Fig. 21.1
Classification of fistula-in-ano. (a), Intersphincteric. (b), Transsphincteric. (c), Suprasphincteric. (d) Extrasphincteric (With permission from Vasilevsky [2])
Table 21.1
Classification of fistula-in-ano
Intersphincteric |
Simple low tract |
High blind tract |
High tract with rectal opening |
Rectal opening without perineal opening |
Extrarectal extension |
Secondary to pelvic disease |
Transsphincteric |
Uncomplicated |
High blind tract |
Suprasphincteric |
Uncomplicated |
High blind tract |
Extrasphincteric |
Secondary to anal fistula |
Secondary to trauma |
Secondary to anorectal disease |
Secondary to pelvic inflammation |
Evaluation and Workup
History
Key Concept: Most routine fistulas you can elicit on history alone. Use the history to tease out those that are more concerning for sources other than cryptoglandular.
A patient with a fistula-in-ano will often recount a history of an abscess that has been drained either surgically or spontaneously. Patients may complain of drainage, pain with defecation, bleeding due to the presence of granulation tissue at either opening, swelling, and/or decrease in pain with drainage. Additional bowel or systemic symptoms (i.e., abdominal pain, bloody diarrhea, weight loss, immunosuppression) may be present when the fistula is not cryptoglandular.
Physical Examination
Key Concept: Although you may not always readily find the internal opening on physical examination, you can often get a feel for the extent of the disease process and exclude other sources of pathology.
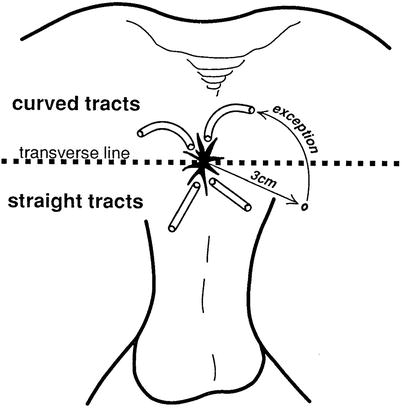
The external or secondary opening may be seen as an elevation of granulation tissue discharging pus. This may be elicited on digital rectal examination. In most cases, the internal or primary opening is not apparent. The number of external openings and their location may be helpful in identifying the primary opening. According to Goodsall’s rule (Fig. 21.2), an opening seen posterior to a line drawn transversely across the perineum will originate from an internal opening in the posterior midline. An anterior external opening will originate in the nearest crypt. Generally, the greater the distance from the anal margin, the greater the probability of a complicated upward extension [3]. Digital rectal examination may reveal an indurated cord-like structure beneath the skin in the direction of the internal opening with asymmetry between right and left sides. Internal openings may be felt as indurated nodules or pits leading to an indurated tract [3]. Look for posterior or lateral induration that may be palpable, as this may indicate fistulas deep in the postanal space or horseshoe fistulas [3, 4]. Bidigital rectal examination will define the relationship of the tract to the sphincter muscles and provides information as to preoperative sphincter tone, bulk, and voluntary squeeze pressure that need to be assessed.
