Fig. 14.1
Renal biopsy showing a large cell with CMV inclusions and surrounding interstitial inflammation (H&E, 60×)
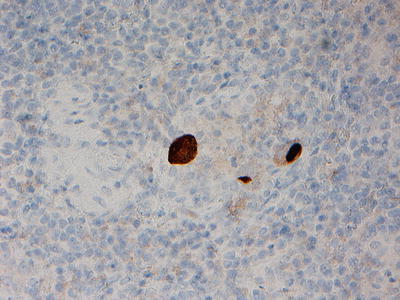
Fig. 14.2
Immunohistochemistry (immunoperoxidase staining) showing CMV-infected cells
Other Immunocompromised Hosts
Besides renal transplant recipients, CMV-associated tubulointerstitial nephritis has been reported in an allogeneic stem cell transplant patients. These patients typically present with fever and hematuria, and kidney biopsy usually reveals focal necrosis of the tubules, with acute and chronic inflammatory infiltrates around the tubules. Intracellular inclusions can be seen in the tubular cells, and immunofluorescent staining for CMV antigens is frequently positive in the degenerating tubular epithelium [21] (Figs. 14.1 and 14.2). However, the frequency of CMV nephritis in stem cell transplant recipients, even in the setting of active CMV infection, is unlikely to be high [18]. In an autopsy series of 33 immunocompromised patients with manifest CMV infection at the time of death [19], CMV was detected in the kidney in only eight patients. Moreover, characteristic “owl’s eyes” cells were present in three cases only. All the patients in this study had evidence of CMV involvement of other organs aside from the kidney.
Treatment and Outcomes
The prognosis of CMV nephritis depends on multiple factors, including but not limited to the immune competence of the host, the presence of preexisting kidney disease, concomitant CMV disease involving other organs, and whether the infection is in a native kidney or renal allograft. It is well known that CMV disease, even if outside of the kidney in renal transplant recipients, is associated with an increased risk of allograft rejection. Moreover, active CMV disease is associated with increased risk of other opportunistic infections. Some renal transplant recipients with CMV-infected allografts may eventually lose all allograft function. Besides, CMV infection of the renal allograft may be accompanied by allograft vasculopathy.
Based on above considerations, we believe it is prudent to treat CMV infection of the kidney similar to other end-organ CMV disease in the immunocompromised hosts. The cornerstone of CMV treatment is antiviral agents with good anti-CMV activity, including ganciclovir, valganciclovir (the valyl ester prodrug of ganciclovir), cidofovir, and foscarnet [22]. However, drug-induced bone marrow suppression and nephrotoxicity are major limiting factors in using these antiviral agents in CMV disease. Therefore, CMV nephritis is best managed in consultation with an infectious diseases specialist.
Treatment benefit of CMV therapy in immunocompetent hosts is questionable. In the few reported cases of CMV-associated interstitial nephritis in immunocompetent hosts, the renal dysfunction is usually self-limited. Therefore, antiviral agents are usually not prescribed and are unlikely to be beneficial in the majority of these cases.
BK Virus
BK is a small (30–45 mm), nonenveloped, double-stranded DNA polyomavirus belonging to Papovaviridae virus family. BK virus was first isolated from the urine of a kidney transplant recipient in 1971 [23]. Polyomaviruses are ubiquitous and primary infection is usually acquired in the early childhood or adolescence via oral or respiratory exposure. Majority of the primary infections are asymptomatic in children. However, mild upper respiratory tract infection symptoms have been reported in up to 30 % of the immunocompetent patients with primary BK virus infection. By adulthood, 60–80 % of the population in the USA is seropositive for BK virus. However, BK virus can remain latent in lymphoid cells, renal tubular cells, and epithelial cells and can reactivate in immunocompromised host to cause symptomatic disease.
BK virus has tropism for urinary epithelium and has been implicated in three distinct genitourinary diseases in immunocompromised patients [24]. These include tubulointerstitial nephritis, ureteral stenosis, and hemorrhagic cystitis. While tubulointerstitial nephritis and ureteral stenosis are mostly seen in renal transplant recipients, hemorrhagic cystitis is typically described in bone marrow transplant patients. Non-renal infections attributed to BK virus in immunocompromised hosts include rare cases of hepatitis, pneumonitis, meningitis, vasculitis, and retinitis [25].
