Fig. 14.1
Diagram showing the six approaches to the superior mesenteric artery. S superior approach, A anterior approach, P posterior approach, L left posterior approach, R right/medial uncinate approach, M mesenteric approach. This figure was reproduced from [4] with a permission of the publisher
Based on the six primary methods of artery-first approaches to PD (Fig. 14.1), many other variants have been described; for example, we have adopted an artery-first approach to distal pancreatectomy with celiac artery resection [5]. Herein we define “artery-first pancreatic resection ” as any pancreatic operation in which any technique is used to ligate the feeding arteries before the division of the pancreas with the intent of reducing blood loss and of achieving a more oncologically complete resection.
Advantages of Artery-First PD
Although the majority of surgeons initiate the dissection in a standard PD with a Kocher maneuver, an increasing number of surgeons are performing an artery-first PD. Putative merits of this general approach include (1) reduction of intraoperative blood loss, (2) early determination of arterial involvement by tumor, (3) clearance of surgical margins along the arteries, and (4) ultimately, a more oncologically complete resection.
In a case-matched study comparing artery-first PD using a posterior approach (n = 21) versus standard PD (n = 21), there was a significantly lower mean blood loss (P = 0.0314) and a shorter operative time (P = 0.0002) associated with artery-first PD. There were no significant differences identified in rates of early morbidity and mortality, length of hospitalization, overall survival, and survival according to tumor type [6]. In another series reported by Kurosaki and his colleagues, there were no significant differences in operating time, blood loss, hospital stay, or overall morbidity between the group of patients who underwent artery-first PD by the left posterior approach (n = 40) and that of patients who underwent standard PD (n = 35). However, artery-first PD was associated with fewer recurrences (P = 0.006) and improved 1- and 3-year survival rates (P = 0.004) compared to standard PD [7]. Though additional retrospective studies exist in which artery-first PD and standard PD are compared, their results are inconsistent and no conclusions can generally be drawn from them. Controlled randomized trials are needed to validate the potential advantages of artery-first PD.
Role of Artery-First PD
Artery-first PD has been practiced for two decades by enthusiastic pancreatic surgeons and it is routinely used in many specialized institutions in Japan [4]. In contrast, artery-first PD has been less popular in Western countries, possibly due to difficulty in identification and dissection of arteries in obese patients. Worldwide, there is an increasing role for artery-first PD given the increasing role for surgical resection in the setting of borderline resectable pancreatic cancer [8]. Indeed, consensus exists that operative exploration and resection may be indicated, in high-volume centers with surgical and multidisciplinary expertise, in the case of involvement of SMV and/or portal vein or limited involvement of the SMA, but not in that of arterial encasement [9]. Therefore, it has become necessary to assess for the presence or absence of arterial encasement at an early stage of pancreatectomy. The artery-first PD is an ideal approach to this clinical problem.
The artery-first PD is also appropriate following administration of chemotherapy and/or chemoradiation [10]. Indeed, pancreatic surgeons are asked to resect ever-increasing numbers of patients with borderline resectable and locally advanced pancreatic cancer who have received prior induction therapy. It is not possible to predict pathologic involvement of the SMA after neoadjuvant therapy by imaging alone because fibrotic tissue may often remain after treatment even in the setting of a significant pathologic response. Upon surgical exploration, surgeons must dissect around the major arteries such as the SMA and GDA and decide whether or not to resect before the “point of no return.” In such a situation, the artery-first PD is the choice of operation. Meticulous dissection of the SMA by the artery-first PD may increase margin negative rates and influence locoregional control [11].
Surgical Techniques
At our institution, we use the artery-first PD for all cases so that surgeons in training become familiar with the surgical principles and gain experience. The surgical techniques we use are presented here in detail.
Tora-no-Ana Approach
In the setting of pancreatic cancer, we utilize the “Tora-no-Ana” approach. In a Chinese text from the Han dynasty, there is a proverb: “One cannot get a tiger’s cub without entering the tiger’s lair.” The term of Tora-no-Ana represents the tiger’s lair in the Japanese language. Here, it also refers to an anatomic opening (= Ana) created by division of the ligament of Treitz (= Tora).
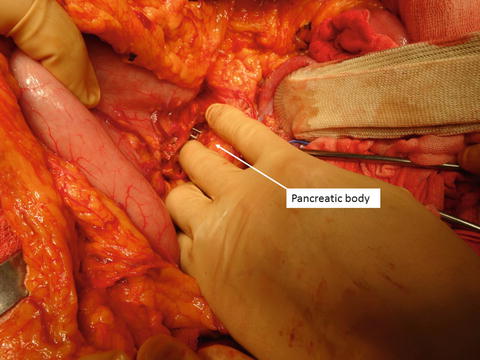
In the operating room, the operator stands on the right side of the patient, the first assistant stands on the opposite side, and the second assistant stands cranial to the operator (Fig. 14.2). The transverse colon is lifted upward by an assistant surgeon and the ligament of Treitz is divided along the lateral margin of the upper jejunum (Fig. 14.3) and the retroperitoneal space is entered. Thus, the Tora-no-Ana is created and the para-aortic lymph nodes are sampled for frozen section. If needed, the inferior mesenteric vein (IMV) may be divided; the inferior posterior margin of the pancreatic body is then mobilized to open the Tora-no-Ana widely. Now, the surgeon can insert his or her right four fingers into the Tora-no-Ana and palpate the SMA and its branches by grasping the mesentery of the upper jejunum between the thumb and fingers (Fig. 14.4). In this way, the surgeon can locate the SMA no matter how obese the patient may be. During the procedure for further dissection, this palpation may be repeated so that the location of SMA is clearly identified.
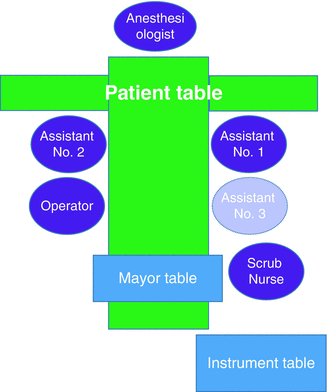
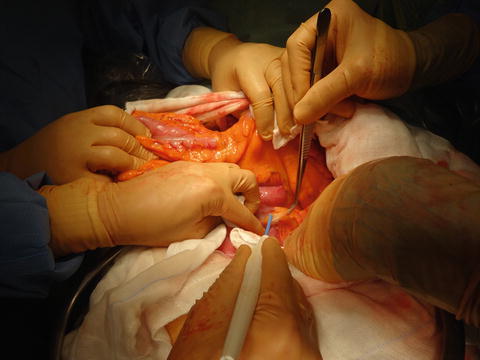
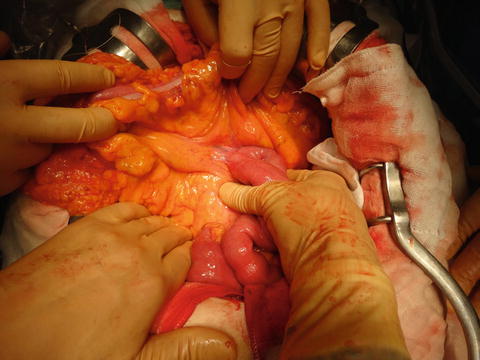