Anoscopy is done prior to operation in an attempt to identify the primary opening, while sigmoidoscopy may locate a proximal internal opening and excludes underlying pathology such as proctitis or neoplasia. If you see either of these (or findings concerning for either), you need to redirect your workup and likely perform biopsies, cultures, etc., as appropriate. Colonoscopy or barium enema is indicated in patients with symptoms of inflammatory bowel disease, in patients with multiple or recurrent fistulas, and in patients who merit colorectal cancer screening. Anal manometry is normally not required, but may be useful in women with previous obstetric trauma, elderly patients, a patient with Crohn’s disease or AIDS or in a patient with a recurrent fistula [5]. Preoperative imaging is used infrequently, with options including fistulography, CT scanning, endoanal ultrasound, and MRI.
Imaging Studies
Key Concept: I will use imaging studies in recurrent fistulas occasionally when I think it may be helpful to demonstrate clinically undetected sepsis, to serve as a guide at the time of the initial surgery, to determine the relationship of the fistula tract to the sphincter mechanism, and to reveal the site of sepsis in a recurrent fistula.
Fistulography
Key Concept: Although fistulography may help in select cases, it is plagued by difficulties, is invasive, and may result in the dissemination of sepsis.
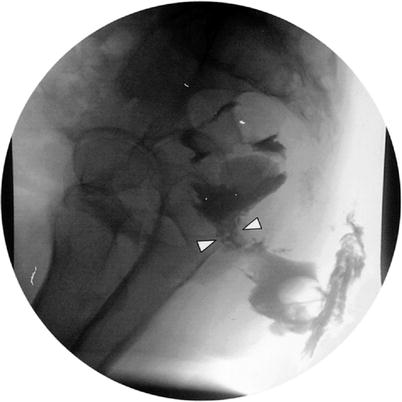
Fistulography, which involves cannulation of the external opening with a small feeding tube or catheter and injection of water soluble contrast, may be useful in the evaluation of recurrent fistulas or in Crohn’s disease where previous surgical forays or disease may have altered anorectal anatomy (Fig. 21.3) [6]. Contrast is introduced at low pressures to avoid tissue disruption and allow secondary tracts to fill. Localization of an associated abscess or the level of the internal opening may be difficult due to the absence of precise landmarks. Contrast may reflux into the rectum wrongly suggesting an extrasphincteric tract with a rectal opening. Accuracy rates in identifying the internal openings and extensions have ranged from 16 to 96 % with a false-positive rate of 12 % [6, 7].
CT Scan
Key Concept: CT is most helpful to look at surrounding structures rather than the fistula itself.
CT scanning performed with intravenous and rectal contrast is a less invasive method to assess the perirectal spaces and may differentiate an abscess from perirectal cellulitis [2]. It does not permit visualization of tracts in relation to the levators, but may be helpful in assessing the degree of rectal inflammation in patients with inflammatory bowel disease.
Endoanal Ultrasound
Key Concept: Ultrasound often can show you the presence and extent of the fistula, though should be interpreted by an experienced provider.
Endoanal ultrasound can establish the location of the primary opening and the relation of the primary tract to the anal sphincters, determine if the fistula is complex, and identify areas of suppuration [8]. A study conducted in 2002 using a 10 mHz probe along with injection of hydrogen peroxide was able to identify the internal opening in 93 % (Fig. 21.4) [8]. Ultrasound is rapid and well tolerated and can be transported to the operating room for challenging cases. Unfortunately, it is operator dependent, and scars or defects caused by previous sepsis, surgery, or trauma may confuse ultrasonographic interpretation [10].
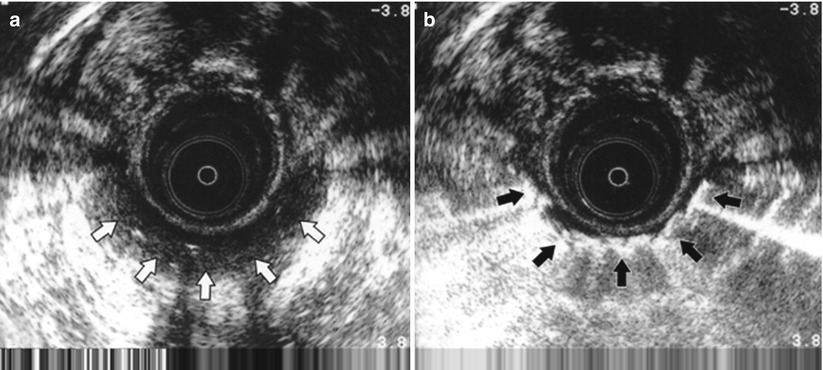

