Grade
Change in clinical management
A
No or minimal
B
Radiographic or endoscopic intervention or Grade A for > 1 week
C
Operative intervention
Controlled and Uncontrolled Biliary Leaks
A controlled biliary leak occurs when there is no communication with an intraperitoneal collection. In contrast, an uncontrolled biliary leak communicates with an intraperitoneal collection or flows freely throughout the peritoneal cavity. An uncontrolled biliary leak generally requires further drainage or manipulation of extant drains to establish control and prevent secondary infection.
Source
The source of the biliary leak can either arise from an incomplete division or disrupted side branch of the involved duct that is contiguous or communicates with that duct distally or a complete division of the involved duct that becomes discontiguous and does not communicate with that duct distally. The former sources are likely to resolve without operative intervention while the latter typically require such intervention. Discontiguous ductal injuries from an entire liver segment or more that lack communication with the central biliary tree are also termed excluded or orphan leaks [3]. These biliary leaks generally persist because the parenchyma harboring that duct maintains its vascularity. A classic example of this biliary leak would be a divided right segmental or sectional biliary duct after laparoscopic cholecystectomy. Regardless, the source of the biliary leak has implications on its diagnosis and management.
Risk Factors and Prevention
Risk factors and prevention of biliary leaks from the extrahepatic biliary system during laparoscopic cholecystectomy and bilioenteric anastomoses will be followed by discussion of bile leaks following hepatic resection.
Biliary leaks after laparoscopic cholecystectomy is estimated at 0.3–0.5 % [4]. Given the large number of cholecystectomies performed annually, this operation is associated most commonly with biliary leaks. The cystic duct stump, ducts of Luschka, other ducts in the gallbladder fossa, and major extrahepatic bile ducts comprise the potential sites of biliary leakage. The cystic duct stump represents the most common site of leak and reported risk factors include emergency surgery, incomplete or disrupted closure of the cystic duct, width and degree of inflammation of the cystic duct, and presence of common bile duct stones [5, 6]. The risk factors for biliary leaks related to major bile duct injury and classification of biliary injury following cholecystectomy have been reported previously and a complete discussion is beyond the scope of this chapter [7]. Commonly cited factors include operator inexperience and technical errors, inflammation, and most importantly anatomic misidentification of the extrahepatic bile duct.
Prevention
To prevent cystic duct stump leaks, one must ensure the cystic duct stump is appropriately identified and secured. A variety of methods of securing the cystic duct stump have been described. These include use of titanium clips, locking clips, harmonic scalpel, suture ligature, and endovascular stapling devices. There is no evidence-based data to suggest that any one method is vastly superior. The technique used should ensure that the clip or device is securely fastened, will not be dislodged, and should span the entire cystic duct. The cystic duct wall must be vascularized and free of significant inflammation. For a wide cystic duct, additional clips, ligature, or an endovascular stapler may be used after confirming the anatomy. Cautery injury proximal to the site of ligation should be avoided by careful dissection and use of cautery. Gentle traction should be used to avoid avulsion of the cystic duct from the common hepatic duct. Leakage from ducts of Luschka or from the fossa is likely related to dissection into the liver parenchyma. Efforts to stay in the correct plane and ligation of accessory ducts entering the gallbladder from the fossa should decrease peripheral biliary leaks. Preventing biliary leaks from major bile duct injuries requires correct identification of the anatomy. The critical view of safety in which the cystic duct and cystic artery are isolated and the cystic plate is exposed has been shown to decrease the rate of major biliary injuries [8]. The role of routine cholangiography to prevent injury is controversial [9]. Clearly cholangiography can define anatomy and unsuspected injury intraoperatively. Selective intraoperative cholangiography based on operative conditions and lack of anatomic clarity is utilized most frequently but such use does not address misidentification errors.
