Fig. 4.1
(a) Endoscopic photograph of thick mucus extruding from the orifice of the major papilla. This finding is compatible with main-duct intraductal papillary neoplasm, a condition strongly associated with pancreatic ductal adenocarcinoma. (b) Pancreatogram revealing a long cast-like filling defect in the main pancreatic duct, reflecting the presence of intraductal mucus. Also note the presence of a ductal stricture in the head of the pancreas
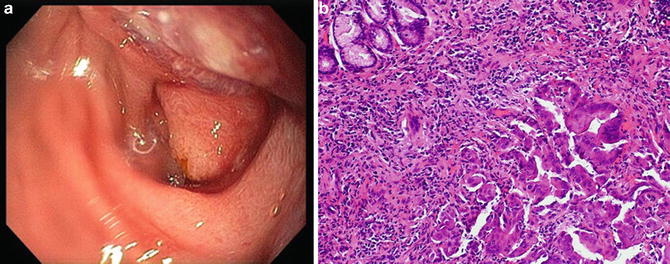
Fig. 4.2
(a) Endoscopic photograph of direct invasion of the duodenal wall caused by a pancreatic head mass. Note the uninvolved orifice of the major papilla seen downstream. (b) Standard forceps biopsies confirmed adenocarcinoma invading the duodenal wall
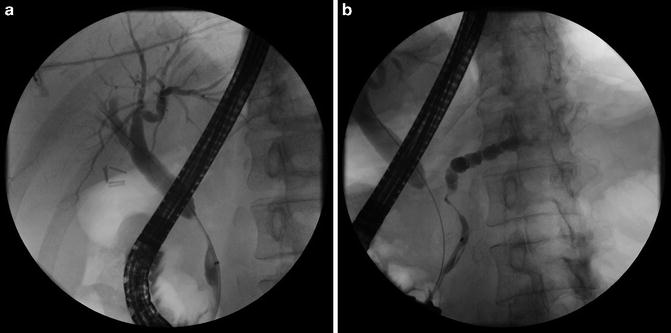
Fig. 4.3
(a) Double-duct sign. Cholangiogram revealing a common bile duct stricture with upstream dilation. (b) Pancreatogram revealing a long irregular stricture in the head of the pancreas with upstream dilation. Simultaneous obstruction of the common duct and pancreatic duct is highly suggestive of a mass lesion in the head of the pancreas
Tissue sampling techniques during ERCP include brush cytology, forceps biopsy, aspiration of bile or pancreatic juice for cytology, or a combination. In patients in whom a plastic stent has already been placed, the stent can be spun and the cells obtained can be evaluated [8]. Exfoliated malignant cells may be adherent to the surface of the stent as they become entrapped within biofilm and sludge. The sensitivity rate for ERCP-directed brush cytology or biopsy is 30–50 %, with a combination of techniques achieving sensitivity rates of approximately 70 % [9, 10]. This is considerably less than EUS-guided fine needle aspiration (FNA) which has a sensitivity of approximately 85–90 % for the diagnosis of pancreatic cancer [11]. Several studies have shown that diagnostic yield during ERCP can be increased by using a combination of different tissue sampling methods [12, 13]. Unfortunately, the negative predictive value in tissue sampling during ERCP using a combination of techniques is nearly 40 % [12].
