Fig. 27.1
Perineal Crohn’s disease in an 8-year-old male showing multiple fistulas
Hemorrhoids
Key Concept: Hemorrhoids are normally from constipation and should almost always be treated nonoperatively.
Hemorrhoids are extremely uncommon in children, with the prevalence increasing in older kids and teens [12]. Symptoms are similar to adults including bleeding, prolapse, itching, pain, blood-streaked stools, and a bulge or dilated venous plexus at the anal orifice. Treatment in children is usually conservative with diet modification, increased liquid intake, and sitz baths. Laxatives can also be helpful, and glycerin suppositories work well for younger children. The vast majority of hemorrhoids resolve with medical treatment for constipation alone. Operative intervention is reserved for the rare failure and consists of the procedures as adults [12].
Anal Fissure
Key Concept: Anal fissure presentation and treatment is similar to adults.
While anal fissures can occur at any age, they most commonly present around 2 years old [12]. Symptoms are similar to adults, with rectal pain, bleeding, streaked stools, as well as crying with defecation. The lesions in infants may be found anywhere, although they are more common in the posterior midline. Females, similar to adults, more commonly have anterior midline fissures [12]. If the lesions are off the midline or are multiple, consider a workup for Crohn’s disease or a culture and/or biopsy to exclude malignancy, tuberculosis, venereal disease, or immunodeficiency in the appropriate setting. Recognize, however, that most anal fissures are secondary to constipation and will heal spontaneously with a simple focus on maintaining adequate fluid intake along with a proper bowel regimen, and that more intensive will only be required in a small subset of patients. Similar to adults, glyceryl trinitrate 2 % (GTN) has been associated with improvements in fissure healing and symptom relief compared with lidocaine or placebo in both the short term (10 days) and at 8 weeks [12]. Healing rates have been up to 84 %, with symptomatic relief reported in 91–94 % with GTN [12–15]. Botulinum toxin can also be used for resolution of anal fissure, though most data consists of case reports. Keshtgar showed in their study that 4 children who underwent treatment for anal fissure with transcutaneous botulinum toxin had resolution of their lesion [16]. Finally, successful use of lateral subcutaneous sphincterotomy has also been described in children, with a complete resolution of all fissures in small series [12, 17].
Our approach is as follows: Asymptomatic lesions are likely to resolve and should be managed expectantly with dietary and fluid management. If intervention is necessary, an initial trial of topical therapy (lidocaine, GTN) should be used. Recurrences can be treated medically (GTN is not used or botulinum toxin), with surgery reserved for continued failures. You should remember that any surgical intervention involving a small child or infant should be done so with the most extreme caution, as incontinence and anal stenosis are reported in up to 30 % [12].
Rectal Prolapse
Key Concept: Rectal prolapse in children is most commonly secondary to constipation, and the majority will resolve with treatment directed towards proper bowel habits. Surgery should be reserved only for refractory cases with severe symptoms.
Rectal prolapse is a common problem in children, with a peak incidence in the first year of life and most occurrences taking place under 4 years old around the time of toilet training [18–20]. Prolapse in this cohort is a symptom of an underlying condition, usually constipation, but can also be caused by increased abdominal pressure (i.e., chronic cough), acute or chronic diarrhea, parasites, neoplastic disease of the rectum, malnutrition, cystic fibrosis, or pelvic floor weakness [12]. While the exact etiology is unknown, the rectosigmoid in children with prolapse tends to be ~1.5-fold longer than their normal counterparts [21].
Most cases are mild and typically resolve spontaneously shortly after the rectum prolapses [18, 19]. You should use manual reduction prior to the onset of edema for an acute prolapse that does not reduce on its own. Firm steady pressure may be necessary to decrease the swelling and allow reduction. Importantly, perform a digital examination to ensure the reduction is complete, and exclude any distal source of pathology. Another helpful tip in children is to approximate the buttocks with adhesive tape for several hours to reduce recurrence [18]. Roughly 90 % of rectal prolapse cases in children under the age of 4 will resolve with these simple steps alone and rarely continues after 6 years old [18]. Parents should be instructed to use stool softeners and/or laxatives, while avoiding prolonged straining and ensuring the use of a proper toilet size for their child to prevent recurrent episodes. We also tell parents to promptly reduce a prolapse if it were to reoccur and to bring the child in for reevaluation.
