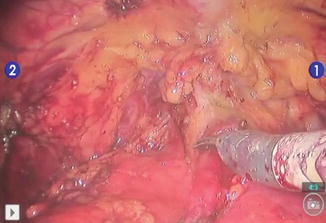
Fig. 8.1
Laparoscopic circumscription (a) of the tumor under endoscopic guidance (b)
Cautery lines are kept superficial until four 2–0 vicryl stay sutures have been placed lateral to the proposed resection area (Fig. 8.2).
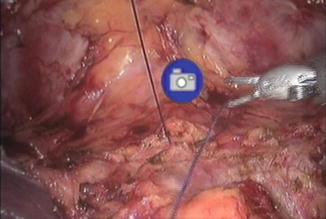

Fig. 8.2
Placement of stay sutures
The bladder is then drained and if possible (e.g., favorable anatomy, lobulated bladder, or bladder diverticulum), a 60-mm Echelon Endopath stapler (Ethicon Endo-surgery, Cincinnati, OH) is brought in through either the left or right 12 mm ports and used to divide the bladder at the proposed lines of resection (Fig. 8.3). If not amenable to the use of a stapler, scissors are used instead to cut sharply along the marked lines of resection with care not to spill bladder fluid/urine into the peritoneum. Pulling up on the stay sutures will facilitate the resection and decrease the risk of fluid spillage into the peritoneal cavity. Multiple specimens are also sent for intraoperative frozen section analysis to ensure negative margins have been achieved. Once negative margins have been confirmed, the specimen is then placed in an Endo-catch bag (Covidien, Mansfield, MA) and removed through an extended port incision at the end of the case.
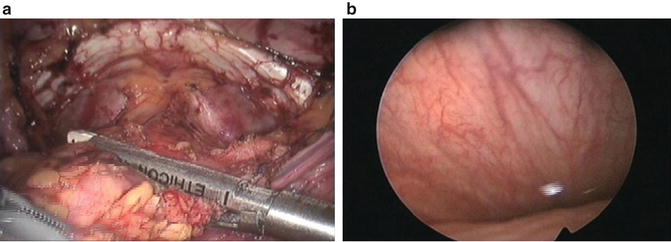

Fig. 8.3
Laparoscopic view (a) and endoscopic view (b) of bladder resection using Endo-GIA staplers
If the stapler has been used, the remaining suture line on the native bladder is excised while tension is maintained on the stay sutures to prevent any urine spillage and contamination of the peritoneal cavity. This maneuver removes the staples in the suture line that could potentially serve as a nidus for stone formation if left in place. The bladder is emptied completely prior to the resection. Once resected, it is sent for histopathological analysis as the final margin. The bladder is then closed in two watertight layers in a running fashion using 3–0 monocryl/vicryl for the mucosal layer and 2–0 monocryl/vicryl for the outer layer. Moreover, the bladder could be closed in a full thickness, continuous fashion using 2–0 unibarbed V-loc suture (Covidien, Mansfield, MA) [19].
Subsequently, the bladder is tested for any leakage by filling with 250 ml of normal saline while being monitored cystoscopically and laparoscopically (Fig. 8.4). A JP drain is also placed via the 5-mm port.
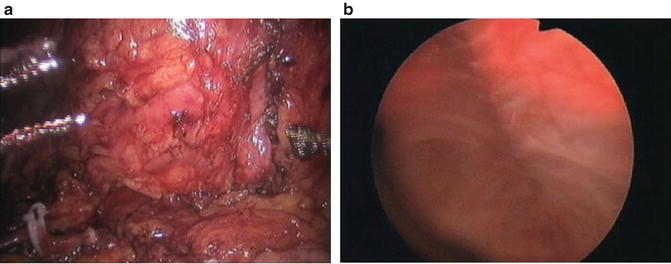

Fig. 8.4
Laparoscopic (a) and endoscopic (b) monitoring of testing of bladder closure
Finally, bilateral pelvic lymphadenectomy is performed as previously described in other sections of this book.
Postoperative Care and Follow Up
The following standard postoperative care is applied toward all PC, VSRC, and PSRC patients. In our experience, PC patients normally remain hospitalized on average for 1–2 days; while those who undergo VSRC and PSRC remain hospitalized longer (average of 5–6 days). The nasogastric or orogastric tube is typically removed immediately after surgery. Patients are allowed then to chew gum and have ice chips, and their diets are gradually advanced over the course of their hospitalization. Intravenous antibiotic prophylaxis is maintained for at least the first 24 h after surgery, while DVT prophylaxis with subcutaneous heparin is continued after surgery and for the duration of the hospitalization as long as hematocrit levels remain stable. Additionally, patients are encouraged to ambulate as soon as possible, preferably on postoperative day #1. Pain control is initially achieved with ketorolac with IV narcotics for severe breakthrough pain and then quickly converted to oral medications once the patient is tolerating a diet. Daily chemistry and hematocrit levels are routinely checked for the first 48 h and if stable, are then obtained at the surgeon’s discretion. JP drain is typically removed after output is minimal and creatinine level of the fluid is consistent with serum creatinine. Urethral foley catheter can be removed in 7–13 days following a normal cystogram.