Although aspiration of bile or pancreatic juice is simple to perform, fluid cytology alone has a low sensitivity and is not performed by most endoscopists. Fluid specimens are often acellular, likely due to the desmoplastic nature of certain tumors or failure to invade the ductal epithelium. Techniques to increase tumor exfoliation prior to collecting specimens, such as stricture dilation or saline irrigation, have not demonstrated increased cancer detection rates in prospective comparative trials [12]. Forceps biopsies have a higher yield, but generally require a sphincterotomy to gain access to the bile duct or pancreas. When performing forceps biopsies, it may be helpful to first place a guidewire across the stricture to maintain access and for use as a guide for cannulation and positioning of the biopsy forceps (Fig. 4.4). Performing intraluminal forceps biopsies during ERCP can be technically challenging as the device cannot be passed over a guidewire. It may also increase the risks of the procedure, including bleeding, pancreatitis, and perforation. By comparison, biliary brush cytology is relatively easy to perform as the brush passes over a prepositioned guidewire to acquire a specimen within the stricture. The overall technical success rate of biliary brush cytology is >90 %. Brush cytology in the pancreatic duct is sometimes helpful but is frequently more difficult to perform. Pancreatic cancer often causes tight strictures of the main pancreatic duct which prohibit passage of the brush through the tumor in greater than 25 % of patients [12] (Fig. 4.4). Because of the aforementioned challenges, most practitioners perform biliary brush cytology alone, which has sensitivity as low as 30 %. Although the sensitivity of brush cytology or forceps biopsy alone is suboptimal, both techniques are almost 100 % specific [13]. Advanced techniques such as digital image analysis may enhance the accuracy of routine cytology [14], but is not widely available. Additional methods to improve the diagnostic yield such as the molecular analysis of the components of pancreatic juice and bile remain experimental [9, 15, 16]. Although the overall performance of tissue sampling techniques during ERCP in patients with suspected pancreatic cancer is significantly lower than EUS-FNA, it remains an important modality and should be performed whenever a diagnosis has not been established at the time of the procedure.
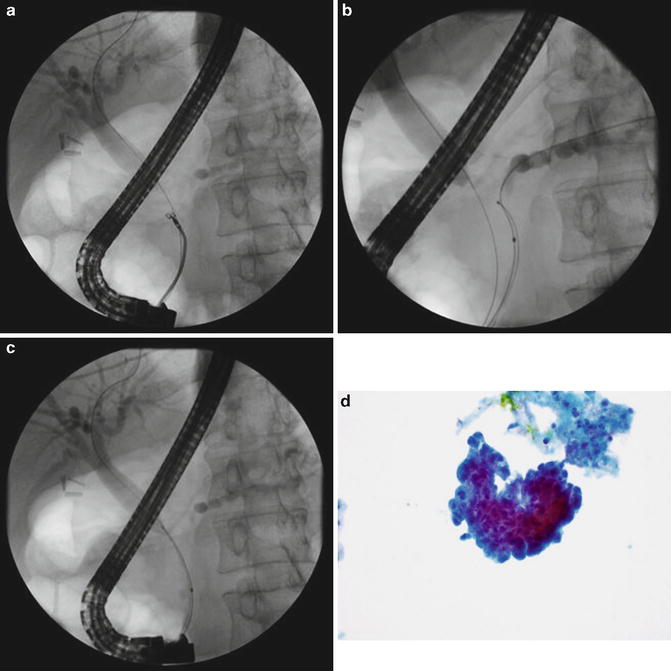

Fig. 4.4
(a) Biliary forceps biopsy. A guidewire passed through the biliary stricture is used as a guide for cannulation and positioning of the biopsy forceps. (b) Pancreatic duct brush cytology. A second guidewire has been passed through the pancreatic duct stricture and is used to position the cytology brush. Brushings within pancreatic duct may be challenging to perform due to the tight nature of the stricture. (c) Biliary brush cytology. A cytology brush has been passed over a prepositioned guidewire to acquire a specimen within the stricture. (d) Photomicrograph of a specimen obtained during biliary brush cytology revealing crowding and overlapping of cells, compatible with adenocarcinoma
Rationale for Preoperative Biliary Drainage
Historically, major hepatobiliary surgical procedures in patients with obstructive jaundice have been associated with significant morbidity and mortality, largely due to the development of postoperative complications such as sepsis, bleeding disorders, and renal failure. Biliary obstruction has been regarded as a risk factor that can worsen the outcome after surgery [17]. The primary rationale of PBD for patients with biliary obstruction due to pancreatic cancer is to reduce the risk of postoperative complications. The concept of PBD was introduced by A.O. Whipple and colleagues in 1935 when they published one of the first case series of PBD for patients with periampullary cancer [18]. The two-staged technique involved performing a preliminary open biliary diversion procedure (cholecystogastrostomy) to reduce jaundice, followed by resection of the primary tumor at a later stage, depending on the severity of jaundice. The goal of this approach was to optimize the overall physical status of the patient prior to definitive resection.
