rIFN: recombinant-interferon.
Figure 27.1 Meta-analysis of interferon therapy for HBeAg positive chronic hepatitis B: effect of treatment, measured as risk difference, on HBeAg clearance.
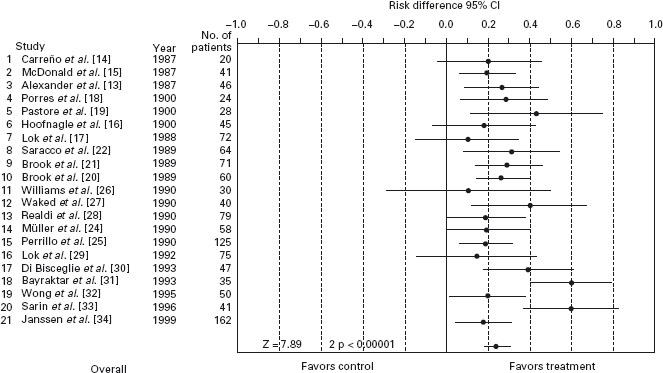
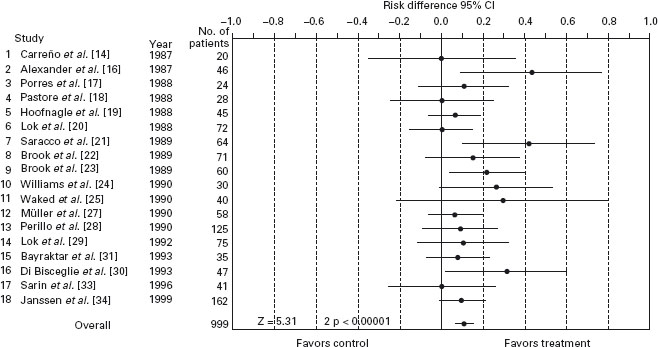
Figure 27.2 Meta-analysis of interferon therapy for HBeAg positive chronic hepatitis B: effect of treatment, measured as risk difference, on loss of HBsAg.
Pegylated IFN in HBeAg positive chronic hepatitis B
Only two RCTs comparing peginterferons to IFN in patients with HBeAg positive chronic hepatitis B have been published so far [38, 39]. The first study used different doses of pegylated IFN α-2a (90,180 and 270 μg sc weekly for 24 weeks); in the second one patients received pegylated IFN α-2b at a dose of 1.0μg/kg weekly for 24 weeks. The rate of combined response at the end of follow-up (HBeAg negative, HBV DNA < 5log10 copies/ml, and ALT normalization) was significanlty higher in patients treated with pegylated IFN (21.3% vs 11.2%; OR, 2.16; 95% CI: 1.34–3.51). A1d
IFN and corticosteroids in HBeAg positive chronic hepatitis B
An alternative approach to chronic HBV infection is based on the induction of a brief period of immunosuppression by steroids [40,41], then a withdrawal to provoke an abrupt ALT elevation due to the host immune reconstitution, and a subsequent decline of HBV-DNA. IFN administration is then started 2–4 weeks after stopping steroids. The sequential schedule has been studied in some RCTs and their results have been pooled in a meta-analysis [42]. The overall rate of HBeAg loss in 13 RCTs was significantly higher in the prednisone-IFN group (34.7% vs 29.1%, OR 1.41; 95% CI: 1.03–1.92). Similar results were observed for loss of HBV-DNA (39.7% vs 31.6%; OR, 1.51; 95% CI: 1.12–2.05). However, the loss of HBsAg was similar across the two groups (7.3% vs 5.4%; OR, 1.41; 95% CI: 0.77–2.59). Pre-treatment ALT levels had no impact on evaluated outcomes but only the low dose of prednisone was significantly associated with HBsAg loss (OR, 2.76; 95% CI: 1.18–6.43) as compared to high dose (OR, 0.69; 95% CI: 0.29–1.66).
Even if there could be an advantage in pre-treatment with steroids of this subset of patients, this must be balanced against the risk of flare of liver disease after steroid withdrawal. A severe, sometimes fatal “seroconversion hepatitis” has been reported in subjects with pre-existing cirrhosis [43, 44]. This treatment algorithm is not recommended.