Children over 4 years of age are more likely to have neurologic (i.e., spinal cord lesions) or muscular defects and require intervention. You should also consider screening for cystic fibrosis (CF), as up to 11 % of children with rectal prolapse will have CF [22]. Indications for surgery include failure of conservative measures, continued prolapse after 1 year despite nonoperative therapy, greater than 2 episodes requiring manual reduction, or chronic pain, bleeding, and perianal excoriation from the recurrent prolapse [18]. The simplest, most benign, and still efficacious, intervention is sclerosing injections. Several different agents have been used with efficacy rates ranging from 83 to 100 %, depending on the sclerosant (i.e., 30 % saline, 5 % phenol in almond oil, ethanol 70 %) and number of treatments [23–28]. However, this should be used with caution, as reported complications include temporary fecal incontinence, temporary limping, bleeding, perirectal inflammation, urinary retention, necrosis of the rectal mucosa, and abscess formation [23, 28].
Both perineal and abdominal approaches are available for refractory cases; however, similar to adults, no single operation has been shown to have significantly better results [20, 22]. Although the Thiersch procedure is not used much in adults anymore, it may be ideal for select children with prolapse secondary to weak pelvic floor muscles or those associated with cystic fibrosis [18]. A less invasive procedure somewhat unique to children (and not used extensively anymore) is linear cauterization, which has a reported success rate up to 94 % [29]. Some institutions also advocate a combination of these techniques, with linear cauterization used in conjunction with sclerotherapy injection and a Thiersch procedure [20].
Although you may not have much experience in any of these, other procedures you may hear include posterior sagittal transsphincteric rectopexy, Ekehorn’s rectosacropexy (placing a U-shaped suture through the rectal ampulla and tying the suture outside at the sacrococcygeal junction), and a transcoccygeal rectopexy with puborectalis plication. Success and recurrence rates remain highly variable in limited series [30–32]. More commonly used procedures in adults, such as the Altemeier perineal proctosigmoidectomy, have also been associated with good results in the pediatric population.
Laparoscopic approaches such as suture rectopexy, posterior mesh rectopexy, and resection of the sigmoid colon with or without rectopexy have also been associated with good outcomes in children [21]. Recurrence rates are ~5 %, with failures perhaps higher in children with cystic fibrosis [33]. In general, we do not use prosthetic mesh initially in the pediatric population. Adding a sigmoid resection with rectopexy in children has been also controversial, although it has been shown to be safe, eliminates the risk of volvulus, and has a low morbidity and low recurrence rate [21]. Finally, similar to adults, the addition of a resection is preferred in those with intractable constipation and prolonged transit studies.
Constipation
Key Concept: Similar to adults, constipation therapy should be guided by a thorough history and physical examination, with initial dietary, fluid, and nonoperative management. Select diagnoses with severe nonresponsive symptoms may be considered for operative intervention.
Constipation is a common problem in childhood, affecting 7–30 % of children [34]. You should remember that constipation is a symptom rather than a diagnosis, manifesting with painful defecation, difficulty with passage of stools, or decreased stool frequency [35]. It is a major problem, responsible for ~3 % of visits to outpatient clinics and up to 20 % of pediatric gastroenterological complaints [35]. When not treated adequately, constipation can lead to psychological problems, low self-esteem, withdrawal, and social isolation [36–38].
Evaluation
Key Concept: History and physical examination guide most of the evaluation, with ancillary testing used mostly for directed findings, difficult cases, or those who have nonresponsive severe constipation.
Functional constipation is the underlying reason in more than 95 % of children [39], with the diagnosis confirmed by having two or more of the symptoms listed in Table 27.1 [38]. A thorough history and physical examination is typically sufficient to rule out most pathological causes, and extensive additional testing prior to medical and dietary treatment is unnecessary (Table 27.2) [38]. Similar to adults, a comprehensive history should include the frequency and character of the stools, withholding behavior, timing of incontinence, straining, and pain with defecation. Children who present with constipation under 6 months of age, especially who have delayed passage of meconium greater than 48 h after birth, or those who present with abdominal distention and refusal to feed are concerning symptoms for Hirschsprung’s disease, anal stenosis, or ectopic anus (Table 27.3) [35, 39]. Other concerning symptoms that should prompt you to consider a more extensive workup are accompanying urinary tract infections, weight loss, neuromuscular development, and psychological or behavioral problems (Table 27.2) [39].