Biliary obstruction is associated with several deleterious effects. Animal studies have shown that obstructive jaundice leads to a proinflammatory state resulting from portal and systemic endotoxemia [19]. Decreased bile in the intestinal lumen causes increased permeability of the intestinal mucosal barrier, promoting bacterial translocation and the occurrence of endotoxemia [20]. Systemic endotoxemia leads to impaired cellular immunity and increased concentrations of proinflammatory cytokines such as interleukin-6 (IL-6), interleukin-10 (IL-10), interleukin-8 (IL-8), and tumor necrosis factor (TNF) [21–23]. The overall effects of obstructive jaundice in humans on endotoxin and cytokines may be different from those seen in animal models [24]. Biliary obstruction also causes a reduction in hepatic reticuloendothelial system function leading to a diminished clearance of endotoxin by Kupffer cells [24, 25]. Persistent elevation of cytokines has been associated with protein calorie depletion, a factor associated with higher surgical complications which could potentially be reversed by biliary decompression. Malignant biliary obstruction may also adversely affect coagulation due to bile acid-induced hepatocyte damage [26] as well as impaired hepatic synthesis of vitamin K-dependent coagulation factors secondary to reduced vitamin K absorption from the intestine. Despite these effects favoring bleeding complications, a recent study has shown that patients with severe biliary obstruction may also develop a procoagulant state which was almost completely reversed by preoperative endoscopic biliary drainage [27]. In addition to impairment of immune function and coagulopathy, biliary obstruction is also associated with renal dysfunction. Cholestatic jaundice is known to have deleterious effects on cardiovascular function, blood volume, and vascular reactivity. The overall effect of obstructive jaundice predisposes the kidney to prerenal failure and acute tubular necrosis. Most evidence suggests that the constituents of bile (cholesterol, bilirubin, bile acids) do not exert a direct nephrotoxic effect [28]. A multivariate analysis has shown that renal dysfunction in patients with obstructive jaundice is associated with the degree of biliary obstruction as well as the age of the patient [29]. Biliary obstruction may also be associated with impaired myocardial function and is associated with increased plasma levels of atrial natriuretic peptide (ANP). Internal biliary drainage results in improvement in cardiac function and normalization of ANP [30].
The adverse effects of biliary obstruction on multiple organ systems and immune function may adversely impact the outcome after major surgery for patients with pancreas cancer. Preoperative biliary drainage has the potential to improve surgical outcomes by reversing the detrimental effects via restoration of bile flow.
Methods of Preoperative Biliary Drainage
Endoscopic stent placement and percutaneous biliary drainage have largely replaced surgical biliary bypass for management of biliary obstruction due to pancreatic cancer. These techniques are generally considered less invasive, less expensive, and have a shorter recovery time as compared to surgical procedures. The choice between endoscopic vs. percutaneous biliary drainage is often a matter of a local expertise and patient anatomy, although endoscopic stent placement is preferred whenever possible due to fewer procedure-associated complications [31]. Percutaneous biliary drainage is more often used when endoscopic stent placement is unsuccessful or not technically possible due to altered anatomy (e.g., duodenal obstruction, tumor invasion of the ampulla, or previous surgical bypass procedures).
Percutaneous Biliary Drainage
Percutaneous transhepatic biliary drainage (PTBD) was introduced in the 1960s and was the treatment of choice for biliary drainage for over two decades [32, 33]. PTBD drainage is most often performed using fluoroscopic guidance although ultrasound can be helpful for the initial puncture when the bile ducts are dilated [34]. The technique involves passing a skinny needle (21 or 22 gauge) through the hepatic parenchyma until reaching a dilated intrahepatic bile duct. A percutaneous cholangiogram is performed by injecting contrast as the needle is slowly withdrawn, followed by passage of a small diameter (0.018 in.) guidewire to secure the position in the biliary tree. Once the dilated duct has been accessed with the needle, the needle is exchanged for a coaxial system to upsize the 0.018-in. access guidewire to a larger guidewire (e.g., 0.035 or 0.038 in.) which is more stable and can be used for further interventions.