Follow-up for these patients is dictated by the pathology. Typically, these patients are followed closely every 3–6 months for the first 2 years with history & physical (H&P), voided cytology, labs (CBC, Chemistry, and LFT), and cystoscopic evaluation. Abdominal-pelvic cross-sectional imaging and chest radiographs are dictated by the pathology of the disease and standard NCCN recommendations are followed.
Discussion
Historically, PC had been advocated as a viable alternative to radical cystectomy due to its technical simplicity, decreased perioperative morbidity, and preservation of urinary and sexual functions. However, its popularity was short lived because of its high local rate of recurrence, ranging from 38 to 80 % [20], and low overall 5-year survival [17]. Suboptimal patient selection coupled with advances in surgical techniques associated with radical cystectomy, such as nerve-sparing procedures and continent reservoirs, all contributed to its downfall.
Nevertheless, as society ages and survival from bladder cancer increases, there has been a strong paradigm shift toward improving the quality-of-life issues without sacrificing oncological efficacy. This rekindles the interest in bladder-preserving procedures to be used in the primary setting or as part of a multimodal approach. To avoid the same pitfalls, many experts advocate for stricter selection criteria that include solitary, primary tumors located far away from the ureteral orifices or bladder neck and in an easily resectable area that allows for an adequate resection margin. Additionally, tumor multifocality and the presence of concomitant CIS must be ruled out.
In a retrospective study of 58 patients who had undergone PC from 1995 to 2001, Holzbeierlein et al. reported an overall 5-year survival rate of 69 % at a mean follow up of 33 months and local recurrence rate of only 19 % [16]. Univariate analysis demonstrated that tumor multifocality and the presence of concomitant CIS were significant predictors of recurrence. Similarly, in another retrospective study of 37 patients with a mean follow-up of 72.6 months, Kassouf et al. reported the overall 5-year, disease-specific, and recurrence-free survival rates to be 67 %, 87 %, and 39 %, respectively [21]. On multivariate analysis, higher pathological stage was associated with shorter overall recurrence-free survival; whereas, adjuvant chemotherapy was associated with prolonged advanced recurrence-free survival. In another retrospective study by Fahmy et al. looking at 714 patients with muscle-invasive bladder cancer who had undergone PC from 1983 to 2005 among different institutions, the 5-year overall survival between PC and radical cystectomy groups were similar (49.8 vs. 51 %). At a median of 17.6 months, 23.7 % of these patients recurred and required salvage radical cystectomy [22]. However, these patients had a 50 % increased risk of dying compared to those who underwent radical cystectomy initially.
These selected publications and others all emphasize the importance of optimal patient selection to achieve good oncological control. In carefully selected patients, 5-year overall survival is similar to that of radical cystectomy plus the benefits of decreased morbidity and preservation of urinary and sexual functions. It’s worth mentioning that due to the paucity of data, no differences in overall survival, local recurrence rate, and functional outcomes have been demonstrated between different PC approaches [laparoscopic (robotic) versus open]. Rather, the decision to select a specific approach depends on the surgeon’s experience and comfort level. However, with the success achieved in robot-assisted prostatectomy, the authors anticipate similar outcomes in robot-assisted PC.
Vaginal-Sparing Radical Cystectomy
Indications
Successes of female radical cystectomy have largely been measured by oncological and urinary outcomes, with little regard to sexual outcomes. Zippe et al. [23] reported that up to 52 % of female patients experienced sexual dysfunction after RC and that the nature of the dysfunction encompassed both organic and psychosocial domains, such as decreased lubrication, decreased orgasm, lack of sexual desire, and dyspareunia. In a recent review of the literature, Elzevier et al. [24] reported that female sexual dysfunction rate after RC ranged from 20 to 82 % with no difference among the types of urinary diversion. As a result of these data, there has been an increased interest in modifying current surgical techniques to improve sexual outcomes. Anatomic studies have localized the neurovascular bundles to be along the lateral walls of the vagina [25]. Additionally, the removal of the distal urethra is associated with significant devascularization of the clitoris, which could adversely impact sexual arousal and orgasm [26]. Armed with this knowledge, some experts have modified their surgical approaches to include techniques of vaginal-sparing, ovary-sparing, urethral-sparing, neurovascular preserving, and tubular vaginal reconstruction [27–31].