Risk Factors for Bile Leaks After Extrahepatic Bilioenteric Anastomosis
Excluding the transplant population, few studies have investigated risk factors for biliary leaks after bilioenteric anastomoses. Biliary reconstruction to the small bowel is undertaken through Roux-en-Y hepaticojejunostomy or hepatico- or choledochoduodenostomy. Typically, Roux-en-Y hepaticojejunostomy is favored because anastomotic leaks actually represent a pure biliary fistula as reflux of enteric content through the fistula is rare with appropriate length of construction of the Roux limb. In contrast, biliary anastomoses to the duodenum are not pure biliary fistulae because gastroduodenal contents are constantly exposed to the fistula site and comprise part of the effluent. The volume and contents of the effluent from these biliary leaks likely contribute to their severity. Roux-en-Y hepaticojejunostomy is the most versatile biliary reconstruction method. It can be used in any operation after transection of the extrahepatic bile duct. In a review of 519 hepaticojejunostomies performed for a wide range of indications including pancreatic cancer, chronic pancreatitis, cholangiocarcinoma, and transplantation, bile leaks occurred in 5.6 % of patients [10]. Independent risk factors for leakage were preoperative radiochemotherapy, preoperative low cholinesterase levels, simultaneous liver resection, and reoperation after liver transplant. Another high-volume center reported hepaticojejunostomy leak rates of 2.2 % after pancreaticoduodenectomy [11]. The only risk factor identified was a low preoperative albumin.
Hepatico- or choledochoduodenostomy either in an end-to-side or side-to-side fashion can be used in the setting of biliary calculus, strictures, bile duct cysts, and malignancy. Concerns about choledochoduodenostomy include sump syndrome and potential for duodenal fistula in the event of anastomotic leak. Sump syndrome can occur with side-to-side choledochoduodenostomy with the accumulation of debris in the distal blind end of the bile duct. One recent retrospective study demonstrated fewer anastomotic complications when the duodenum was used for biliary reconstruction [12] and no difference in frequency of biliary fistulae following end-to-side choledochoduodenostomy (8 %) when compared with Roux-en-Y jejunal reconstruction (16 %). It should be noted, however, that Roux-en-Y was used more frequently when the reconstruction was above the confluence. In another retrospective review from India, 270 patients underwent side-to-side choledochoduodenostomy with a biliary leak documented in 2 % of patients [13]. Sump syndrome was not observed in any of these patients, the majority of whom underwent preoperative endoscopic retrograde cholangiography (ERC) and papillotomy. While retrospective, these studies suggest the frequency of biliary leaks may be comparable to Roux-en-Y reconstruction with low incidence of sump syndrome.
Choledochocholedochostomy is primarily used in the setting of orthotopic liver transplantation (OLT), but has also been described in the repair of bile duct injuries. In general, end-to-end choledochocholedochostomy has not been favored for repair of iatrogenic bile duct injuries given concern for subsequent stricture formation [14]). Limited data exist to support this technique outside of transplant. One retrospective study comparing Roux-Y hepaticojejunostomy with end-to-end reconstruction in 94 patients demonstrated similar rates of biliary leaks and bilomas (10 and 7 % respectively) [15]. End–end reconstruction was used with ducts greater than 4 mm in diameter in the absence of inflammation. With a mean long-term follow-up of 62 months, no significant difference in strictures was identified.
In contrast to bile duct injuries, choledochocholedochostomy is commonly used in orthotopic liver transplantation. A recent systematic review of over 11,000 orthotopic liver transplants documented biliary leaks in 8 % of cases [16]. The use of T-tubes to reduce biliary complications remains an area of debate. Numerous prospective randomized trials have been performed to address this issue with conflicting results [17]. Many centers have abandoned routine use of T-tubes given the evidence from several prospective randomized trials indicating the anastomoses can be performed with similar or lower rates of strictures and biliary leaks. Indeed, in the systematic review by Akamatsu et al., 82 % of over 6000 deceased donor liver transplantations with duct-to-duct anastomoses were performed without a T-tube. In contrast, many centers continue to use splinting stents for biliary reconstruction during live donor liver transplantation, and use of a transcystic stent for biliary reconstruction during OLT has also been described [18].
Prevention
Specific criteria for optimal construction of bilioenteric anastomoses are sparse and techniques vary widely. Primary recommendations for prevention of biliary leaks after bilioenteric anastomoses are (1) well-vascularized bile duct, (2) absence of cholangitis and inflammation of the bile duct, (3) tension-free anastomosis, (4) well-vascularized duodenum or jejunum, and (5) atraumatic suture placement. For dilated bile ducts, a single running layer of absorbable monofilament suture is effective and efficient. For nondilated ducts or for complex biliary anastomoses involving multiple ducts, interrupted absorbable sutures are used. If multiple duct orifices are present, adjacent ducts can be joined with interrupted absorbable sutures to reduce the number of bilioenteric anastomoses. Biliary stenting to bridge the bilioenteric anastomosis has not been shown to reduce biliary leaks after biliary reconstruction. Stents, however, may be indicated to bridge anastomoses after R1–2 resection to ensure biliary access for subsequent intraluminal therapy or dilatation.