PTBD can provide biliary drainage in three ways. The simplest of these is external drainage which involves decompressing the biliary tree through a percutaneous tube which exits the skin, but the intraductal tip is left upstream to the site of biliary obstruction. The method is typically used when a tight stricture cannot be traversed with a guidewire after percutaneous access to the biliary tree has been achieved. A major disadvantage of external drainage is the fact that bile flow to the duodenum is not restored. For internal–external drainage, a directional catheter is inserted through the percutaneous sheath and advanced over a hydrophilic guidewire through the biliary obstruction and into the duodenum. The catheter can then be exchanged over a stiffer guidewire (e.g., Amplatz) for a multiside-hole drainage catheter which is passed through the stricture into the duodenum. The internal–external catheter allows bile to drain externally into a bag and/or internally into the duodenum, thereby preserving the normal enterohepatic circulation of bile (Fig. 4.5). The third technique establishes internal drainage by percutaneous placement of a plastic or self-expandable metal stent (SEMS) across the biliary stricture. Recent studies have shown percutaneous SEMS placement to be a safe and effective technique [35–38]. Although it is common practice to establish initial internal–external drainage prior to SEMS placement, some experienced centers have reported good results with percutaneous SEMS insertion as a single-stage procedure [35, 36]. A retrospective study from the UK reported an overall technical success rate of 79 % among 67 patients undergoing percutaneous short SEMS placement for biliary obstruction due to pancreatic or periampullary tumors [35]. The complication rate was 9.4 % although all complications were managed conservatively and none precluded subsequent surgery.
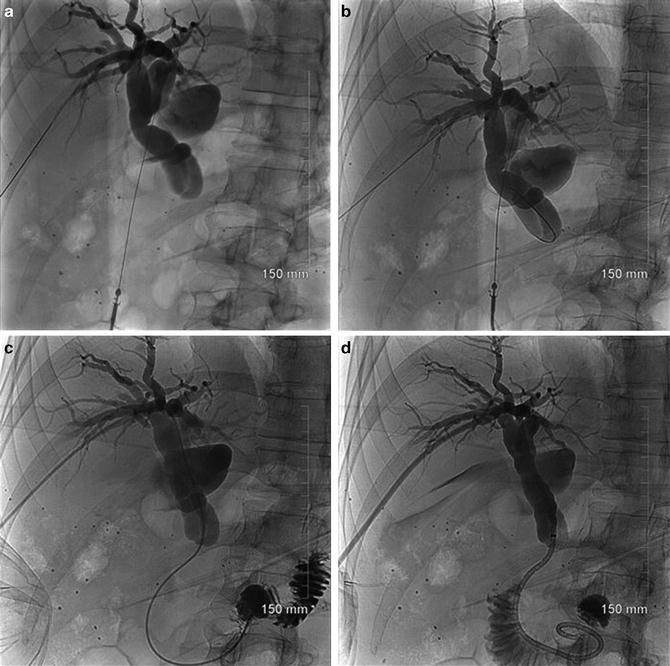

Fig. 4.5
(a) The patient is a 60-year-old male with borderline resectable pancreatic head cancer who underwent unsuccessful ERCP due to failed bile duct cannulation. A percutaneous transhepatic cholangiogram was performed by injection of contrast through a 22 gauge Chiba needle. Needles are shown entering left and right intrahepatic ducts. (b) Initial attempts to pass a guidewire through the high-grade bile duct stricture in the head of the pancreas were unsuccessful. No contrast flowed through the stricture. (c) A stiff 0.035 in. hydrophilic guidewire and 5 Fr catheter were ultimately passed through the stricture into the duodenum. (d) Following placement of a 0.035 Amplatz guidewire and dilation of the tract to 10 Fr, a 10 Fr multiside hole internal-external drainage catheter was placed with tip reaching the transverse duodenum
One disadvantage of PTBD is that it cannot be used in the presence of moderate or severe ascites [39]. PTBDs can be cumbersome for patients to manage and require significant maintenance. External drains require periodic emptying, flushing of the drain, and drain exchanges to prevent occlusion [40]. PTBDs can also be prone to leakage, dislodgement, and complications such as hemobilia and infection. A recent prospective study involving 109 patients with advanced malignancy showed that PTBD improved pruritus and hyperbilirubinemia, but not overall quality of life [41]. Despite potential drawbacks, PTBD continues to have an important role for management of biliary obstruction, especially when ERCP is unsuccessful [42].