Risk of BK virus reactivation and symptomatic disease depends on degree of immunosuppression [26]. Patients with allogenic stem cell transplant are higher risk of reactivation disease and hemorrhagic cystitis compared to patients with autologous stem cell transplant (50 % vs. 15 %). Reported rate of BK nephritis in kidney transplant patients ranges from 1 to 10 % [27]. Infection of native kidneys in patients with other organ transplants, such as liver or lung, is rare.
Risk factors for BK virus nephropathy [28] are summarized in Table 14.1.
Table 14.1
Risk factors for BK virus nephropathy
Age (young children or elderly) |
Male gender |
White ethnicity |
Diabetes mellitus |
Degree of immunosuppression (allogenic stem cell transplant recipients are at higher risk compare to patients with autologous bone marrow transplant. Similarly, patients with kidney transplant who are receiving combination therapy with multiple agents such as mycophenolate mofetil, tacrolimus, cyclosporine, or azathioprine are at higher risk) |
Ureteral trauma |
Organ donor with BK virus seropositivity |
Absence of HLA-C7 in donor and recipient |
JC virus, another DNA polyomavirus belonging to Papovaviridae virus family, can cause a similar illness in immunocompromised patients [29]. However, chronic inflammation and fibrosis are more pronounced in renal disease due to JC virus [30].
Clinical Manifestations
The clinical presentation of BK virus reactivation disease involving kidneys is nonspecific. Typically patients with BK allograft infection present 10–13 months after transplant (range 6 days to 5 years) [31]. Asymptomatic viruria and viremia usually precede symptomatic BK nephropathy. Patients usually present with fever and subacute renal failure due to interstitial nephritis. BK virus is clinically indistinguishable from allograft rejection. BK virus related hemorrhagic cystitis in bone marrow transplant patients usually presents with fever and microscopic or macroscopic hematuria.
Diagnosis
Differentiating BK virus nephropathy from allograft rejection in kidney transplant patients can be quite difficult. While ureteral stenosis can be identified on ultrasound or computed tomography (CT) imaging, the diagnosis of interstitial nephritis requires a combination of urinalysis, molecular testing, and frequently renal biopsy.
Seropositivity to BK virus is very prevalent in the general population, and therefore, serology is not helpful in establishing the diagnosis of reactivation disease in transplant patients [32]. However, seropositive individuals tend to have less severe disease [33].
Urinalysis typically reveals pyuria, microscopic hematuria, cellular casts, and renal tubular cells, suggestive of interstitial nephritis. Cytology may be helpful in detecting viral shedding in the urine. Decoy cells, termed due to their resemblance to renal carcinoma cells, may be seen on urine microscopy [31]. These are characterized by enlarged nucleus with a single large basophilic intranuclear inclusion. Although decoy cells may be observed with cytomegalovirus (CMV) infection, viral inclusions are cytoplasmic and not intranuclear in CMV-infected cells [34]. Presence of white blood cells in urine and >10/cytospin decoy cells has high correlation (up to 70 %) with biopsy-proven BK interstitial nephritis [35].
Urine electron microscopy (EM) may reveal cast-like three-dimensional polyomavirus aggregates, termed Haufen. Presence of Haufen has high correlation with BK virus nephropathy (99 %) [36]. Although EM may help to differentiate between asymptomatic viral shedding and active BK infection, it is not readily available at most centers, is labor intensive, and needs experienced eyes for appropriate interpretation.
Polymerase chain reaction (PCR) testing for BK virus in plasma and urine is perhaps the most helpful molecular diagnostic tool available in routine clinical practice. Prolonged and high-grade viremia usually precedes and has good correlation with BK virus nephropathy. Therefore, plasma PCR to detect BK viremia has high sensitivity (100 %) and specificity (88 %) with renal disease [37]. With 98 % negative predictive value, absence of sustained DNAemia makes BK nephropathy highly unlikely [38].
Quantitative blood PCR is also useful for monitoring response to therapy in transplant patients. However, it should be noted that 1–2 log change in viral load is not significant and trends, rather than single values, are important in making critical treatment decisions. PCR on kidney biopsy specimens is not helpful because it can detect latent virus in asymptotic individuals and is not helpful in making treatment decisions [39].
Considering that most patients with BK nephritis have viral shedding in the urine, one practical approach to make the diagnosis is to start from BK virus PCR from urine sample. If urine PCR reveals >10 [7] cop/mL, one should proceed with plasma testing. In patients with blood BK viral load of >10 [4] cop/mL and evidence of allograft dysfunction, it is reasonable to make a presumptive diagnosis of BK virus nephritis and treat accordingly. However, renal biopsy is frequently needed to accurately differentiate between BK nephritis and allograft rejection [40].