Risk Factors for Bile Leak After Liver Resection
Risk factors for biliary leak after hepatic resection have been confounded by the lack of uniform definition of biliary leak in the literature. Several recent studies have retrospectively investigated the incidence and risk factors for biliary leaks. In a review of 2628 consecutive resections, preoperative jaundice, portal vein embolization, liver resection for biliary tumors, repeat hepatectomy, extended hepatectomy, caudate resection, two-staged resection, en bloc diaphragm resection, bile duct resection and reconstruction, longer operative duration, greater estimated blood loss (EBL), larger tumors, portal lymph node dissection, and intraoperative transfusion were risk factors for biliary leak on univariable analysis [19]. Of these factors, repeat hepatectomy, bile duct resection, intraoperative transfusion, en bloc diaphragm resection and extended hepatectomy were found to be independent predictors for biliary leak on multivariable analysis. It is unclear whether factors such as increased blood loss or intraoperative transfusion are simply surrogates for increased operative complexity or directly affect biliary leaks.
In another study of 505 consecutive liver resections without bile duct resection, biliary leaks were identified in 6.7 % of patients. Multivariable analysis identified repeat hepatectomy, a large cut surface area, and intraoperative blood loss as independent predictors for biliary leaks [20]. In 610 patients undergoing liver resection without bile duct resection, peripheral cholangiocarcinoma, left hepatectomy including segment 1, transection plane outside of the main portal scissure, and hepatectomies including the caudate or segment four were independent predictors of biliary leakage. On multivariable analysis, peripheral cholangiocarcinoma and resection of segment 4 were risk factors for bile leaks. Use of fibrin glue and cirrhosis were found to decrease the incidence of leaks [21].
Collectively, these studies suggest that complex liver resections involving the caudate, extended hepatectomy, and increased blood loss increase the risk for biliary leak and may warrant additional methods to assess for biliostasis intraoperatively.
Prevention of Biliary Leaks After Hepatectomy
Apart from intraoperative blood loss and transfusion, most identified risk factors are not modifiable. Techniques used to reduce intraoperative blood loss and transfusion requirements include use of intermittent inflow occlusion, low central venous pressure, and meticulous hemostatic technique. A variety of parenchymal transection techniques have been described. These include the clamp crush, Cavitron ultrasonic surgical aspirator (CUSA-Tyco Healthcare, Mansfield, MA), water-jet dissection, stapling devices, and energy devices, e.g., Ligasure (Valleylab, Tyco Healthcare, Boulder, CO, USA), Harmonic scalpel (Ethicon Endosurgery, Cincinnati, OH, USA), TissueLink (Salient Surgical Technologies, Portsmouth, NH). No parenchymal transection method has been shown superior in reducing the rate of biliary leaks. One randomized control trial assessing the impact of parenchymal transection technique in 120 patients who were allocated to either clamp crushing or Ligasure showed no difference in biliary leak between groups [22]. A retrospective analysis of 141 patients undergoing hepatic resection without bilioenteric anastomosis compared clamp crushing, stapling, and Tissuelink with no difference in the rate of biliary leakage [23]. Among 300 patients undergoing stapler hepatectomy, the incidence of biliary leak was 8 % and was claimed to be comparable to other parenchymal transection techniques [24]. For open or laparoscopic hepatectomy, identification and stapling transection of lobar, sectional, or segmental biliary duct provide secure closure. Minor or intrasegmental biliary ducts are secured with clips or suture ligature. Transection methods should avoid trauma to the hilar ducts that can predispose to late leaks. Laparoscopic hepatic resections utilize the Harmonic scalpel and endovascular staplers. The former technology likely fuses small ductules; however, conflicting data exist on the incidence of biliary leaks ranging from 24 to 1 % [25, 26].