Endoscopic Biliary Drainage
The most common and generally preferred method of achieving preoperative biliary drainage is by ERCP with stent placement. Endoscopic stents are often used as a bridge to surgery for patients with resectable or borderline resectable disease as well as for long-term palliation for unresectable pancreatic cancer. The main advantage of an endoscopic approach over PTBD is the avoidance of skin and liver punctures as well as the risk of tumor seeding which may occur along the catheter and to the skin [43]. Recent meta-analyses have suggested that endoscopic stenting provides superior results to open surgical bypass in patients with distal biliary obstruction due to pancreatic cancer [7, 44]. Biliary drainage may be achieved using either plastic stents or SEMS and it is now clear that stent luminal diameter is a critical factor for both types as the risk of stent occlusion correlates with stent diameter. In general, wider diameter stents have a lower risk of short-term occlusion, whether plastic or metal.
Plastic biliary stents have been used since their development in the 1980s and are now commercially available in a wide variety of diameters, lengths, and designs (Fig. 4.6). They may be composed of various materials including polyethylene, polyurethane, and Teflon. Plastic biliary stents are available in diameters ranging from 5 to 12 Fr and lengths from 1 to 18 cm [45]. The primary advantages of using plastic stents for malignant biliary obstruction are that they are effective, have lower costs, and are easily removed or exchanged. Plastic stents are often selected when a diagnosis has not been established or the patient’s resectability status is unknown at the time of initial endoscopic treatment. The major disadvantage of plastic stents is that they have a high rate of occlusion due to formation of bacterial biofilm, sludge, as well as dietary fibers [46] (Fig. 4.7); this leads to the need for repeat procedures and stents exchanges. In general, 7 Fr plastic stents remain patent for approximately 8 weeks whereas 10 Fr plastic stents remain patent for an average of 3–5 months [47]. It is important to note that plastic biliary stents often do not maintain patency during the time required for most patients to complete neoadjuvant chemoradiotherapy for pancreatic cancer. A recent retrospective study reported that among 49 patients treated with plastic stents who were undergoing neoadjuvant therapy, 55 % required repeat ERCP for stent malfunction at a median of 82.5 days after initial stent placement [48]. Studies evaluating stent designs have compared stents composed of Teflon without side holes to standard polyethylene stents with side holes. No difference in patency rates was found based upon stent composition or design [49, 50]. Although it is generally accepted that larger diameter plastic stents (10 Fr or greater) have a longer patency than smaller diameter stents, a study comparing 10–11.5 Fr stents found no difference in patency rates [51]. A Cochrane meta-analysis found that choleretic agents such as ursodeoxycholic acid (UDCA) and/or antibiotics do not appear to improve plastic stent patency rates [52].