However, the benefits of these organ-sparing approaches must be weighed against the risks of compromising oncologic outcome. Many studies have reported the incidence of urothelial cancer involvement of internal genitalia (vagina, uterus, and ovaries) to be between 2.6 and 5 % and the risk of having concomitant primary genital malignancy to be low as well [32–34]. Therefore, at our center, we only perform vaginal, ovary-sparing, and neurovascular preserving robot-assisted radical cystectomy in those patients who meet the following criteria.
Good performance status (ECOG ≤ 2) with manual dexterity and willingness to self-catheterize neobladders if needed.
Non-obese patients (BMI <30) with minimal comorbidities as these could restrict the patient’s cardiopulmonary tolerance of the surgery.
No previous intra-abdominal/pelvic surgeries or prior pelvic radiotherapy.
Demonstrate T2 disease or better with non-bulky tumors.
No gynecologic malignancy such as cervical or ovarian cancers.
Are sexually active with intentions to continue after surgery.
Steps of the Procedures
Port Placement, Establishment of Pneumoperitoneum, Ureteral Mobilization, Posterior Dissection, Control of Round Ligaments, Hysterectomy, and Control of Bladder Pedicles
These steps are as would be performed during anterior pelvic exenteration or female cystectomy and are described in detail in other sections of this book.
Dissection of Vesicovaginal Space
During the posterior dissection step as described in other sections of this book, antegrade dissection through the cul-de-sac allows for the separation of the posterior bladder from the uterus. This dissection plane is carried as far posteriorly as possible, preferably to the junction of the corpus uteri and cervix. The superior portion of the sacro-uterine ligaments along with the round ligaments is transected. However, the cardinal ligaments that attach to the lateral walls of the vagina along with the ovaries are left intact to maintain support to the vagina and to preserve hormonal function, respectively. Next, the bladder is dropped in a standard fashion similar to that of robotic prostatectomy and is described in other section of this book. During this step, the endopelvic fascia is identified and opened to expose the dorsal venous complex (DVC), urethrovesical junction, and lateral walls of urethra. The DVC is controlled using 0-vicryl suture and transected. Using both blunt and sharp dissection, a space between the urethra and the anterior wall of the vagina is created. Control of the bladder pedicles are achieved with Hem-o-lock clips (Teleflex Medical, Research Triangle Park, NC) or a stapler. It’s important to know that while the vagina receives its arterial blood supply from multiple sources, vaginal branches from the uterine and inferior vesical arteries are among the important contributors. Additionally, since these branches travel in close proximity to the nerve supply of the vagina and clitoris, it’s important to spare them during this step to avoid potential devascularization and denervation.
Using the lateral sulci of the vagina and lateral walls of the urethra as landmarks, dissection of the vesicovaginal space is performed sharply with minimal monopolar coagulation to prevent inadvertent thermal injury to the neurovascular bundles. The authors typically utilize a sponge stick dipped in betadine and inserted into the vaginal vault to help identifying the vaginal apex and anterior vaginal wall during the course of dissection. Hemostasis is obtained with pinpoint monopolar coagulation and suturing. Once the vesicovaginal space is fully developed, the urethra is encountered and transected.
Vaginal Stump Fixation: Variation of the Mansoura Technique
In 2002, Ali-El-Dein et al. [35] introduced the Mansoura modification in hope of preventing postoperative chronic urinary retention or hypercontinence that frequently plagued those who had received a neobladder. The authors demonstrated that by attaching the preserved ends of the round ligaments to the vaginal stump, this effectively fixed and provided support to the vaginal vault as well as prevented posterior and caudal displacement of the neobladder as demonstrated by cystogram. This has resulted in a reported 55 % reduction in the incidence of urinary retention among their patients. Armed with this knowledge, we also perform vaginal stump fixation among all patients who will receive a neobladder. During the standard hysterectomy as described in other section of this book, transection of the round ligaments is made close to its origins in the uterine horns to ensure adequate length for the vaginal fixation. Closure of the vaginal stump is performed horizontally with 2–0 vicryl to prevent narrowing of the vagina. The free ends of the round ligaments are then sutured to both ends of the vaginal apex, in effect suspending and supporting the vagina.
Bilateral Extended Pelvic Lymphadenectomy and Orthotopic Urinary Diversion
These steps are similar to those that have been described in detail in other sections of this book.
Postoperative Care and Follow Up
Postoperative care for VSRC is similar to that of PC except that these patients would have undergone an orthotopic urinary diversion. The ureteral stents of the neobladder are removed at approximately POD #10 and JP drain is maintained to drain any potential extravasation of urine as a result. A pouchogram is performed on POD #14 and if no leakage is noted, both the JP and urethral Foley catheter are removed subsequently. The patient is taught intermittent catheterization and pouch irrigation. Follow-up is similar to that of PC as described above and is also dictated by the pathology. More importantly, patients should continue to follow up with their Ob–gyn for periodic vaginal cytology and gynecologic examination.