Fig. 21.4
(a) Anal endosonogram (b) with hydrogen peroxide. (Courtesy of Dr. Julio Faria) White arrows demonstrate unenhansed fistula tract. Black arrows highlight hydrogen peroxide enhansed fistula tract
MRI
Key Concept: MRI is becoming the preferred imaging technique in many centers for recurrent and complex fistulas.
MRI may be of value in the assessment of patients with complex fistulas and in those with anatomic distortion resulting from previous surgery (Fig. 21.5). MRI has been found to accurately delineate the site of the internal opening and the presence and course of primary and secondary fistulous tracts [11]. It may also significantly decrease recurrence rates in surgery for recurrent fistulas [12]. I find MRI to be useful in those cases that I feel require imaging and will often use it as a first modality.
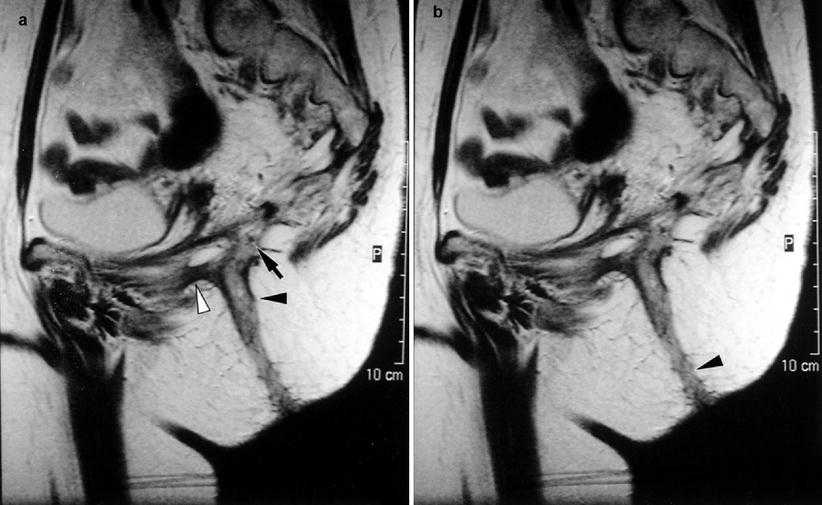