Viral cultures are cumbersome and only performed in research setting. Their role in clinical practice is yet unproven [41].
Histologic features of BK nephropathy are quite variable and range from minimal changes to patchy tubulointerstitial inflammation and graft sclerosis [42]. Although BK nephritis has certain characteristic features (Table 14.2) [24, 26, 35, 40, 42–44], these are not pathognomonic and can be found in other viral infections such as adenovirus, CMV, and HSV. However, location of viral inclusions may provide important diagnostic clues towards viral etiology of underling nephritis. While cells infected with BK virus have intranuclear inclusions, CMV-infected cells have cytoplasmic and HSV results in both intranuclear and cytoplasmic inclusions (Fig. 14.3).
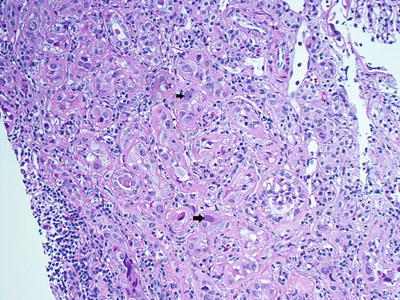
Table 14.2
Characteristic histologic features of BK virus nephropathy
Interstitial, mononuclear or neutrophilic, infiltrate at the site of infection |
Evidence of tubulitis with lymphocyte permeation of tubular basement membrane. Other features indicative of tubular injury include tubular cell apoptosis, cell dropout, desquamation, and flattened epithelial lining |
Variation in nuclear size (anisonucleosis), hyperchromasia, and chromatic clumping of infected cells |
Intranuclear basophilic viral inclusions without a surrounding halo |
Electron microscopy may reveal BK viral particles |

Fig. 14.3
Renal biopsy revealing BK virus (black arrows)-infected cells (H&E, 40×)
Both BK virus and allograft rejection can cause severe tubular inflammation. In general, the presence of endarteritis, fibrinoid vascular necrosis, glomerulonephritis, and C4d deposits along peritubular capillaries is more suggestive of allograft rejection [26]. However, vasculopathy has been occasionally reported with BK infection and does not exclude BK nephritis.
Due to focal nature of the disease, diagnosis of BK nephropathy may be missed in one-third of renal biopsy specimens. Therefore, examination of two core biopsy samples (including medulla) may help to increase the yield. In situ hybridization for BK virus can improve the diagnostic yield of renal biopsy (Fig. 14.4). Repeat biopsy may be needed where pathologic findings on first biopsy are inconclusive.
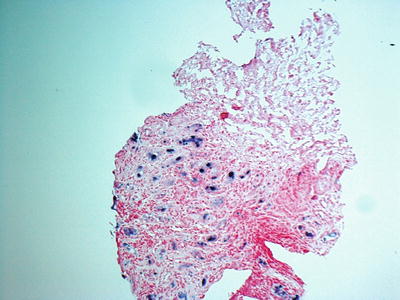

Fig. 14.4
Renal biopsy with in situ hybridization for BK virus showing infected cells (40×)
Treatment
BK nephritis is associated with significant morbidity and leads to allograft failure in 15–50 % of the infected patients [28, 31]. The key to appropriate management of BK nephritis is to accurately differentiate between allograft rejection and BK nephritis, as treatment is diametrically opposite. While increasing immunosuppression is the warranted in acute rejection, reduction in immunosuppressive therapy is the cornerstone of BK nephritis.
Treatment of BK virus nephritis in renal transplant patients can be approached in two different ways [45, 46]. First option is periodic monitoring for BK viremia and preemptive treatment based on rising BK viral load. Optimal interval and duration of screening is unclear but checking BK viral load every 3 months for the first 2 years after renal transplant may be a reasonable approach [26]. Some experts recommend closer monitoring (monthly BK-PCR) for the first 3 months as majority of the patients with BK renal disease develop viremia in the first 3–4 months after renal transplant [47]. The alternative approach is to reserve therapy for patients who develop allograft dysfunction and have biopsy-proven BK nephritis. There is some evidence to suggest that periodic surveillance and preemptive treatment may be a more effective approach [48].
Treatment of BK nephritis includes (a) reduction in immunosuppression, (b) use of intravenous immunoglobulins (IVIG), and (c) antiviral drugs. In patients who have increasing level of BK viremia without allograft dysfunction, reduction in immunosuppression alone may be adequate. This usually involves stopping the antimetabolic agent (such as azathioprine or mycophenolate mofetil) and reducing the dose of calcineurin inhibitor (cyclosporine). However, for patients who have biopsy-proven BK nephritis that is unresponsive to maximal reduction in immunosuppressive therapy, antiviral therapy may be necessary [47].