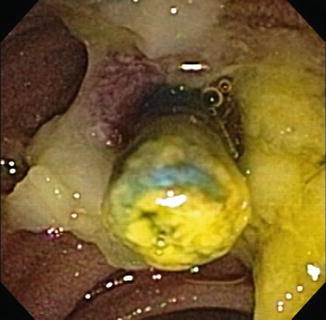

Fig. 4.6
Various plastic biliary stents. Plastic stents are available in a variety of diameters, lengths, and designs and may be composed of different materials. Stents which have a wider luminal diameter generally remain patent longer

Fig. 4.7
Endoscopic photograph of an occluded plastic biliary stent. Plastic stents occlude due to the formation of bacterial biofilm and biliary sludge. High occlusion rates is a limiting factor in the use of plastic stents for preoperative biliary decompression for pancreatic cancer
SEMS are now widely used for management of malignant biliary obstruction. As with plastic stents, SEMS are available in a variety sizes and designs (Fig. 4.8). Multiple studies have shown that when compared to plastic stents, SEMS have a superior patency rate when used for preoperative biliary decompression due to pancreatic cancer [7, 48, 53–58] (Fig. 4.9). The improved patency of SEMS relates to the fact that when fully deployed, SEMS have a roughly threefold wider luminal diameter than most plastic stents. Longer stent patency is especially important as more centers adopt neoadjuvant therapy as a standard of preoperative care. Stent occlusions during this period can result in severe complications such as cholangitis as well as interruptions in therapy, hospitalizations, unplanned procedures, and delays in eventual surgery [59]. In a recent prospective study evaluating SEMS in 55 patients undergoing neoadjuvant therapy for pancreatic cancer, only 15 % experienced stents malfunctioned by 260 days after placement [60]. This compares favorably to a 55 % stent malfunction rate when plastic stents were used for a similar patient population [48]. Another retrospective study evaluating plastic stents and SEMS for preoperative biliary decompression reported a 39 % stent dysfunction rate for those who received plastic stents compared to no stent dysfunction for those who received an SEMS [54]. Adams et al. evaluated stent complications among 52 patients who underwent placement of either a plastic stent or SEMS to receive neoadjuvant therapy for pancreatic cancer [57]. The complication rate was nearly seven times higher with plastic stents than with SEMS. Moreover, the rate of hospitalization for stent-related complications was threefold higher in the plastic stent group than the SEMS group.

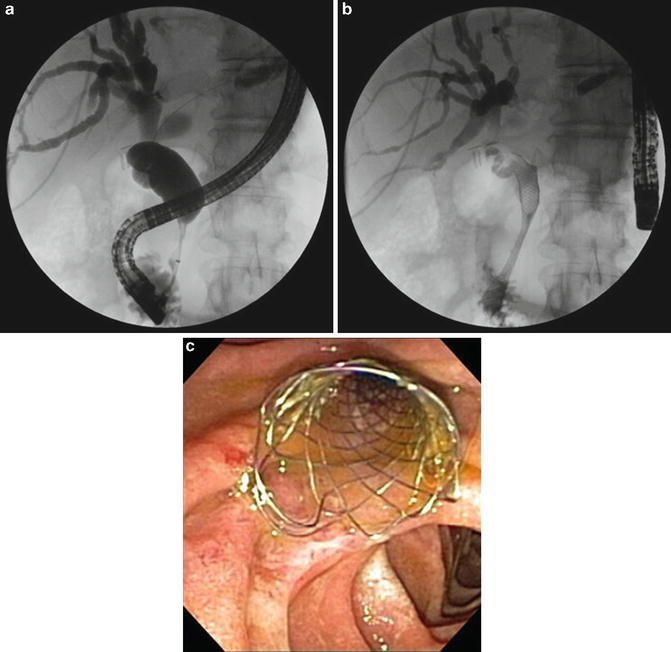

Fig. 4.8
Various self-expandable metal biliary stents. (a) Uncovered Zilver (Cook) (b) uncovered Wallflex (Boston Scientific) (c) partially covered Wallstent (Boston Scientific) (d) partially covered Wallflex (Boston Scientific) (e) fully covered Wallflex (Boston Scientific) (f) fully covered Viabil (ConMed)

Fig. 4.9
(a) A patient with borderline resectable pancreatic cancer underwent ERCP for management of obstructive jaundice prior to neoadjuvant therapy. The cholangiogram revealed a distal common bile duct stricture with upstream dilation. (b) A 10 × 60 mm biliary self-expandable metal stent was placed with subsequent resolution of jaundice. (c) Endophoto of a biliary self-expandable metal stent following placement
One factor that led to the initial use of plastic stents for preoperative biliary decompression was the concern that uncovered SEMS could potentially cause technical difficulties with transecting the bile duct and creating a biliary anastomosis during subsequent pancreaticoduodenectomy. Studies have now shown that placement of a short-length SEMS (typically 4–6 cm length) does not interfere with the outcome of surgery [5, 54, 61–63]. Siddiqui et al. reported the outcome of 241 patients with resectable or borderline resectable disease who underwent preoperative SEMS placement [63]. Uncovered, partially covered, and fully covered SEMS were used. Ultimately, 166 patients underwent curative-intent surgery without any observed technical difficulties during surgery due to the presence of an SEMS. Similarly, Mullen et al. found no difference in intraoperative or postoperative complications, or length of hospital stay among 29 patients who underwent pancreaticoduodenectomy after SEMS placement compared to those who had plastic stents (n-141), no stent (n-92), or biliary bypass (n-10) prior to surgery [64]. It is advisable during stent placement to use the shortest length SEMS possible to bridge the stricture with care taken to leave an adequate length of common hepatic duct un-stented (ideally 2 cm) to simplify any future surgical anastomosis, especially if using an uncovered SEMS. The choice between plastic stent vs. SEMS may ultimately rely on other factors such as cost, expected survival length, and certainty of diagnosis at the time of initial ERCP.