Pegylated IFN and lamivudine in HBeAg positive chronic hepatitis B
Another approach to increase the rate of viral clearance is to add another antiviral drug to the current therapy. Three RCTs have tested this hypothesis and patients were rand-omized to receive peginterferon plus LAM or peginter-feron alone [45–47]. The rate of persistent viral clearance, as evaluated by serum HBV-DNA levels <400 copies/ml at the end of follow-up, was similar across the two groups (11.6% vs 10.9%%; OR, 1.07; 95% CI: 0.73–1.58), and therefore it can be concluded that the association of pegylated IFN with LAM is not superior to peginteferon alone in treating patients with HBeAg positive chronic hepatitis B and this schedule is not recommendable in the clinical practice. A1c
IFN in HBeAg negative chronic hepatitis B
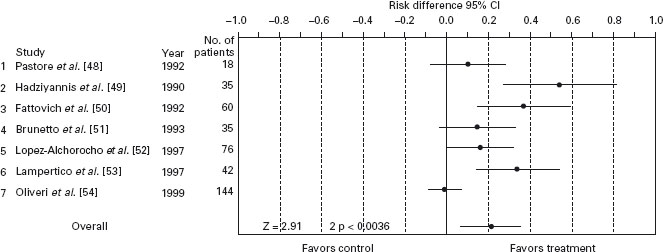
Data on the efficacy of IFN therapy in HBeAg negative chronic hepatitis B are few compared to HBeAg positive. The results of published RCTs remain inconsistent and the overall assessment of the treatment effect is difficult to evaluate. The drawing of firm conclusions based on the results of these studies is hampered by the small sample size of the studies and by heterogeneity of baseline severity of patients and the schedule of treatment. An evaluation of treatment efficacy on HBV-DNA clearance and sustained ALT normalization was performed by a meta-analysis on seven RCTs [48–54] enrolling patients infected by the HBe minus mutant. Five RCTs compared IFN regimens with non-active treatment; two trials compared different doses of IFN. We combined the results of IFN arms of these two trials and made a single pair-wise comparison with the overall control rate of the other five RCTs. All the RCTs were performed in centers from the Mediterranean area, indirectly confirming the high geographical prevalence of this mutation. Pooled data totalling 301 subjects, showed a significant effect of IFN therapy on sustained loss of HBV-DNA (Risk difference 0.17; 95% CI: 0.07–0.27; NNT 5.9) (see Figure 27.3). A1c
IFN or pegylated IFN and lamivudine in HBeAg negative chronic hepatitis B
No study has properly compared peginterferon to IFN therapy in patients with HBV mutant infection, but, similarly to HBeAg positive patients, the combination therapy of IFN or peginterferon plus LAM in comparison to IFN alone has been tested also in anti-HBeAg positive subjects. One study used IFN-α [55] and three PEG IFN [56–58]. Overall, 225 patients received combination therapy and 219 IFN alone. The rate of HBV-DNA undetectability at the end of follow-up was 18.2% and 21.0% respectively (OR, 0.83; 95% CI: 0.51–1.35), so addiction of LAM does not improve efficacy of IFN.
Nucleoside and nucleotide analogs
Evidence of effectiveness from phase III randomized trials is available in the literature for nucleoside analogs (LAM, LdT and ETV), and nucleotide analogs (ADV and TDF). These drugs are administered by the oral route and display a powerful inhibitory effect on HBV-DNA polymerase [59]. The examined “surrogate” endpoints (6-24 months) were ALT normalization, reduction of serum HBV-DNA levels during therapy, the rate of undetectable serum HBV DNA at the end of therapy, the rate of HBeAg loss or anti-HBe seroconversion at the end of therapy for patients with HBeAg positive hepatitis, the rate of HBsAg loss, anti-HBs seroconversion and the histological improvement at the end of treatment. Regarding the values of serum HBV-DNA during treatment and at the end of therapy, the RCTs reported different methods and different units to measure viral load. The serum HBV-DNA load was measured using polymerase chain reaction (PCR) assay or reverse transcription PCR assay or solution hybridization assay. The detection limits to evaluate the response ranged from 1.6pg/ml (150,000 copies/ml) to 300 copies/ml.
