Fig. 1 a–e
Ovarian endometrioma. a Axial fast-spin-echo T2-weighted imaging (FSE T2-WI) shows unilocular right cystic ovarian lesion (asterisk) demonstrating diffuse hypointensity (i.e., shading effect) relating to chronic hemorrhage. b Sagittal and c coronal T2-WI show some fibrous adhesions in the pouch of Douglas (arrows). d Axial fat-suppressed (FS) T1-WI confirms presence of blood product inside the lesion. e Subtraction gadolinium-enhanced FS T1-WI demonstrates absence of mural solid nodules. These findings are suggestive of endometriosis
Dermoid Cysts (Mature Cystic Teratomas)
On US, dermoid cysts may demonstrate several characteristic features such as:
Fat fluid level
“White ball” within part of or all the lesion (representing dermoid plug consisting of hair and sebum)
“Tip of the iceberg” appearance, in which a diffuse or partly echogenic mass is seen with sound attenuation
“Dermoid mesh,” consisting of long, echogenic lines and dots within the cyst fluid, corresponding to hair floating within the cyst cavity.
Often, if shadowing is present, it may be confused with adjacent bowel. US features may overlap with other entities, such as endometriomas or ovarian carcinomas. On CT, they are recognized due to the presence of macroscopic fat and calcifications. On MRI, the fat component will demonstrate high SI on both T1- and T2-WI, with the lipid-laden cyst fluid demonstrating a similar high SI on T1- and intermediate SI on T2-WI. It is important to distinguish a dermoid cyst from an endometrioma, as both can show high SI on T1-WI. Chemical-shift artefact will be seen at the fat-fluid interface in dermoid cysts as bright or dark bands along the frequency-encoding gradient. Use of frequency-selective FS enables dermoid cysts (which appear as low SI) to be distinguished from hemorrhagic lesions or endometriomas (which retain high SI) (Fig. 2). Rarely, a cancer may develop within a dermoid cyst, most frequently a squamous cell cancer.
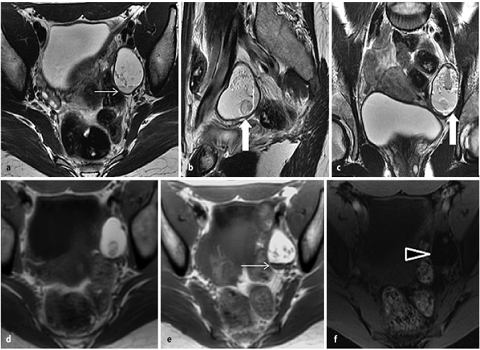

Fig. 2 a–f
Mature ovarian cystic teratoma. a Axial, b sagittal, and c coronal fast-spin-echo T2-weighted images (FSE T2-WI) show high T2- signal-intensity unilocular left cystic ovarian lesion, with hypointense mural protrusions (i.e., dermoid plug; large arrow in b and c). d, e On axial T1-WI, lesion demonstrates high signal intensity characterized signal drop on f fat-saturated (FS) T1-WI (arrowhead) due to lipid content, with evidence of fluid-lipid level (thin arrow in a and e)
Serous and Mucinous Cystadenomas
Serous and mucinous cystadenomas are classified as surface epithelial tumors. Serous cystadenomas are classically described on US as thin-walled unilocular or bilocular cystic lesions, with a homogenous echotexture, thin regular septa (when present), and no vegetations (Fig. 3). Unlike functional cysts, cystadenomas will persist unchanged over one or two menstrual cycles and may increase in size. In comparison with serous cystadenomas, mucinous cystadenomas are more often multilocular, containing fluid of different echotextures. As with serous cystadenoma, they have a smooth wall, thin septa (<3 mm) and no vegetations. On MRI, cysts in mucinous tumors demonstrate various signal intensities on T1- and T2-WI, giving rise to the so-called “stained-glass” appearance. This is because on T1-WI locules with watery mucin generate lower SI than locules with thicker mucin. The opposite is generated on T2-WI.
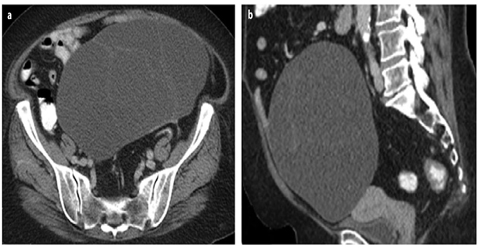