Fig. 21.5
Phase array MRI demonstrating the fistula opening (black arrow) and tract (black arrow head) (With permission from Vasilevsky [2]) White arrow demonstrates secondary entension
Treatment
General Principles
Key Concept: The principles of fistula surgery are to eliminate the fistula, prevent recurrence, and preserve sphincter function. Success is usually determined by identification of the primary opening and dividing the least amount of muscle possible.
Several techniques can help you identify the primary opening in the operating room:
1.
Passage of a probe or probes from the external to the internal opening or vice versa
2.
Injection via a catheter inserted into the fistula tract of contrast such as hydrogen peroxide (Fig. 21.6) or dilute methylene blue and noting their appearance at the dentate line
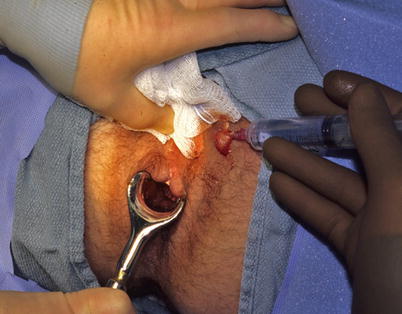

Fig. 21.6
Injection of dilute hydrogen peroxide to identify the internal opening (Courtesy of W. Brian Sweeney, MD)
3.
Following the granulation tissue present in the fistula tract
4.
Noting puckering of an anal crypt when traction is placed on the tract
Patients are educated on the challenges of fistula management and the different approaches available. Depending on the patient and their anatomy, success has varied from 60 to 90 %. In general, most surgeons prefer to accept a recurrence over injury to the patient or their sphincters. For most surgical procedures the patient is placed in the prone jackknife or left lateral (Sims’) position following induction of a general or regional anesthetic. Local anesthesia consisting of mixture of 0.5 % lidocaine or 0.25 % bupivacaine hydrochloride with 1:200,000 epinephrine is injected along the fistula tract for hemostasis following insertion of an anal speculum. The fistula is assessed to determine its location and relation to the sphincters using the techniques described above. Taking into account the characteristics of the fistula and the patient, several options are available.
Operative Management
Lay-Open Technique (Fistulotomy)
Key Concept: Fistulotomy provides the best success for low-lying fistulas, though you will have an open wound for a while that you need to follow-up on.
For simple intersphincteric and low transsphincteric fistulas, a probe is carefully inserted from the external opening through the tract to the internal opening at the dentate line (Fig. 21.7). The tissue overlying the probe is incised with electrocautery and any granulation tissue and the tract pseudoepithelium is curetted or fulgurated. Additional gentle probing is used to identify any high blind tracts or extensions, which are unroofed, if found. If you desire, the wound may be marsupialized on either edge by sewing the edges of the incision to the tract with a running absorbable suture. There is no need to insert packing if an adequate unroofing has been accomplished (Figs. 21.8 and 21.9).
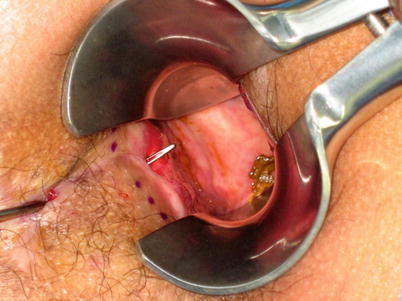
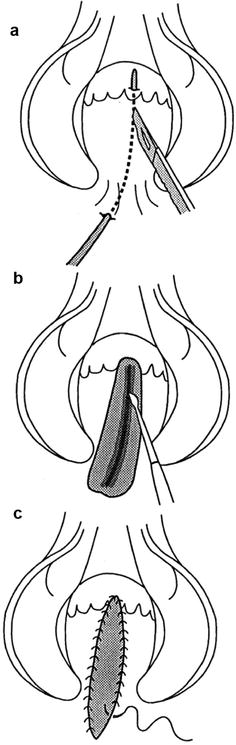
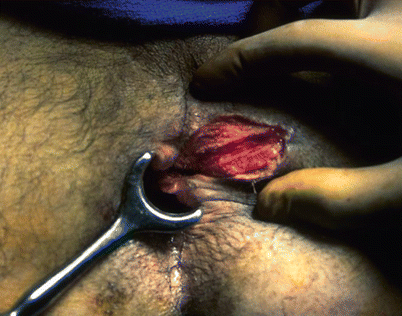

Fig. 21.7
Crypt probe running through the fistula tract

Fig. 21.8
Technique of laying open. (a) Insertion of probe and incision of tissue overlying probe. (b) Curettage of granulation tissue. (c) Marsupialization of wound edges (With permission from Vasilevsky [2])

Fig. 21.9
Fistulotomy (Courtesy of W. Brian Sweeney, MD)
Following the lay-open technique, patients are placed on regular diets, bulk agents, and non-codeine-containing analgesia. Patients are instructed to take frequent sitz baths to ensure perianal hygiene. I see them back in the office at 2-week intervals to ensure that healing has occurred from the depths of the tract. Granulation tissue can be cauterized using silver nitrate sticks, and cotton-tipped swabs are often used to probe the depths of the incision to ensure that adequate healing is occurring.
Fistulotomy has the highest success rate but leaves a wound that must heal by secondary intention. The amount of sphincter muscle that can be safely divided is a matter of surgical judgment that takes into account patient characteristics and the specifics of the fistula. In general, division of muscle distal to the dentate line will not have major alterations in continence. Suprasphincteric fistulas involve the entire external sphincter complex as well as the puborectalis muscle, and laying open the entire tract would render the patient incontinent. Thus other methods should be utilized.
Setons
Key Concept: Setons are great for allowing drainage, allowing the tract to fibrose, and preparing the fistula for a secondary procedure. Cutting setons aren’t used much anymore.
If the fistula tract is seen to cross the sphincter muscle at a high level, the insertion of a seton is an option. A seton may be any foreign substance that can be inserted into the fistula tract to encircle the sphincter muscles. Materials commonly employed include silk or other nonabsorbable suture material, Penrose drains, rubber bands, or Silastic vessel loops. Setons may be used in two fashions.
A draining seton is placed through the tract and left loosely in place to act as a drain to facilitate drainage and delineate the tract (Fig. 21.10). In complex disease, prolonged drainage allows resolution of sepsis and provides an opportunity for additional therapy. Placement of an initial draining seton has improved the success of subsequent procedures (e.g., the LIFT or suprasphincteric fistulae), as described later in this chapter.
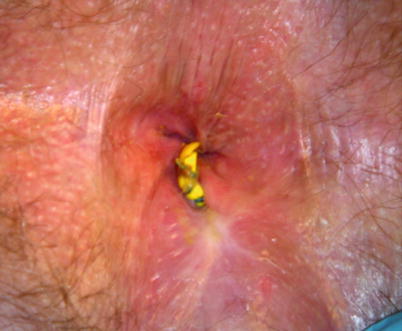