Table 27.1
Rome III criteria for pediatric functional constipation
Two or fewer defecations per week |
At least one episode of fecal incontinence per week |
Stool retentive posturing |
Painful or hard bowel movements |
Large diameter stools that could obstruct the toilet |
Presence of large fecal mass in the abdomen or rectum |
Organic causes |
History: delayed passage of meconium (>48 h after birth), early onset of symptoms (<6 months of age), bilious vomiting, bloody stools, ribbon like stools, delayed or abnormal development, no withholding, no response to conventional treatment, and extraintestinal symptoms (fever, fatigue, eczema, bladder disease, neurological disturbances, oral ulcerations) |
Physical exam: failure to thrive, fever, absent cremasteric reflex, abdominal distension, abnormal position of anus, perianal fistula, absent anal wink, tuft of hair on spine, deep sacral dimple, decreased lower extremity strength/tone/reflex, asymmetry of the lumbosacral region or flattening of the gluteal muscles/lower limb, spina bifida, and explosive diarrhea after rectal exam |
Sexual abuse |
History: smear feces |
Physical exam: extreme fear during anal inspection or rectal examination, anal scars, fissures, and hematomas |
Table 27.3
Causes of constipation in children
Idiopathic constipation (most common) |
Dietary |
Inadequate fluid/fiber intake |
High milk intake |
Dietary protein allergy |
Cow’s milk allergy |
Anal/rectal disorders |
Anal stenosis, anterior ectopic anus |
Anal fissure (acute, chronic) |
Perianal group A streptococcal infection |
Eosinophilic proctitis |
Megarectum |
Neurogenic abnormalities |
Sacral dysgenesis (anorectal anomaly) |
Myelomeningocele (spina bifida) |
Spinal cord abnormalities/trauma |
Static encephalopathy |
Tethered cord |
Hirschsprung’s disease |
Intestinal neuronal dysplasia |
Cerebral palsy |
Endocrine and metabolic disorders |
Hypothyroidism |
Hypercalcaemia |
Renal tubular acidosis |
Diabetes mellitus |
Hypokalemia |
Vitamin D intoxication |
Medications |
Opioids |
Anticholinergic agents |
Antidepressants |
Phenytoin |
Phenothiazine |
Abnormal defecation dynamics |
Fear and withholding habit |
Pelvic floor muscle dyssynergia |
Other |
Anorexia nervosa |
Sexual abuse |
Scleroderma |
Cystic fibrosis |
Celiac disease |
In addition to a complete abdominal and perineal examination, differences in the examination for pediatric patients should involve looking for other signs of congenital syndrome anomalies (i.e., VACTERL) and ensure there is fecal impaction or anal stenosis [13]. A fecal mass either found in the abdomen or rectum is present in 30–70 % of children with constipation [16]. Investigations for children may include a basic metabolic panel to exclude electrolyte imbalance and thyroid or celiac panels to work up patients with severe unremitting constipation. You should note that most laboratory investigations rarely uncover an underlying disease [35, 38]. Plain abdominal x-rays are seldom helpful to assess fecal retention, and actually correlate poorly between symptoms and pathology, leaving more open to misinterpretation [39, 40]. For children over 12 months of age, anorectal manometry can be extremely valuable when Hirschsprung’s disease is in the differential diagnosis. Failure of relaxation of the internal sphincter is an absolute indication for rectal biopsy and has a high positive predictive value for the absence of ganglion cells [35]. Furthermore, in any infant or other patient in whom Hirschsprung’s disease is suggested, a rectal biopsy is the gold standard for diagnosis. You should remember that short-segment Hirschsprung’s disease may manifest later in childhood (or even adult life) with constipation, and work up those select patients accordingly. While ultrasound has been suggested to look at fecal load in the colon, it really is not very useful. This is similar to the colon transit studies, where some authors feel it is helpful [37]. One could argue that in children with normal transit time, surgery is not typically indicated [41]. Others suggest it aids in determining the point at which normal peristalsis is lost [39, 42]. However, we rarely find it useful in the primary workup for constipation. Finally, though the indication for endoscopy for primary constipation symptoms is rare, high-risk or concerning symptoms should direct the need accordingly.