Fig. 3 a, b
Serous ovarian cystoadenoma. a Axial and b sagittal contrast-enhanced computed tomography (CE-CT) images show cystic pelvic mass with smooth margin and internal thin, regular septa, without evidence of solid mural enhancing nodules
Benign Solid Ovarian Tumors
These tumors include ovarian fibromas, thecomas, adenofibromas, and Brenner tumors. Fibrotic tumors account for approximately 5% of all ovarian tumors.
Fibromas/Fibrothecomas
They are the most common solid benign tumors of the ovary. Fibromas and fibrothecomas may mimic pedunculated myomas on US, appearing as well-circumscribed, hypoechoic, and solid round or oval tumors with sound attenuation. On color Doppler, they are usually hypovascular. Fibromas are sometimes associated with ascites and pleural effusion (Meigs syndrome). On MRI, due to the extensive collagen content and hyalinized tissue, fibromas and fibrotic component of fibrothecomas appear as intermediate SI masses on T1- and low SI masses on T2- WI. There may be scattered foci of high SI on T2-WI due to cystic degeneration or edema. Calcification may occur, seen as foci of signal voids on T1- and T2-WI. After administration of gadolinium, there is typically delayed lowlevel enhancement. Thecomas contain a prominent lipid component that can be demonstrated on chemical-shift imaging or on frequency-selective FS-MRI. Due to estrogen secretion, endometrial hyperplasia may be present.
Brenner Tumors
Brenner tumors are transitional cell tumors of the ovary. The majority is benign, but borderline and malignant types can also arise. Benign tumors are usually unilateral, small (<5 cm), and hypoechoic on US. Extensive calcification is characteristic, resulting in posterior acoustic shadowing on US. On MRI, the benign type appears solid and of low SI on both T1- and T2-WI. They demonstrate lower SI on T2-WI than other solid tumors. Ipsilateral ovarian tumors (e.g., mucinous cystadenomas) may be present in 30% of cases, and endometrial hyperplasia can also occur.
Borderline and Malignant Ovarian Tumors
Tumors such as these include surface epithelial tumors (serous cystadenocarcinomas, mucinous cystadenocarcinomas, endometrioid, and clear-cell carcinomas), sex cord-stromal tumors (granulosa cell and Sertoli-Leydig cell tumors), and germ cell tumors. Less common malignant tumors are mixed Müllerian tumors, undifferentiated carcinomas, carcinoids, and lymphomas.
Borderline serous tumors account for 10–15% of all ovarian serous tumors and are bilateral in 30% of cases. Likelihood of malignancy increases with the presence of septations, mural nodules, and papillary projections (Fig. 4). MRI features suspicious for malignancy include: mass >4 cm, cystic lesion with solid component, irregular wall thickness >3 mm, septal thickness >3 mm, presence of papillary projections or nodules, and presence of solid mass with necrosis and early, bright contrast enhancement [23, 24]. In addition, ancillary criteria for suspected malignancy include: direct local invasion, peritoneal deposits, enlarged lymph nodes, and ascites. DW-MRI is highly sensitive and specific in detecting the extent of peritoneal disease. CE-CT remains the technique of choice for staging ovarian carcinoma (Fig. 5).
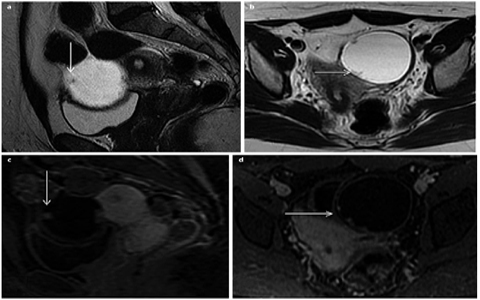

Fig. 4 a–d




Serous borderline ovarian tumor. a Sagittal and b axial fast-spin-echo T2-weighted image (FSE T2-WI) show unilocular, thinwalled cystic lesion, with intermediate signal intensity T2 papillary projections (arrows in a and b). c, d Sagittal and axial gadoliniumenhanced fat-saturated (FS) T1-WI confirm the presence of solid papillary projections, characterized by contrast enhancement
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