Figure 27.3 Meta-analysis of interferon therapy for HBeAg negative chronic hepatitis B: effect of treatment, measured as risk difference, on sustained loss of HBV-DNA.
Finally, the majority of studies evaluated the rate of appearance of mutations in specific regions of the polymer-ase-encoding HBV gene (so-called YMDD mutant and others) which were associated with the loss of efficacy of the drugs [60].
Lamivudine The original observations on the anti-HBV efficacy of LAM came from the treatment of HIV infected subjects who were also HBsAg positive [61–62]. The optimal dosage in immunocompetent patients with HBV chronic hepatitis, found by a dose-ranging study [63] and confirmed by an RCT [64], was 100 mg daily. It is important to consider that only a minority of published full papers are controlled trials with an appropriate control group.
The first two RCTs evaluated the efficacy of LAM therapy in HBeAg positive chronic hepatitis previously untreated and used a control arm treated with placebo. The first RCT performed in China [64], included exclusively Oriental subjects, and the second trial was performed in USA and included prevalently white patients [65]. Both studies reported that a course of 12 months of LAM had favorable effects on histological, virological, and biochemical features of the disease and was well tolerated. At the end of treatment the rate of HBeAg seroconversion was 16% and 17% respectively in treated groups and 4% and 6% in control groups (OR, 3.06; 95% CI: 1.56–6.01). The histological response, evaluated as a reduction of at least two points of grading Knodell score, was more frequent in treated groups with a rate between 49% and 52%. Finally, the overall incidence of YMDD mutations at the end of treatment was of 14% in the Chinese trial and 32% in the American trial.
RCTs which compared the efficacy of 48–52 weeks LAM therapy versus IFN monotherapy (standard or pegylated IFN) or combination therapy with IFN and LAM [66–70], reported that among patients allocated in the LAM arm the rate of ALT normalization ranged between 23% and 78%, the rate of HBe seroconversion ranged between 19% and 28%, and the incidence of YMDD mutations was 8–40%. The identified predictors of virological relapse were older age, male sex and low levels of aminotransferases at the start of therapy.
The only published RCT including patients with anti-HBe positive chronic hepatitis used a schedule of 52 weeks of therapy with LAM [75]. Patients were randomized to receive 100 mg LAM orally once daily for 52 weeks or placebo for 26 weeks. The primary efficacy endpoint was loss of serum HBV-DNA with a cutoff of 1.6pg/ml) plus normalization of ALT at week 24. A significantly higher proportion of patients receiving LAM (63%) had a complete response at week 24 compared with patients receiving placebo (6%) (p < 0.001). In a ranked assessment of pretreatment and post-treatment biopsy pairs 11% improved, 86% showed no change, and 2% worsened in fibrosis. At week 52, 27% of patients receiving LAM had YMDD variant HBV.
There are two major problems with LAM treatment: the occurrence of YMDD mutants under therapy and the stability of viral suppression after drug discontinuation. Appearance of the YMDD mutant was associated with an ALT relapse in 30–70% of cases, and usually both the YMDD mutant and the ALT peak subside rapidly upon stopping LAM. The efficacy of LAM to inhibit HBV replication was limited only to the period of drug exposure [72–75].
Telbivudine LdT is a potent and specific inhibitor of HBV-DNA polymerase. A double-blind pre-clinical study evaluated the efficacy and safety of two different doses of LdT (400 or 600 mg/day) alone or plus LAM (100mg/day) compared with LAM alone in adult patients with HBeAg positive chronic hepatitis [76]. At the end of 52 weeks of therapy, LdT monotherapy showed a significantly greater mean reduction in HBV-DNA levels (6.01 vs 4.57log10 copies/ml; p < 0.05), rate of undetectable HBV-DNA by PCR with a cutoff limit of 105 copies/ml (61% vs 32%; p < 0.05), and rate of ALT normalization (86% vs 63%; p < 0.05) compared with LAM monotherapy. The rate of HBeAg seroconversion (31% vs 22%) was not significantly different. Combination treatment with LdT and LAM was not better than LdT alone. A1d
In a phase III RCT [77], 1370 patients (more than 80% Asian) with HBsAg positive chronic hepatitis (921 HBeAg positive and 446 HBeAg negative) were randomly assigned to receive 600 mg of LdT or 100mg of LAM once daily. At week 52, a significantly higher proportion of HBeAg positive and HBeAg negative patients receiving LdT than of those receiving LAM had a reduction in the serum HBV-DNA levels to fewer than 5log10 copies/ml (60.0% vs 40.4%, p = < 0.001 and 88.3% vs 71.4%, p < 0.001 respectively). The histological response was different in HBeAg positive patients (64.7% vs 56.3%, p = 0.01), but not in HBeAg negative patients (66.6% vs 66.0%, p = 0.90). The rate of HBeAg loss and HBeAg seroconversion was comparable in HBeAg positive patients in both groups of treatment. The incidence of viral resistance during 52 weeks of therapy was lower in patients treated with LdT than in these treated with LAM (5.0% vs 11.0% in HBeAg positive patients, p < 0.0001, and 2.2% vs 10.7% in HBeAg negative patients, p < 0.001). The frequency of adverse events was similar for patients receiving LdT and LAM, and serious adverse events were reported in 2.6% of patients receiving LdT and 4.8% receiving LAM. A1c
In another RCT trial conducted in China, 332 HBeAg positive or HBeAg negative patients were treated with 600mg of LdT or 100mg of LAM daily for 104 weeks [78]. The primary efficacy endpoint was reduction in serum HBV-DNA levels at week 52 of treatment. Secondary end-points included clearance of HBV-DNA to undetectable levels, HBeAg loss, seroconversion and ALT normalization. At week 52, among 290 HBeAg-positive patients, mean reductions of serum HBV-DNA (6.3log10 versus 5.5log10, p < 0.001) undetectable serum HBV-DNA by PCR (67% vs 38%, p < 0.001), ALT normalization (87% vs 75%, p < 0.007), and HBeAg loss (31% vs 20%, p < 0.047) were significantly more common in the LdT group. Viral resistance in LdT recipients was approximately half that observed with LAM, but this difference was not statistically significant. A1c
Finally, a randomized open-label trial compared the antiviral efficacy of LdT and ADV in 135 treatment-naive HBeAg positive patients with chronic hepatitis [79]. Patients were randomly assigned to received 52 weeks of LdT or ADV or 24 weeks of ADV and then LdT for the remaining 28 weeks. The primary efficacy comparison was serum HBV-DNA reduction at week 24 and at week 52. At week 24, mean HBV-DNA reduction was greater in patients with received LdT than in two groups who received ADV (–6.30 vs –4.97 log10 copies/ml; p < 0.001), and more patients treated with LdT had undetectable HBV-DNA by PCR, which has a lower limit of detection of 300 copies/ ml. (39% vs 12%; p < 0.001). At week 52, the rate of undetectable HB-DNA by PCR (60% vs 40%, p = 0.06), the rate of HBeAg loss (30% vs 21%, p = 0.3), HBeAg seroconversion (18% vs 19%, p = 0.3) and the rate of ALT normalization (79% vs 85%, p 0.4) were similar in patients treated with LdT or ADV.
Entecavir ETV is a cyclopentyl guanosine analog that inhibits both the priming and elongation steps of viral replication. In a phase III RCT [80], 715 nucleoside naive patients (60% Asian and 40% white) with HBeAg positive chronic hepatitis were randomized to receive 0.5 mg of ETV or 100mg of LAM once daily for 52 weeks. The primary efficacy endpoint was histological improvement (a decrease by at least two points in the Knodell necroinflammatory score) at week 48. Secondary endpoints included a reduction in the serum HBV-DNA level, HBeAg loss and seroconversion, and normalization of ALT level.
At 48 weeks, the ETV treated patients had higher rates of histological improvement (72% vs 62%, p = 0.009), HBV-DNA reduction (–6.9 vs –5.41og10), undetectable HBV-DNA (67% vs 36%, p < 0.001), and ALT normalization (68% vs 60%, p = 0.02). HBeAg seroconversion occurred in 21% of ETV treated patients and 18% of those treated with LAM (p = 0.33). Safety was similar in the two groups. A1c
Another study showed that the antiviral activity of ETV is greater than that of ADV in patients with HBeAg positive chronic hepatitis who are treatment-naive [81]. In this study 69 patients with high levels of baseline HBV-DNA (>108 copies/ml) were treated with ETV 0.5mg/day or ADV 10mg/day for 52 weeks. ETV was superior to ADV for mean change from baseline in serum HBV-DNA levels at week 12 ((–6.231og10 copies/ml vs –4.421og10 copies/ml respectively; p < 0.0001). The proportion of patients achieving HBV-DNA less than 300 copies/ml were greater in ETV-treated than ADV-treated patients at weeks 12, 24, and 48. At week 48 only 3% of ETV treated patients versus 47% of ADV treated patients had HBV-DNA of 105 copies/ ml or more. A1c
In HBeAg negative chronic hepatitis only one RCT compared ETV and LAM treatment [82]. A total of 648 patients were randomized to receive either ETV 0.5 mg/day or LAM 100mg/day for 48 weeks. More patients in the ETV group than in the LAM group had undetectable serum HBV-DNA levels (<300 copies/ml) (90% vs 72 %; p < 0.001) at the end of treatment. ALT normalization was also observed more frequently with ETV than with LAM (78% vs 71%, p = 0.045), but there was no difference in improvement in fibrosis. There was no evidence of resistance to ETV. Safety and adverse-event profiles were similar in the two groups. A1c
Adefovir dipivoxyl ADV, a nucleotide analog, has a potent in vitro and in vivo effect against herpes virus, retroviruses and hepadnaviruses. The drug, when given orally at a dose of 10mg daily, inhibits both the wild type and LAM-resistant HBV strains with an excellent safety profile [83]. Renal tubular damage has been observed when prolonged treatment with higher doses has been given [84]. Two phase III multicenter RCTs, one in HBeAg positive patients have been published [85,86]. In the first study, 1515 patients worldwide with HBeAg positive chronic hepatitis B were randomized to receive 10mg or 30mg of ADV or placebo for 48 weeks. Most of them were treatment naive, since only 123 had been previously treated with IFN. The observed reduction of HBV-DNA levels was 4.76 with 30mg vs 3.52 log copies/ml with 10mg of ADV; in both cases a significantly greater suppression than that observed in the placebo group. The rate of biochemical and histologi-cal improvement was comparable between two ADV regimens (59% vs 53% and 55% vs 48% respectively). HBeAg seroconversion, although significantly more common in patients receiving ADV (12% at 30mg, 14% at 10mg) than in the control group (6%, p = 0.049 and 0.01 respectively), was relatively uncommon. No ADV-associated resistance mutations were identified in the HBV-DNA polymerase gene. Mild nephrotoxicity was observed with the 30 mg regimen, and thus 10mg was considered to be the best regimen. In the second RCT 480 Chinese subjects with hepatitis HBeAg positive chronic hepatitis were enrolled in a multicenter, double-blind, randomized, placebo-controlled study of ADV 10mg once daily [26]. There was a significant difference in reduction of serum HBV-DNA levels after 12 weeks between subjects who received ADV and those who received the placebo (3.4 and 0.1log10 copies/ml respectively, p < .001). A higher proportion of subjects with undetectable serum HBV-DNA and with normal ALT was observed in ADV treated subjects at week 52 (median HBV-DNA reduction of 4.5log10 copies/ml, 67% with HBV-DNA <105 copies/ml, 28% with HBV-DNA undetectable, and 79% with ALT normalization). Subjects with YMDD mutant HBV at baseline had virological, biochemical, and serological responses to treatment that were similar to those of subjects with wild-type HBV. The incidence of clinically adverse events was similar in nature and severity between the treatment groups, and there was no evidence of renal toxicity. No ADV related HBV mutations were identified. A1c
Patients with chronic HBeAg negative, HBV-DNA positive chronic hepatitis were studied in a multicenter trial [87], comparing the efficacy of 10mg of ADV daily for 48 weeks to placebo. Viral suppression was obtained in 51% of patients on ADV vs none in the placebo group. The median HBV-DNA level of ADV-treated patients at 48 weeks (3.91 log copies/ml) was lower than observed with placebo (1.35log copies/ml, p < 0.001). HBsAg seroconversion was never achieved. Histology changed significantly in the ADV group, with 64% of patients improved as compared with 33% of those on placebo, and alanine ami-notransferase level normalized in 72% on ADV and 29% on the placebo. In both studies, YMDD or other repetitive mutations in the HBV polymerase region did not occur under either dose of ADV during the 48 weeks of treatment.