Treatment
Key Concept: Use medical management and dietary changes as first–line therapy—they will work most of the time.
Years of experience and expert opinion guide most of constipation management. We find that the most useful first step is often helping the patient and family to understand the problem and enlist their cooperation and compliance in what is often a trial and error period [36, 38]. Interestingly, there is an association between not only low-fiber intake and constipation but also a positive family history, obesity, and low levels of parental education with an increased prevalence of childhood constipation [38, 43]. The American Academy of Pediatricians suggests a balanced diet that includes whole grains, fruits, and vegetables as the first-line treatment of constipation; however, when comparing fiber supplementation to placebo, there was no significant difference in defecation frequency or resolution of symptoms [36]. We feel there is very little downside, so we agree with this initial approach, along with ensuring adequate fluid intake and physical activity [44]. Children with a developmental age of at least 4 years should also be instructed to have dedicated toilet time for 5–10 min after each meal (three times per day) to encourage regular toileting [45].
If you encounter children who have severe constipation marred by fecal impaction and/or overflow incontinence, disimpaction can be accomplished using either oral or rectal therapies [44–48]: high-dose PEG 3350 (1.5/g/kg/day, max dose of 100 g/day) orally for outpatient use and milk of molasses enemas (mixed 1: 10 mL/kg with maximum of 500 mL) followed with maintenance PEG 3350 have been compared with relatively similar results beyond the first day [47]. In general, enemas relieve symptoms faster than the oral treatment, but either approach is feasible. Other authors have shown that PEG with electrolytes is more effective than suppositories, rectal enemas, or even manual evacuation [49]. If you would like to avoid rectal therapy or if oral disimpaction fails, you may need to take the child to the operating room for disimpaction under general sedation.
Maintenance treatment must be initiated immediately and may be necessary for several months. You should discontinue this treatment only after there are regular bowel movements for several weeks without effort and the child is toilet trained [34, 44]. Tailored therapy is best and you can choose from a variety of laxatives. Unlike adults, laxatives cannot be assumed to work in a similar manner in children due to the fact that the etiology for childhood constipation is typically different than that of their adult counterparts [36, 38]. PEG with electrolytes is our preferred agent and has been shown to be more effective in achieving treatment success compared to other laxatives (lactulose and milk of magnesia) [50–52]. There is also evidence suggesting that mineral oil is efficacious, though chronic use should be avoided, as it has been associated with vitamin deficiencies [52]. Other agents such as probiotics may be helpful in the treatment of functional constipation, but there is very little data available, and we withhold recommendation. Behavioral therapy alone or biofeedback should not be the sole treatment of constipation in children; however, there is some evidence that behavioral intervention plus laxative therapy improves continence in this age group [38, 44].
Surgery: Sphincter Procedures, Antegrade Continence Enema, and Stoma
Key Concept: Several surgeries somewhat unique to children are available for symptomatic severe constipation that is nonresponsive to medical management.
Surgical options for constipation depend in large part on the underlying etiology. Procedures include anal dilation, intra-sphincteric injection of botulinum toxin, antegrade continence enema (ACE), excision of colon and/or megarectum and diversion, and reconstructive proctocolectomy [35, 53]. While (as previously stated) transit studies and total colonic manometry don’t play a major role in the initial evaluation of constipation, we do use them prior to performing a definitive surgery. We find its major use is to ensure the distal-most segment of the colon or remaining rectum will have a functional motility pattern that will effectively allow for the passage of stool [42]. We also recommend a period of inpatient or close home observation to document objective evidence in order to help identify misreporting and exaggerating symptoms and thus avoiding unnecessary operations [53].
Anal dilation and myomectomy of the internal anal sphincter have been performed with the thought that children with constipation have hypertrophy of their internal anal sphincter, and anal dilation would decrease sphincter tone and allow for painless defecation [35]. Unfortunately two double-blinded randomized controlled trials have shown that anal dilation does not benefit children with chronic constipation [35], and we do not generally advocate its use. Myomectomy of the internal anal sphincter has been associated with weakening of the sphincter in subsequent years, and again, we are not strong proponents. Botulinum toxin sphincter injection has reported rates of symptom resolution in up to 94 %, without the long-term problems [16]. While this is generally well tolerated, ~20 % may require repeat injection [16].