Fig. 21.10
Silastic vessel loop draining seton in place
Prior to the development of additional surgical options, when a fistula involved a significant portion of the sphincter mechanism, a cutting seton was considered. With this technique, the lower portion of the internal sphincter is divided along with the skin to reach the external opening and a nonabsorbable or elastic suture is inserted into the fistulous tract. The ends of the suture are tied with multiple knots to create a handle for manipulation (Figs. 21.11 and 21.12). The cutting seton was traditionally tightened at regular intervals to slowly cut through the sphincter. This allows the tract to become more superficial, converting a high fistula into a low one. The proximal fistulotomy subsequently heals by stimulating fibrosis behind it. This prevents separation or retraction of the sphincter muscle. The seton also allows delineation of the amount of remaining muscle that may be divided at a second operation several weeks later. The technique is very uncomfortable for the patient, and with the availability of other options, cutting setons are rarely used. A recent extensive literature search suggested abandoning the use of cutting setons because of sphincter damage and incontinence rates approaching 12 % [2].
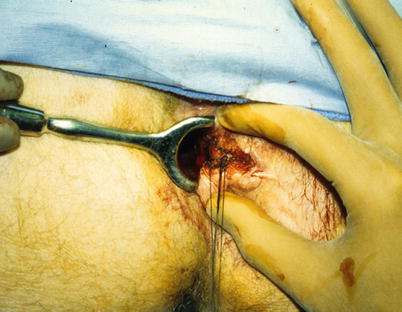

Fig. 21.12
Cutting seton in place (Courtesy of W. Brian Sweeney, MD)
Anorectal Advancement Flap
Key Concept: Anorectal advancement flaps provide a good option for difficult and recurrent fistulas. Ensure your flap is well vascularized by making it wide enough, including some of the underlying muscle and mobilize enough to make it tension-free.
The traditional laying-open technique is often inappropriate for anterior fistulas in women, in patients with inflammatory bowel disease, in patients with high transsphincteric and suprasphincteric fistulas, as well as in those with previous multiple sphincter operations and multiple and complex fistulas. For these patients, an anorectal advancement flap has been advocated (Figs. 21.13a–d and 21.14) [14]. Advantages of this technique include a reduction in the duration of healing, reduced associated discomfort, lack of deformity to the anal canal, as well as little potential additional damage to the sphincter muscles since no muscle is divided [2].
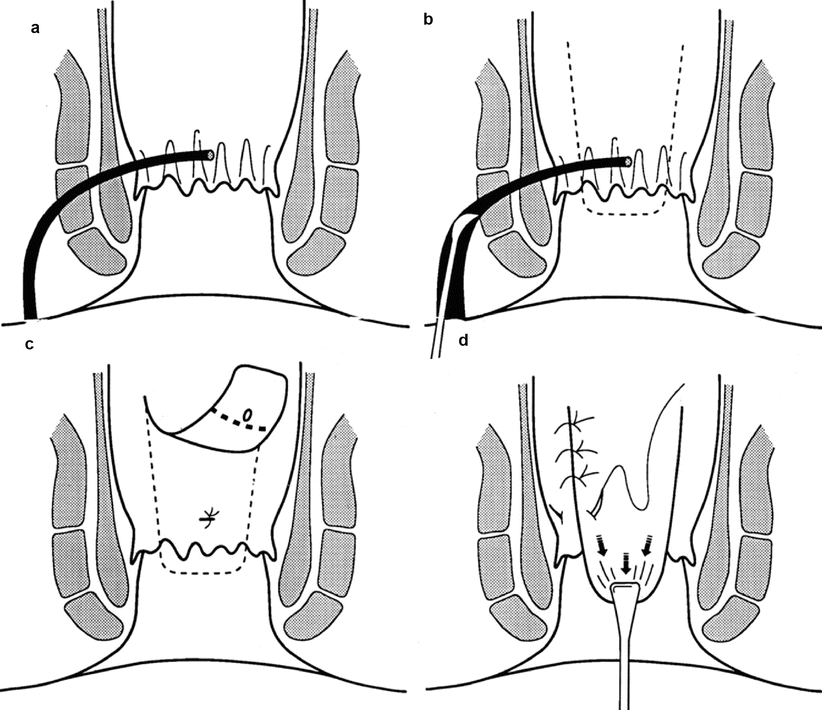
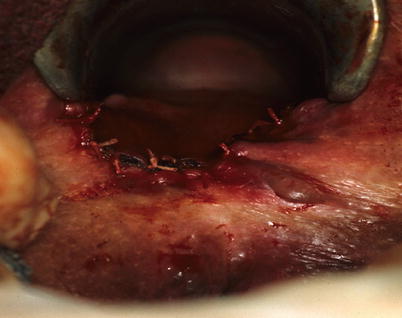