Although SEMS remain patent longer than plastic stents, they are also at risk for occlusion due to tumor ingrowth through the mesh interstices, overgrowth beyond the ends of the stent, or due to a hyperplastic response of normal tissue caused by the stent (Fig. 4.10). For this reason, SEMS were developed which are partially or fully covered with a goal of improving patency by preventing tumor and tissue ingrowth. Coverings include material made of polytetrafluoroethylene (PTFE), expanded polytetrafluoroethylene/fluorinated ethylene propylene (ePTFE/FEP), or silicone membranes. The covering may be on the exterior or interior of the stent. Some fully covered stents have fenestrations in the cover without exposing the metal wires. Unfortunately, covered stents may also occlude due to stent migration, tumor/tissue overgrowth, tumor ingrowth as the covering deteriorates over time, or possibly due to food debris [40]. Reflux of duodenal contents into SEMS is also known to occur [65] and could potentially cause problems in some patients. One of the advantages of uncovered SEMS, which has been shown in several studies, is their low migration rate (0–2 %) [56, 66, 67]. This is presumably due to embedding of the stent into the wall of the bile duct after deployment. Covered SEMS have a higher migration rate of approximately 6–8 %. Partially and fully covered stents have the advantage that they can be repositioned or fully removed using a rat-tooth forceps or snare [45]. SEMS are available in 6, 8, and 10 mm diameters when fully deployed, which is a key feature in determining the risk of occlusion. A large prospective multicenter study randomized 241 patients with malignant biliary strictures to receive uncovered SEMS of different designs in two diameters (i.e., 6 mm Zilver, 10 mm Zilver, or 10 mm Wallflex). SEMS occlusions were much more frequent with a 6-mm diameter SEMS and equivalent in the two 10-mm arms despite major differences in stent design, material, and expansion, suggesting that diameter is the critical feature [68]. Similarly, Yang et al. showed no significant difference in the rate of occlusion when using uncovered SEMS of equal diameter, but different stent design [69].
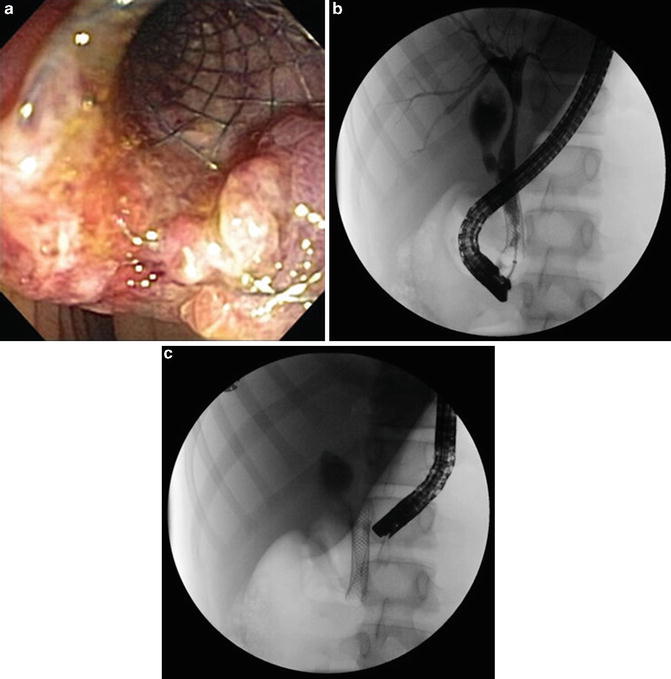

Fig. 4.10
(a) Endoscopic photograph demonstrating tissue overgrowth at the duodenal end of a biliary self-expandable metal stent. (b) Balloon occlusion cholangiogram revealing a biliary stricture caused by tumor or tissue ingrowth through the interstices of the existing metal biliary stent. (c) A second SEMS was deployed within the existing SEMS with resolution of biliary obstruction
Studies comparing the differences in patency rates between covered and uncovered SEMS in patients with malignant distal bile duct obstruction have shown conflicting results. For example, two randomized multicenter trials found no difference in patency rates [70, 71]. Another randomized trial showed longer patency with covered SEMS [72]. A meta-analysis concluded that covered SEMS have a significantly longer patency compared with uncovered SEMS [73]. However, a subsequent meta-analysis found no difference in patency between covered and uncovered SEMS at 6 and 12 months, although covered stents had a higher rate of stent migration [74].