Antegrade enemas are a therapeutic option for children who have intractable constipation or fecal incontinence and have failed either (a) a maximal medical management or (b) a sphincter procedure [35, 38, 53, 54]. Originally described by Malone in 1990 [54], subsequent modifications include an appendicostomy placed in an open or laparoscopic fashion, the cecum or rectum reconstructed to create a continent conduit, or a percutaneous cecostomy tube or button placed laparoscopically or open (Fig. 27.2) [35, 54]. The concept remains the same: to deliver antegrade enemas to clean out the entire colon to reduce soiling, with reported success rates of 52–92 % [53]. Each procedure has their own complications including stenosis, prolapse, skin infections, granulation tissue formation, leakage, and prostheses dislodgment [16, 19]. Results do not seem to differ in the complications or outcomes between a right and left ACE [55]. Ultimately, most children with idiopathic constipation can progress to a point they use no oral medications, and the stoma can be no longer used or taken down.
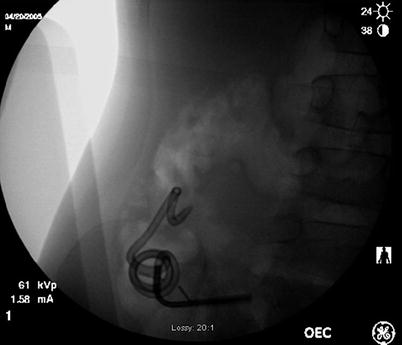

Fig. 27.2
Plain film of an antegrade cecostomy tube in place
Patients with Hirschsprung’s disease should be managed by the appropriate surgical therapy, which is most often a pull-through procedure that can be performed open or laparoscopically (Figs. 27.3, 27.4, and 27.5) and is one or several stages. One rare, but obviously significant, complication you may see is in children with chronic constipation; a small percentage may develop dilation of the rectum and sigmoid (megarectum). Resection of the dilated segments may be performed in the most extreme cases that have failed all other options, including ACE [53, 57]. Unfortunately, this rarely results in a complete or immediate cure, as children can still have fecal incontinence or constipation and may still need further resection [38]. Yet, almost all will be able to reduce their laxative need, and roughly half will be able to eliminate their laxative requirement altogether [56]. Finally, when combined with the physiological manometric data, the need for permanent stomas secondary to failure for these procedures can be reduced [20].
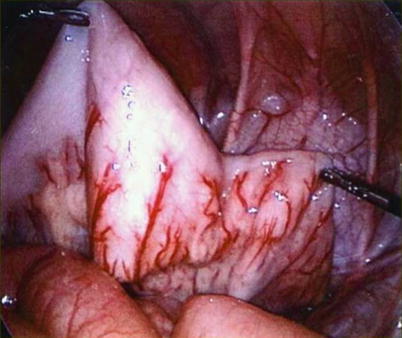
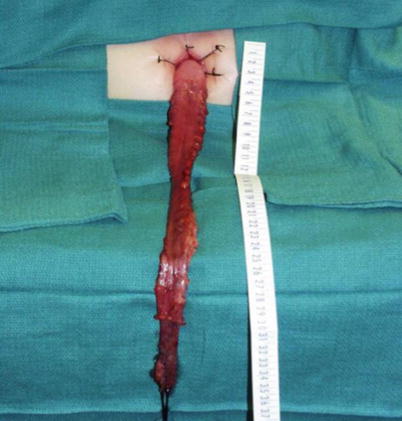
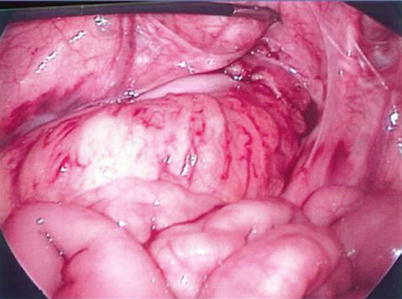

Fig. 27.3
Abdominal view at the beginning of laparoscopic-assisted pull-through procedure. The transition from dilated/normal sigmoid colon (right on the image) to contracted/aganglionic colon (left on the image). We will typically perform a few submucosal biopsies to confirm the presence of ganglion cells (Courtesy of David Gourlay, MD)

Fig. 27.4
Mobilized aganglionic sigmoid-rectum pulled through the anus. We have determined the level where ganglion cells are present (pictured here close to the anus). After transecting the aganglionic segment, a hand-sewn coloanal anastomosis is performed above the dentate line (Courtesy of David, Gourlay, MD)

Fig. 27.5
Abdominal view of the completed pull-through (Courtesy of David Gourlay, MD)
For those patients with severe problems that are nonresponsive and significantly affect their quality of life, a stoma can be performed. In this case, we prefer to use a temporary diverting loop ileostomy in hopes the child will wish to consider alternative treatments [16]. Finally, there are case reports of a restorative proctocolectomy performed in this final group with generally good results; however, we have little experience with this and feel it should not be undertaken lightly [57].