Fig. 21.13
Anorectal advancement flap. (a) Transsphincteric fistula-in-ano. (b) Enlargement of external opening and curettage of granulation tissue, (c) mobilization of flap and closure of internal opening, (d) suturing of flap in place covering internal opening (With permission from Vasilevsky [2])

Fig. 21.14
Completed anorectal advancement flap (Courtesy of W. Brian Sweeney, MD)
Following preparation and positioning, the fistula tract is identified with a probe and either cored out or curetted. The internal opening is identified and excised and the external opening is enlarged to allow for drainage. A full-thickness flap of rectal mucosa, submucosa, and part of the internal sphincter is raised. The residual internal opening is closed with absorbable suture. The flap is then advanced 1 cm below the internal opening. The tip of the flap containing the fistulous opening is excised, and the flap is sewn into place with absorbable sutures ensuring that the mucosal and muscular suture lines do not overlap. The base of the flap should be twice the width of the apex to maintain good blood supply. Successful results have reported in over 90 % of patients [15]. Factors associated with poor outcomes include Crohn’s disease and steroids [16]. The flap is a great option for recurrent or difficult fistula-in-ano, but you need to ensure your technique allows for great mobilization and preservation of the blood supply to optimize your success.
Fibrin Glue
Key Concept: With time and experience, fibrin glue has had low rates of incontinence but high rates of failure and recurrence.
The use of fibrin glue as a primary treatment alone or in combination with an advancement flap was appealing since it is a simple, noninvasive approach that avoids the risk of incontinence associated with fistulotomy. In the case of failure, it may be repeated several times without jeopardizing continence. As with fistulotomy, the fistula tract along with its internal and external openings is identified and curetted (with curettes or flexible brushes). Fibrin glue is injected into the fistula tract through a Y connector so that the entire tract is filled and the glue can be seen emerging from the internal opening (Fig. 21.15). The injecting catheter is slowly withdrawn so that the entire tract is filled (Fig. 21.16).
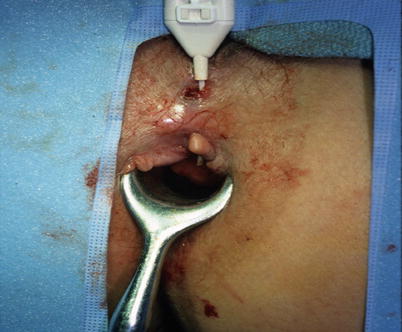
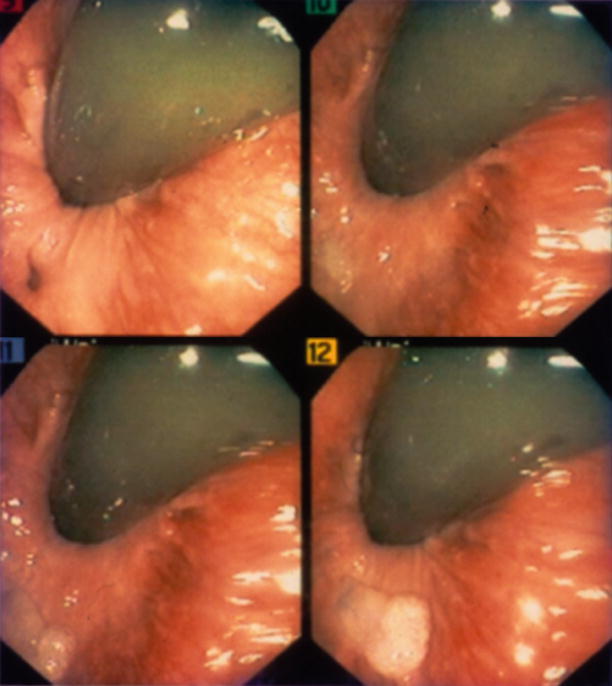