Unfortunately, antiviral drugs have limited activity and efficacy against BK virus. Drugs with known activity against BK virus include quinolones, leflunomide, and cidofovir [49]. Quinolone therapy has been shown to reduce BK viremia in transplant patients [50]. Considering that quinolones are fairly nontoxic, these should be tried first, with or without IVIG [51]. Pooled IVIG have neutralizing antibodies against BK virus and can be helpful, especially in patients who have hypogammaglobulinemia. Quinolones and IVIG can be used in combination. Use of IVIG therapy has the additional benefit of treating concomitant allograft rejection [52]. This is particularly important in cases where it may be difficult to differentiate between cellular rejection and BK nephritis.
Antiviral drug, leflunomide, inhibits BK virus replication in vitro and had limited success in the treatment of BK nephritis in a small study in transplant patients [53]. However, more data is needed before it can be used in routine clinical practice. Cidofovir, approved for CMV retinitis, is also active against BK virus. There is limited data to support its benefit in BK nephritis with improved allograft survival [54]. However, cidofovir is quite nephrotoxic [55] and its use should be limited to refractory cases. In cases where treatment is strongly indicated, use of lower doses (1 mg/kg every other day or three times per week) instead of standard dose of 5 mg/kg weekly should be considered to reduce risk of nephrotoxicity. Patients should be aggressively hydrated before and after each infusion. (Please see the section on treatment of adenoviruses for details.)
Hantavirus
Hantavirus is an enveloped, negative-sense, RNA virus that belongs to the Bunyaviridae family of viruses. Twenty species of hantavirus exist in nature and 11 have been reported to cause disease in humans. Among these, most human infections are cause by Hantaan, Puumala, Seoul, Dobrava, Sin Nombre, and Andes viruses.
Hantaviruses have worldwide distribution. Humans usually acquire hantavirus infection by inhalation or contact with secretions, urine or feces, of infected rodents [56]. Activities that may predispose to such exposures include farming, forestry, animal trapping, military activity, crisis conditions, and camping. Mice and rats are the most commonly implicated rodents in human cases. In the USA, most cases are reported from the southwestern states including New Mexico, California, Washington, and Texas and are due to Sin Nombre virus, which is usually associated with cardiopulmonary disease [57]. However, cases of hemorrhagic fever with renal syndrome have also been reported in the USA [58]. Epidemics of hantavirus infection from laboratory exposure have also been reported.
Clinical Manifestations
Typical incubation period of hantaviruses ranges from 2 to 4 weeks. Fever, malaise, and abdominal or back pain are the first signs of illness in most cases. However, infection can rapidly progress from fever to hypotensive shock and acute renal failure. Severity of illness partly depends on the causative species of hantavirus [59]. Severe renal disease is mostly caused by Hantaan and Dobrava viruses [60]. This was previously termed as nephropathia epidemica, epidemic hemorrhagic fever, and hemorrhagic nephrosonephritis. However, these cases are now collectively referred to as hemorrhagic fever with renal syndrome or HFRS based on World Health Organization (WHO) classification [56]. Puumala virus related renal disease is usually mild and carries good long-term prognosis [61].
Sin Nombre virus, the most common species of hantavirus in the USA, usually begins with fever, flushing, and pharyngitis but can quickly progress to acute respiratory distress and heart failure [62]. Respiratory failure in acute hantavirus infection is caused by noncardiogenic pulmonary edema due to diffuse capillary leak syndrome. Severe disease with this viral strain is termed hantavirus-related cardiopulmonary syndrome (HCPS). However, organ involvement is not limited to heart or lungs and renal dysfunction is frequently reported. Low-grade proteinuria and hematuria are found in up to 50 % of the cases and are usually associated with moderate increase in serum creatinine value [63]. Severe renal disease is less common with this particular species of hantavirus.
The clinical presentation of severe hantavirus infection closely mimics septic shock with fever, hypotension, and acute renal failure. These manifestations are primarily caused by increased vascular permeability leading to decreased peripheral vascular resistance and increased cardiac output [64]. As a result, there is poor renal perfusion and abrupt decline in glomerular filtration rate (GFR). Disseminated intravascular coagulation (DIC) is a complication of severe hantavirus infection and manifests as hemorrhage, petechiae, ecchymoses, hemoptysis, hematemesis, and melena.