Tenofovir TDF is an acyclic nucleotide analog with a molecular structure related to that of ADV. Data from several small studies suggest that TDF might be more potent than ADV in inducing the early and rapid suppression of HBV-DNA in LAM resistant patients [88–90]. In two double-blind, phase III RCTs [91], patients with HBeAg positive or HBeAg negative chronic hepatitis were assigned in a 2:1 ratio to receive 300mg of TDF or 10mg ADV once daily for 48 weeks. The primary efficacy endpoint was HBV-DNA level less than 400 copies/ml and histologic improvement (a reduction in the Knodell necroinflamma-tion score of two or more points without worsening of fibrosis) at week 48. The first RCT included 176 HBeAg positive patients which were treated with TDF, and 90 HBeAg positive patients treated with ADV. At 48 weeks, a higher proportion of patients in the TDF arm than in the ADV arm achieved HBV-DNA levels <400 copies/ml (76% vs 13%, p < 0.001). The respective rates for ALT normalization were 69% vs 54% (p = 0.03) and for HBeAg seroconversion were 21% vs 18% (p = 0.02). A higher proportion of patients treated with TDF had HBsAg loss (3% vs 0%, p = 0.02). The incidence of severe adverse events was similar in the TDF and ADV arms. A1c
In the second RCT, 375 HBeAg negative patients with chronic hepatitis were randomized to receive TDF 300 mg (250 patients) or ADV 10mg (125 patients) for 48 weeks. At week 48, a significantly higher proportion of patients treated with TDF achieved the primary endpoint, compared with patients treated with ADV (71% vs 49%, p < 0.001). At the end of treatment, 93% of the patients in the TDF group had HBV-DNA levels greater than 400 copies/ml, compared with 63% of patients in the ADV group (p < 0.001). The rates of ALT normalization were similar in both treatment groups. No patients treated with TDF had a confirmed 0.5 mg increase in serum creatinine level or creatinine clearance of <50ml/min. A1c
What are the long-term benefits of anti-viral therapy for chronic hepatitis B?
IFN therapy and HCC development
We reviewed the available literature to estimate whether IFN reduces the incidence of HCC and liver-related mortality in HBV-related cirrhosis. All potentially relevant papers were initially classified into two subsets (1: controlled trials; 2: cohort studies).
Subset 1: controlled trials The rate of HCC in treated and untreated patients with HBV cirrhosis was reported in 11 studies, including 2560 patients [92–102]. The benefit of IFN on cancer development is shown in Figure 27.4. IFN seemingly decreased the rate of HCC occurrence in all but one trial, and a significant difference was observed in three studies. The pooled estimate of the preventive effect of treatment was significantly in favor of IFN (rate difference: –0.04; 95% CI: –0.01~0.07). A remarkable heterogeneity was detected among studies (heterogeneity: 39.12 with 10df;p< 0.001).
Since the trials showed a significant inconsistency, subgroup analyses were performed in relation to the ethnic origin of patients (European versus Oriental studies). Consistent results were observed only when assessing data pooled from European reports; in this subgroup no preventive effect of HCC was found. A1c
Subset 2: cohort studies It has been suggested from long-term cohort studies that IFN treatment may have a protective effect against HCC development in patients with chronic HBV infection. We reviewed the clinical course and outcome of patients enrolled in long-term, follow-up studies [98,99,102,103–111]. The 12 studies (11 prospective and one retrospective) included a total of 1952 patients, 1187 of them not receiving active treatment. Length of follow-up ranged from 2.1 to 8.9 years. Meta-analysis of longitudinal studies with prolonged follow-up showed no significant differences in the rate of HCC between treated patients (1.9%; 95% CI: 0.8-3.0%) and controls (3.16%; 95% CI: 1.8–4.5%). A1c
All data dealing with HCC development coming from studies with prolonged follow-up must be interpreted with caution because possible biases can lead to erroneous estimates:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