Another concern for patients undergoing placement of a covered SEMS who have an intact gallbladder is the potential for developing cholecystitis due to obstruction of the cystic duct origin. Although the rate of developing cholecystitis as a complication after SEMS placement has been low in most studies, rates of up to 10 % have been reported [75, 76]. Some endoscopists routinely perform a biliary endoscopic sphincterotomy (B-ES) to facilitate SEMS placement and to help avert the risk of pancreatitis due to SEMS occlusion of the pancreatic duct. On the other hand, B-ES may itself be a risk factor for procedure-related complications including pancreatitis, bleeding, perforation, and stent migration. Studies comparing the outcome of SEMS placement in patients with and without a preceding B-ES have shown the following: (1) SEMS (covered and uncovered) may be placed without a B-ES with very high success rates equal to those who underwent B-ES prior to stent placement, (2) Avoiding a B-ES prior to SEMS placement may reduce the risk of complications, especially short-term complications such as bleeding and perforation [77, 78].
EUS-Guided Biliary Drainage
Despite a success rate of >90 % in most reports, ERCP with stent placement for malignant biliary obstruction occasionally fails owing to anatomical or technical problems. Surgically altered anatomy, gastric outlet obstruction, tumor infiltration of the ampulla, and periampullary diverticula may result in inability to reach or visualize the ampulla during ERCP. PTBD or surgical interventions are conventionally performed after unsuccessful ERCP. EUS-guided biliary drainage (EUS-BD) has recently emerged as an effective biliary drainage technique in cases of unsuccessful ERCP. Following the first report of EUS-BD by Giovannini et al. in 2001 [79], many groups have subsequently reported on the efficacy of EUS-BD as an alternative biliary drainage modality after unsuccessful ERCP [80–88]. EUS-BD is accomplished using one of three techniques. Transluminal biliary drainage involves accessing the common duct or a dilated left intrahepatic duct under EUS guidance, followed by dilation of the tract and placement of a stent between the common duct and duodenum (cholecystoduodenostomy) or the stomach and a left hepatic lobe duct (hepaticogastrostomy). The stent drains the biliary tree into the GI tract without crossing the site of biliary obstruction. In the EUS-BD rendezvous procedure, the biliary tree is accessed via the common duct or a left hepatic lobe duct and a guidewire is passed via the bile duct across the papilla into the duodenum. The EUS-placed duodenal guidewire is then used to perform ERCP in the usual retrograde fashion. It should be noted that the EUS guided rendezvous technique is possible only when the papilla can be reached endoscopically. With the EUS-guided antegrade technique, transgastric puncture of a dilated intrahepatic duct is performed followed by tract dilation and transpapillary placement a stent across the level of obstruction in antegrade fashion. The antegrade technique may be useful when the papilla cannot be reached endoscopically. EUS-BD is a technically complex procedure requiring advanced skills in interventional EUS. The overall success and complication rates are approximately 81 % and 15 %, respectively, in expert hands [47].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