Incontinence
Key Concept: Fecal incontinence in children encompasses both encopresis (the expulsion of a normal bowel movement in inappropriate places in someone 4 years or older developmentally) and soiling (involuntary leakage of small amounts of stool), with the difference arising from the quantity of feces lost.
Kids with fecal incontinence have significant social consequences (like adults) such as restriction of social activities, as well as an increase in behavioral problems, and lower educational levels than expected when compared to their continent counterparts [59–62]. There are five main groups of children with fecal incontinence: constipation with fecal retention and overflow incontinence, functional non-retentive fecal soiling, anorectal malformations, spinal problems, and sphincter damage or dysfunction [59, 60]. The pathophysiology and treatment strategies differ for each subgroup. While we understand that you may not be the one primarily managing these patients, we will attempt to give you a brief overview and some general evaluation and management tips.
Functional Retentive Overflow Incontinence
Key Concept: Functional fecal retention with overflow incontinence is the most common cause of fecal soiling. Treatment should be aimed at treatment of constipation.
Constipation can lead to a vicious cycle that begins with painful defecation and ends in voluntary withholding and accumulation of stool in the rectum [16]. With chronicity, the rectum may become dilated, and sensation is gradually lost; therefore, when the softer stool arrives from the more proximal colon, it cannot be accommodated and leaks around the bolus of hard stool [46]. Because of the lack of sensation, this passage of soft stool is not sensed until the incontinence has occurred. This functional problem is typically self-limiting but requires aggressive treatment of constipation [59, 60].
Functional Non-retentive Fecal Soiling
Key Concept: This is a subgroup of patients who lack a history or physical exam findings consistent with constipation or functional fecal retention, but who have evacuation of large bowel movements in their underwear at least once a week. Treatment should be aimed at toilet training and positive feedback.
Confounding things, these children often have daily bowel movements in the toilet with near complete evacuation of stool and normal colonic transition studies [59]. Historically this was considered a manifestation of emotional disturbances in school-aged children; however, studies have not shown any serious psychological disorders, and treatment with psychotherapy alone has not been effective [63]. Treatment for these children should involve parental education and encourage defecation 5 min after each meal, a rewards system, and usage of a diary with avoidance of accusatory toilet training and guilt [59]. Laxatives have been shown to worsen outcomes, and functional non-retentive fecal soiling should be treated differently from retentive soiling [64]. Involvement of a mental health professional may be beneficial [59]. Resolution of the symptoms occurs in only 30 % of the children after 2 years and in 70 % of patients after 4 years with the problem persisting into young adulthood in up to 22 % of the population [65].
Anorectal Malformations
Key Concept: These patients present with a wide range in abnormalities, depending on the level of malformation and associated defects, with the management and outcomes often contingent on the malformation. However, a trial at medical therapy is still warranted in this group.
These malformations can be subdivided into three categories: [1] low anomalies, when the rectum has descended below the level of the puborectalis—typically without a fistula to the genitourinary tract and either with or without a cutaneous perineal fistula; [2] intermediate anomalies located at or just below the puborectalis muscle—often having a fistulous connection to the distal genitourinary tract; and [3] high anomalies with the rectum ending above the puborectalis (which may or may not be developed)—with a fistula to the urinary tract or vagina with no perineal opening (Fig. 27.6) [59]. Dependent upon the degree of pelvic floor development, there is great variation in the functional outcomes after repair of these malformations [60]. Generally, lower malformations have good functional outcomes, although they still suffer from occasional incontinence, constipation, an inability to control flatus, or sexual dysfunction [59]. Patients with lower defects mostly have voluntary bowel movements; however, up to 30 % can still experience soiling. Contrast this with patients with high anomalies, where 30 % may experience voluntary bowel movements and up to 90 % experience soiling [59, 66]. Major advances in pediatric surgery in recent decades have allowed for improved anatomic reconstruction primarily due to the posterior sagittal anorectoplasty (PSARP) for higher anorectal anomalies (Figs. 27.7 and 27.8) [59, 67]. These patients have improved outcomes, with up to half of patients experiencing full continence and only ~20 % having frequent soiling [59, 67], although they may still suffer from severe problems with constipation [66].