Intraoperative Tests for Bile Leaks
A variety of methods have been used to detect biliary leaks intraoperatively. A simple method to assess for biliary leaks is to place a white surgical sponge on the cut surface to detect bile staining. Identified sites of bile leakage are oversewn with suture. However, this method is dependent upon bile flow. Evacuation of bile from the biliary ducts intraoperatively may lead to falsely negative findings. Other intraoperative biliary leak tests utilize perfusion of the ducts. These methods include the injection of saline, methylene blue, or indocyanine green retrograde through the cystic duct [27]. The efficacy of these intraoperative tests remains unclear. One randomized trial of 103 consecutive patients undergoing hepatic resection without biliary enteric anastomosis showed no difference in bile leaks when isotonic saline was injected through the cystic duct [28]. Another report indicated decreased incidence of bile leaks with indocyanine green and fluorescent cholangiography in 102 randomized patients [29]. Finally, a recent report of 223 patients, half of which underwent an intraoperative air leak test by introduction of air through a transcystic cholangiogram catheter, demonstrated a reduction in biliary leaks from 10 to 1.9 % [19]. Further validation of various techniques and comparative trials will be required to confirm optimal.
Postoperative Drains
The role of postoperative drains after hepatic resection remains controversial. Proponents claim that drains are infrequently the source of morbidity and, if appropriately positioned and of appropriate type, control biliary leaks. Conversely, opponents claim drains are unnecessary given the low frequency of biliary leaks and can cause morbidity, specifically surgical site infection, biliary erosion, and leak or inadequate biliary leak control if present. Many centers place drains prophylactically at the parenchymal transection site. Some evidence suggests that routine placement of drains is unnecessary and may be associated with increased infection [30, 31]. In the largest series, an audit of over 1000 liver resections failed to demonstrate increased morbidity in the 80 % of patients who were not drained. Intraoperative drains were placed if patients underwent a simultaneous thoracotomy, an uncontrolled bile leak was encountered, bilioenteric anastomosis was performed, or if the field was infected. A metaanalysis did not demonstrate a statistically significant difference in rate of biliary fistula, reoperation, intraabdominal collections requiring aspiration or drainage, with or without an operatively placed drain [32]. Minor liver resections are drained rarely. Biliary reconstruction combined with major hepatectomy, and major resections leaving a large cut surface are drained.
Diagnosis
The diagnosis of a biliary leak is generally straightforward if an operative drain was placed and it communicates with the site of bile leakage. In general, inspection of the effluent is confirmatory of a biliary leak. If diagnostic uncertainty exists, analysis of bilirubin concentration is performed. If a drain was not placed or does not communicate with the site of leakage, patients may present with signs of systemic inflammatory response (fever, tachycardia, tachypnea, leukocytosis), localized abdominal pain, expression of bilious fluid from the incision, or more subtle findings such as delayed gastric emptying or ileus. Suspicion for a biliary leak should be raised in the setting of an unexplained leukocytosis or increase in bilirubin in the postoperative period. In this setting, an ultrasound or contrast-enhanced computed tomography (CT) scan may demonstrate the presence of perihepatic fluid collections. Image-guided drain placement with bilious output is generally diagnostic of a biliary leak.
Investigations
A number of imaging modalities are used to evaluate biliary leaks. These include abdominal ultrasonography (US), CT scan, fistulogram/sinogram, magnetic resonance cholangiography (MRC), ERC, and percutaneous hepatic cholangiography (PTC). Hepatobiliary iminodiacetic acid (HIDA) scans, though used, rarely provide useful anatomic information.
Ultrasonography or CT Scan
When a biliary leak is suspected, abdominal ultrasonography or crosssectional imaging is recommended. Initially, a noninvasive imaging modality is used to detect an undrained collection and may provide clues to the origin of the leak (Fig. 18.1). Identification of biliary dilatation and assessment for concomitant vascular injury are also important.
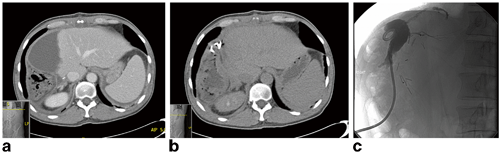





Fig. 18.1
a CT abdomen demonstrates perihepatic fluid collection after right hepatectomy. b CT-guided percutaneous drain placement of biloma. c Sinogram demonstrates opacification of a cavity communicating with the biliary tree
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