Fig. 14.2
Setting in operation room. A setting of the operation table, surgeons, scrub nurse, and anesthesiologist in the operation room. The assistant No.3 is optional

Fig. 14.3
Tora-no-Ana approach. The ligament of Treitz is divided along the lateral margin of the proximal jejunum, while the assistant surgeon retracts the transverse colon upward

Fig. 14.4
Palpation of the superior mesenteric artery. The surgeon inserts his or her right four fingers into the Tora-no-Ana and palpates the superior mesenteric artery and its branches between the thumb and fingers
The anterior peritoneum and adipose tissue of the mesentery is divided at the base of transverse mesocolon (Fig. 14.5). Usually, it is easier to first expose the SMV, which runs parallel to the SMA. The middle colic artery is divided at its origin for the better exposure of SMA (Fig. 14.6). It is important to identify the first jejunal vein in order to avoid incidental injury and bleeding. The SMV and first jejunal vein are looped (Fig. 14.7).
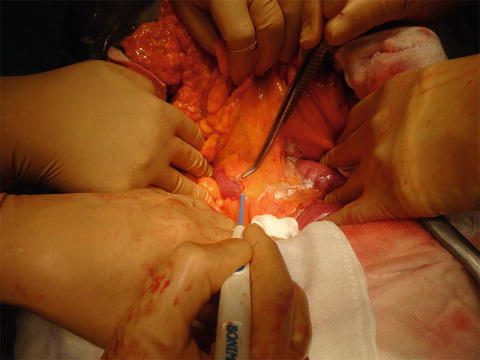
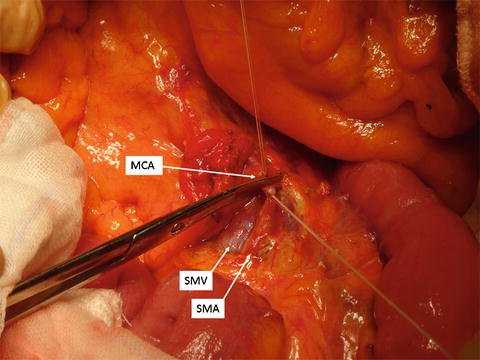
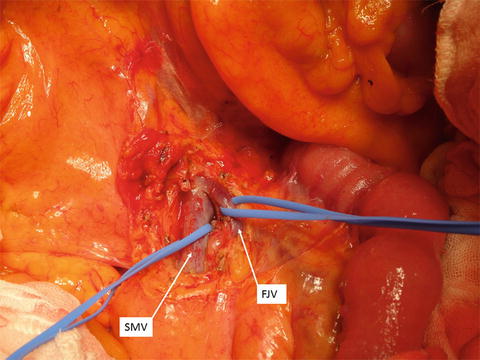