Discussion
Standard female exenteration routinely requires the removal of the internal genitalia (vagina, uterus, and ovaries) along with the bladder. However, surgical modifications sparing these internal organs allow for the preservation of fertility and hormonal functions as well as improvement of functional outcomes. Multiple studies have demonstrated the low incidence of recurrence and concomitant urothelial cancer involvement of these organs. In a retrospective review of 609 female radical cystectomy specimens, Ali-el-dein et al. [34] reported the gynecologic organ involvement to be 2.6 % (16/609). Furthermore, no local vaginal recurrence was found at a mean follow-up of 4.3 years. Similarly, Salem et al. and Varkarakis et al. [32, 33] reported the incidence to be 4.4 % and 5.7 %, respectively. Vagina was most commonly involved, with the exception of one uterus. No vaginal recurrence and major sexual problems were encountered at the last follow-up (mean 6 years).
In terms of functional outcomes, it is believed that preservation of the vagina may decrease the risk of neobladder–vagina fistula and improve incontinence by preventing the posterior displacement of the neobladders [35, 36]. Koie et al. [37] reported 80 % (24/30) complete dryness (day and night continence) in those who had undergone vagina-, uterus-, and ovary-sparing radical cystectomy. Likewise, Chang et al. [28] reported 72 % continence rate (daytime and nighttime) in 15 out of 21 patients who had undergone vagina-sparing radical cystectomy. The results of these selected studies demonstrate that successful outcomes can be achieved without compromising oncological control in carefully selected patients.
Prostate-Sparing Radical Cystectomy
Indications
Since its first description, radical cystectomy has gone through various modifications as a means of improving postoperative continence and potency rates. Rossetti et al. [38] first described supra-ampullar cystectomy techniques by sparing the vasa deferentia, seminal vesicles, and prostatic capsule. Spitz et al. [39] reported the first US series of modified radical cystectomies that preserved the vasa deferentia, seminal vesicles, posterior prostate, and most importantly neurovascular bundles in four young men who had non-urothelial malignancy. These and many other series have in common the attempt to minimize dissection near the neurovascular bundles and urinary sphincter. Initially, PSRC was recommended for men without a primary urothelial malignancy. Successful functional outcomes in these patients sparked interest in applying this approach to those patients with primary urothelial malignancies. However, concerns for long-term oncological efficacy of this approach have been questioned. While the exact selection criteria have not been agreed upon, what has been known is that successful oncologic outcomes of PSRC rest mainly with optimal patient selection. Typically, these patients undergo PSRC in conjunction with an orthotopic neobladder. As such, we believe that optimal candidates should include the followings based on the data in the literature, which is discussed in the later section:
Young, healthy, and potent patients whose potency and fertility remain a priority.
Good manual dexterity with a willingness to self-catheterize neobladders when needed.
Demonstrate clinical T2 disease or better without bladder neck, prostatic urethral involvement, or multifocal CIS disease.
Absence of prostate cancer based on low serum prostate-specific antigen (PSA) levels, negative digital rectal exam (DRE), and negative standard transrectal ultrasound (TRUS)-guided prostate biopsies.
Steps of the Procedures
Standard port placement, pneumoperitoneum establishment, ureteral mobilization, bladder mobilization, control of bladder pedicles, and extended pelvic lymph node dissection are meticulously described in other sections of this book.
Bladder Neck Dissection
After ureteral and posterior bladder mobilizations, the bladder pedicles are carefully controlled as they come off the internal iliac artery using an athermal technique with Hem-o-lock clips (Teleflex Medical, Research Triangle Park, NC) or a stapler to prevent inadvertent thermal injury to the prostatic neurovascular bundles. The dissection is carried towards the prostate base, and only the internal iliac pedicles and superior vesical arteries are clipped and divided while the inferior vesical arteries along with its prostatic branches are spared to promote sexual potency recovery. The bladder is then dropped in a fashion similar to robotic prostate surgery and as described in other chapters of this book. To expose the prostatic–vesical junction in preparation for the bladder neck dissection, one must first develop the lateral pelvic spaces as previously described in sections on developing the endopelvic space on either side of the prostate. Minimal to no dissection is done at the apex of the prostate. Once completed, this allows for the identification of levator ani muscles on the lateral pelvic side wall and the lateral and posterior bladder walls medially. “Defatting” of the prostatic–vesical junction can be made with a combination of blunt dissection and point cautery. Occasionally, an accessory pudendal artery is encountered and should be spared. Additionally, once the prostatic–vesical junction is cleanly exposed, the bladder neck is dissected circumferentially (Fig. 8.5) and ligated to prevent tumor spillage as it is being dissected (Fig. 8.6). The bladder is transected at the bladder neck and placed in an Endo-catch bag (Covidien, Mansfield, MA) which will later be removed with an extended midline port incision.