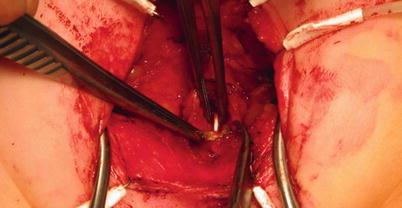
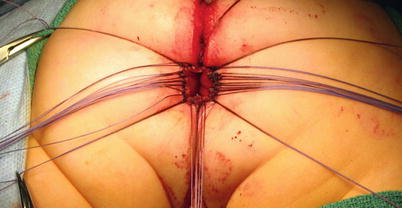

Fig. 27.6
Imperforate anus in an infant with no external opening

Fig. 27.7
PSARP procedure demonstrating the recto-urethral fistula and catheter in the urethral opening

Fig. 27.8
Completed imperforate anus anastomosis with restoration of intestinal continuity to the perineum
Remember that three elements are necessary for continence: functional sphincters, normal rectosigmoid motility, and intact sensation [59, 66]. Children with anorectal malformations who have subsequent defecation disorders should undergo both colonic and anorectal manometric testing to tailor their treatment [68]. It’s not surprising that patients who have a functional internal anal sphincter have higher anal resting tone and better outcomes in continence [69]. However, presence of severe sacral defects is one of the most important negative prognostic factors for continence [59]. These patients (i.e., incontinence from anorectal malformations with minor defects) are ideal for biofeedback, as they are more likely to respond. On the downside, this is time consuming, requires full cooperation from patients, and may not be helpful in patients with hypoplastic sphincters [70]. Therefore, medical treatment should first be used to modify the consistency of stool, with antidiarrheals for loose stools and enemas for those patients that have constipation and overflow incontinence [59, 60]. The enemas may be given retrograde or in an antegrade fashion via an ACE, which tends to be more effective than retrograde enemas [59, 60]. Many patients undergo a second surgery for their incontinence; however, long-term outcomes are no better and may be worse than those who only had one reconstruction [71].
In patients where the anal canal is not surrounded by the sphincter complex, a PSARP has been advocated. The long-term outcomes are variable in this cohort, with a small group of patients having complete resolution of their symptoms and most patients still relying on other measures for continence [72]. These patients may also get fecal soiling due to intractable constipation and overflow incontinence. They may also have a resulting megarectum either from distal obstruction or inadequate treatment of constipation. Although invasive, resection of this dilated portion of bowel can lead to improvement of symptoms of constipation and overflow incontinence. This should be considered only in children who have a functional internal anal sphincter, good anal sphincter tone, and no signs of neuropathy [73].
Spinal Pathology
Key Concept: The degree of bowel functional disorders from spinal pathology depends on the level of the defect. Although medical therapy is again a good first–line therapy, many will require a surgical procedure.
The most common causes of neuropathic bowel in children are spina bifida and myelomeningocele. The most common type of spina bifida affects the lumbosacral region causing bowel and bladder dysfunction [59]. Typical anal changes for these patients are poor voluntary sphincter function, poor colonic motility (specifically the left colon), and poor anorectal sensation [60]. However, the involuntary internal sphincter function is usually spared [60]. More than 50 % of patients with myelodysplasia suffer from fecal incontinence [60]. High lesions affect voluntary sphincter function, sensation, and colonic motility, and these patients are prone to fecal retention [60]. In patients with lower lesions, the functional problem is that patients cannot control their bowel emptying and stools evacuate spontaneously without warning; however, patients can rectally stimulate themselves or strain and have some degree of defecation control [60]. Dietary changes and toilet routines should be established; however, enemas are frequently necessary in these patients, and many of these patients can benefit from an ACE procedure [60].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