Besides poor perfusion, hantavirus infection may also result in direct renal injury. Tubular and interstitial damage can be caused by cytokines and other humoral factors including tumor necrosis factor (TNF) [65]. Renal manifestations of hantavirus infection may vary based on genetic predisposition. Published data suggest that HLA-B8 and DR3 alleles are associated with severe renal failure [66], frequently requiring hemodialysis, where patients with HLA-B27 are more likely to have milder disease [67].
Differential diagnosis for hantavirus-related renal disease is broad and includes leptospirosis, renal failure to nonsteroidal anti-inflammatory drugs (NSAIDs), and vasculitis (Wegener’s granulomatosis, Goodpasture’s disease, etc.).
Diagnosis
Laboratory findings in acute hantavirus infection are quite nonspecific and may include leukocytosis, thrombocytopenia, high C-reactive protein (CRP), increased LDH, elevated liver enzymes, and elevation in serum creatinine [68]. Chest X-ray may reveal pleural effusion, atelectasis, and interstitial infiltrates in patients with HCPS. Abnormal EKG and echocardiographic findings are frequently reported in severe cases [69, 70]. Abdominal ultrasound may reveal an increase in the length of kidneys, restrictive indices, and peritoneal fluid collection [71].
Urinalysis typically reveals marked proteinuria (mean 2.6 g/day) [72]. Up to 25 % of the patients may present with nephrotic-range proteinuria. Although microscopic hematuria is present in 50–85 % of the cases [73], depending on the infecting strain, macroscopic hematuria is uncommon and is usually seen in the setting of severe thrombocytopenia.
Diagnosis is usually confirmed by serologic testing. Available serologic tests include enzyme-linked immunosorbent assay (ELISA), strip immunoblast test (SIA), Western blot, indirect immunofluorescence (IFA), complement fixation, hemagglutinin inhibition, and plaque reduction neutralization assays. IgM antibodies, to nucleocapsid or N antigen, are usually positive by the time of admission [74]. Many patients will also have positive IgG at the time of initial presentation. Acute infection can be confirmed by either presence of IgM antibodies or four field rise in IgG titer in convalescent sample 3–4 weeks later.
Sin Nombre virus can also be detected by RT-PCR (reverse transcription-polymerase chain reaction) using peripheral blood mononuclear cells (PMBC) or serum during early stages [75]. However, viremia is typically short lived and a negative test does not exclude the diagnosis.
Kidney biopsy is generally not necessary in making the diagnosis of acute hantavirus infection. Majority of reports describing histopathologic findings in acute hantavirus infection are from autopsy series. Typical histologic features include tubulointerstitial nephritis with mononuclear cells and CD8 lymphocytes infiltration. Congestion and dilatation of the medullary vessels is frequently observed [76]. Hantavirus typically localizes in the glomerular capillary endothelium, and its presence in the renal tissues can be confirmed by immunohistochemistry using antibodies to viral N antigen [77]. The N antigen stains in punctate pattern in the cytoplasm. Nested RT-PCR is another methods to detect the virus in frozen or fixed, paraffin-embedded tissues [78].
Treatment
Treatment for hantavirus infection is mostly supportive care. Platelet transfusion may be necessary in severe hemorrhagic shock. Also, patients with several renal failure may require hemodialysis. NSAIDs should be avoided as they can exacerbate the renal injury. Patient should be closely monitored in critical care for electrolyte imbalance, heart failure, and risk of cardiac arrhythmias. With improvements in supportive care, mortality in HFRS has reduced from 15 % in 1960s to <5 % in developed countries.
There is no proven, effective, and specific antiviral therapy for hantavirus infection. Although intravenous ribavirin use has been studied in the clinical trials, results have been variable and are not considered the standard of care at present [79].
Prognosis
The severity of hantavirus-related illness at initial presentation does not predict long-term sequelae [80]. The majority of the patients who survive the acute phase recover completely, and renal function returns to the baseline over time [61]. Acute infection is frequently followed by a diuretic phase that may last for 10–14 days. Thereafter, convalescent period may last for several weeks. However, a minority of patients may develop chronic renal insufficiency [81].
Adenovirus
Adenoviruses are medium-sized (90–100 nm), nonenveloped viruses that belong to family Adenoviridae. Adenoviruses were first isolated in 1953 from human adenoids (hence the name). These viruses contain a linear, non-segmented, double-stranded DNA genome.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