Fig. 21.15
Injection of fibrin glue down a fistula tract (Courtesy of W. Brian Sweeney, MD)

Fig. 21.16
Endoscopic view of fibrin glue filling the internal opening. The internal opening can be seen in the left upper picture as the dark area at ~7o’clock, with subsequent images showing the fibrin glue filling this opening (Courtesy of W. Brian Sweeney, MD)
Enthusiasm generated because of initial short-term success rates of 70–74 % has been tempered due to delayed fistula recurrence despite initial apparent healing [17–19]. Slightly better results were obtained with a 2-stage approach consisting of seton placement followed by glue injection at a second stage. Although the exact mechanisms responsible for failure have not been entirely appreciated, it has been suggested that curettage may not adequately remove all granulation or epithelialized tissue thus failing to provide the correct environment for the glue to work [20]. Other adverse factors shown to influence healing include the presence of a short tract which may make it easier for the fibrin glue plug to become dislodged as well as the presence of a cavity on endoanal ultrasound [21]. The latter was associated with a complication of perianal abscess since the tract may not have been entirely filled with glue [22]. Fibrin glue is associated with a low incontinence rate as well as a disappointing low cure rate and not used much anymore.
Anal Fistula Plug
Key Concept: Plugs provide an option for sphincter preservation, although recurrence and failure rates are 40–60 %.
Two bioprosthetic plugs are currently available to treat anal fistulas. The Biodesign® anal fistula plug (lyophilized porcine intestinal submucosal, Cook Biotech, West Lafayette, IN) and the Gore® Bio-A fistula plug (synthetic bioabsorbable, W L Gore, Flagstaff, AZ) provide a scaffold for colonized by host tissue cells, blood vessels, and connective tissue.
Following preparation and positioning, the internal and external openings are delineated. A probe is gently passed from the external to the internal opening to confirm the position of the tract and facilitate insertion of the plug. Debridement or curettage of the tract should not be performed. A seton should be used temporarily if there is acute inflammation or drainage. A 2-0 suture is placed through the tapered end of the plug and the ends of this suture are attached to the fistula probe at the primary opening. The suture is pulled from the primary opening, through the fistula tract to exit at the secondary opening. For patients with a “horseshoe” fistula, an incision may be made over the fistula tract distal to the anal verge to create a secondary opening that the ends of the suture are brought through. With gentle traction on the suture, the plug is pulled into the primary opening of the fistula until it is snug but not forced tightly. Excess plug is removed by transecting the plug at the level of the primary opening. The plug is secured in the primary opening using a 2-0 absorbable suture placed in a figure of 8 fashion with the suture crossing through the center of the plug and incorporating a generous portion of the sphincter mechanism on both sides. Any plug protruding through the secondary opening is also excised. The distal end of the plug is not sutured to the fistula tract and the distal opening is left open for drainage (Fig. 21.17). Patients are advised to avoid vigorous physical activity for 2 weeks after plug placement to minimize the chance of plug dislodgement. No dietary restrictions are necessary nor are topical antibiotics indicated.
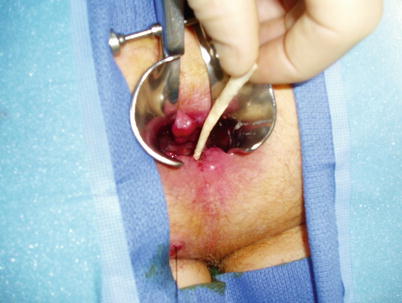

Fig. 21.17
Anal fistula plug (Courtesy of David Armstrong, MD, Atlanta, GA)
As with many of these alternative techniques, initial results were successful [23, 24]. Unfortunately, subsequent experience has been less successful with healing rates averaging 40–60 %. Contraindications for the use of the plug include fistulas with a persistent abscess cavity or infection, allergy to porcine products, and inability to identify both the external and internal openings. The latter constitutes an absolute contraindication for use of the plug. The plug is useful in certain situations of failure or recurrence or in people with borderline continence that you want to ensure no damage to the muscle occurs.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree