reported from a valid multidimensional questionnaire.
†The risk of bias in the trial was judged as A = low, B = unclear or C = high, using the Cochrane risk of bias tool assessing allocation concealment.71 The tool allows an author to judge whether the randomisation process described in the study has clear evidence that the treatment allocation was concealed to the person randomising the patient before the patient is entered into the study.
‡This randomised controlled trial had three treatment arms and subsequently two comparisons.
Intubation
Intubation is probably the most widely used form of palliation of malignant dysphagia at present and allows rapid relief of dysphagia with associated low morbidity. Prostheses may be placed endoscopically, radiologically or surgically at laparotomy, although there is little place for open insertion of a prosthesis when a tumour is unexpectedly found to be irresectable because endoscopic insertion is safer and has fewer complications. Self-expanding metal stents are now routinely employed for this purpose and plastic rigid tubes are generally no longer in use.21,24–26
Self-expanding metal stents (SEMS): The design of SEMS has evolved since they were first introduced for the palliation of malignant dysphagia in the early 1990s with the introduction of covering and also fixtures to allow endoscopic stent removal. They are made from a flexible metal mesh that expands after deployment for up to 48 hours, leading to rapid relief of dysphagia and creation of an internal luminal diameter of 16–25 mm. Early disadvantages of tumour ingrowth and stent migration have been largely overcome by newer materials and designs, although migration may still occur when stents are placed at the oesophagogastric junction. Although initially stents were expensive (about £500–800), the cost is now reducing. Current design developments are centred upon using expandable plastic rather than metal to reduce the manufacturing costs, and developing removable stents and possibly dissolvable stents to use in temporary settings.27 Stents nowadays, therefore, may be fully or partially covered (partially covered self-expanding metal stents, PCSEMS). Several studies have investigated the addition of a valve in the distal part of the stent to reduce acid reflux.28
Method of insertion: Self-expanding metal stents may be inserted endoscopically or radiologically. There are several designs with very similar delivery devices. The Ultraflex Esophageal Stent (Boston Scientific Inc.) is made of an alloy of titanium and nickel and has a shape ‘memory’ as well as superelastic behaviour. It is loaded in a small-diameter delivery catheter, constrained in a compressed form by a double plastic membrane. During expansion the stent shrinks by approximately one-third. It is available either uncovered or partially covered. The design incorporates a proximal flare for secure placement and to reduce the possibility of food entrapment. The conical ‘Flamingo’ Wallstent is designed to reduce problems with migration, and the proximal and distal 1.5 cm of the stent remain uncovered. It may be recovered during deployment and repositioned, provided less than 50% of the endoprosthesis has been released. The Gianturco Z stent also uses stainless steel and it is entirely coated with a polyethylene film. It has long wire hooks at its mid-portion to facilitate anchoring. Unlike the Ultraflex and Wallstents it undergoes very little shortening upon release. A ‘windsock’ design to reduce the possibility of gastro-oesophageal reflux is available. Other stents are variations on these basic designs. Comparative studies show that reintervention rates for tumour ingrowth are higher with uncovered than covered stents. Other comparative studies of SEMS show conflicting results and although these trials may have design weaknesses, there is currently no good evidence that one design is superior to another in terms of morbidity or relief of dysphagia.
Preparation: Endoscopic prosthesis insertion is usually possible under intravenous sedation, although some endoscopists continue to use general anaesthesia. Routine monitoring is required with intravenous sedation, as is continual attention to the airway. Saliva and regurgitated fluids should be constantly removed to prevent aspiration during the procedure.
Endoscopic insertion with fluoroscopy: After endoscopic assessment and measurement of the tumour, a guidewire is passed into the stomach (after successful negotiation of the tumour with the endoscope or under fluoroscopic control). Occasionally, dilatation may be required to a minimum of 10 mm before passage of the delivery system over the guidewire. The proximal and distal extents of the tumour may be marked with radio-opaque skin markers or the tumour limitations injected with contrast. The slim delivery device is advanced over the guidewire until the radio-opaque markers of the compressed stent are correctly aligned with the tumour. Once in position the stent is deployed. It is possible to reposition some of the stents after partial deployment. The guidewire and delivery device are then carefully removed under fluoroscopic guidance. After release of the stent, the endoscope may be reinserted to check the final position. Immediate balloon dilatation is recommended to improve expansion and prevent early migration, but may still be performed up to several days after stent insertion.
Radiological insertion: Morphological imaging of the malignant stricture with oral contrast is performed prior to stent insertion. This assesses length and position of the tumour. A fine steerable catheter is then negotiated over a guidewire through the stricture to the stomach and skin markers aligned. The proximal and distal ends of the tumour are marked (similar to endoscopic positioning). Balloon dilatation to 10–15 mm may be performed if the stricture is very narrow. The stent insertion device is then passed safely and positioned radiographically over the guidewire and released according to the type of stent.
Postoperative management: After stent insertion the patient must be instructed to sit upright. Oral fluids are usually allowed the same day unless there is concern about complications or symptoms or signs of perforation. Clinical and radiological examination may be performed to exclude perforation before oral fluids are commenced. Patients should receive written dietary information with advice to chew food carefully and drink regularly during and after meals. A daily intake of 10 mL hydrogen peroxide (20 vol.) is sometimes recommended.
Complications: Even in experienced hands, intubation with SEMS has a procedure-related mortality of about 1–2% and early complication rates of between 0% and 30%. Complications are listed in Box 10.2.
1. Malposition of the stent may require insertion of a second or even third stent (if the tumour is long). This may overlap the malpositioned stent to adequately cover the tumour.
2. Incomplete stent expansion and early dysphagia may require balloon dilatation if no improvement is seen within 48 hours.
3. Early stent migration. This occurs in about 1% of patients and is more prone in stents placed at the oesophagogastric junction than in stents with both ends anchored within the oesophagus. Endoscopic retrieval may be performed safely, especially with the newer devices. Stents that have migrated into the stomach may also be safely left as they rarely obstruct the pyloric channel or cause intestinal perforation.
4. Oesophageal perforation is the most serious complication and is more likely if the stricture has been dilated before stent insertion, there has been prior use of radiotherapy and/or chemotherapy, if the tumour is sharply angulated or if it extensively encases the oesophagus. Rapid development of subcutaneous emphysema, severe pain, radiological evidence of pneumomediastinum, air under the diaphragm or a pleural effusion should all raise suspicion. The extent of the leak is confirmed by contrast radiography. The most appropriate form of therapy depends on the time of detection and the extent of the leak. If recognised at endoscopy, the insertion of the prosthesis itself may seal off the perforation and prevent mediastinitis. Alternatively, the procedure may be abandoned and conservative treatment undertaken. This involves administration of broad-spectrum antibiotics, cessation of oral intake and feeding either parenterally or by jejunostomy. An intercostal drain may need to be inserted if there is evidence of pleural contamination. Specific management of this serious complication is covered in detail in Chapter 19.
5. Severe upper gastrointestinal haemorrhage occasionally occurs. This is difficult to treat, and only supportive measures may be possible.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