Fig. 14.5
Mesenteric approach. The peritoneum of the mesentery is divided between the inferior duodenal angle and proximal jejunum, while the assistant surgeon retracts the transverse colon upward

Fig. 14.6
Division of the middle colic artery. The middle colic artery (MCA) is divided close to the origin from the superior mesenteric artery (SMA). SMV superior mesenteric vein

Fig. 14.7
Taping of the superior mesenteric vein and first jejunal vein. The superior mesenteric vein (SMV) and first jejunal vein (FJV) are exposed and taped at the base of transverse mesocolon
Division of the Transverse Mesocolon
The gastrocolic ligament is divided and the lesser sac is entered (Fig. 14.8). The middle colic vessels and right aberrant colic vessels are divided at their takeoffs from the inferior margin of the pancreatic body. The transverse mesocolon is widely divided along the anterior inferior margin of the pancreatic head and body (Fig. 14.9). In this way, a part of the mesentery of transverse colon, where the head and proximal body of the pancreas are attached, is resected en bloc. In order to prevent ischemia of the colon, it is mandatory to exercise caution for preserving the arcade of vessels along the transverse colon.
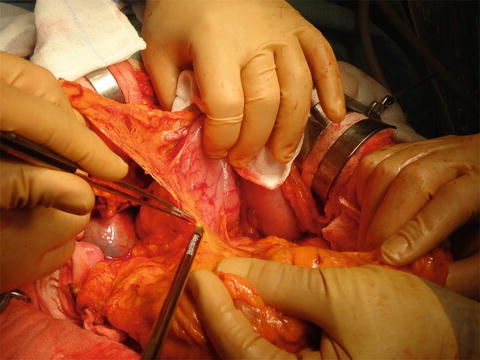
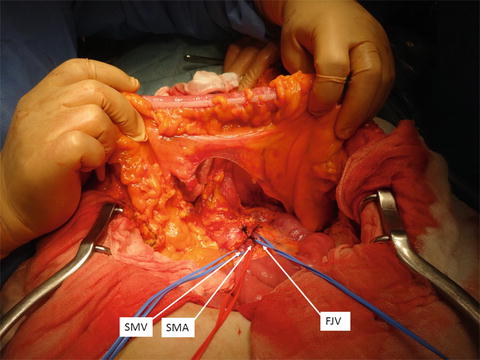

Fig. 14.8
Division of the gastrocolic ligament. The gastrocolic ligament is divided with a vessel sealing device and the lesser sac is entered. The superior mesenteric vein should not be exposed from the lesser sac or else the surgical margin may be compromised

Fig. 14.9
Opening of the transverse mesocolon. The transverse mesocolon is divided along the anterior inferior margin of the pancreatic head and body and widely opened. SMA superior mesenteric artery, SMV superior mesenteric vein, FJV first jejunal vein
Hanging Maneuver of the Pancreatic Body
After making a wide opening within the mesocolon, the transverse colon is retracted downward and the pancreas is well exposed. By retracting the stomach body upward, an avascular area on the left side of the left gastric artery and superior to the splenic artery is dissected with a cautery for the preparation of later passage of a Penrose drain. Large Kelly forceps are inserted into the dissection plane between the pancreatic body and the SMA and are passed toward the avascular area that was dissected previously (Fig. 14.10). As long as the forceps are passed under the fusion fascia of Treitz, they should meet no resistance. One should not attempt to progress the forceps between the pancreatic body and splenic artery, or an injury to the splenic vessels or dorsal pancreatic artery can result. A Penrose drain is passed with the large Kelly forceps (Fig. 14.11). A hanging maneuver is performed by lifting the Penrose drain cephalad, upon which hang the pancreatic body together with the splenic artery and vein (Fig. 14.12). With the hanging maneuver , the inferior vena cava is well exposed from the left side, and the left renal vein is exposed thorough the Tora-no-Ana.