Fig. 29.1
An isolated apoptotic body in a crypt. Less than six apoptotic bodies per ten crypt cross sections are seen in cases graded as indeterminate for acute rejection (H&E 20×)
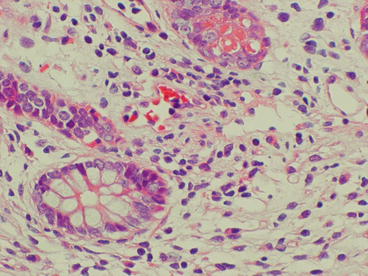
Fig. 29.2
Indeterminate for acute rejection. A mild mononuclear inflammatory infiltrate including blastic or activated lymphocytes is present (H&E 20×)
Acute Cellular Rejection, Mild: Grade 1/Score 2
Crypt injury is more evident. There are six or more epithelial apoptotic bodies per ten crypt cross sections together with other features correlated with mucosal damage such as mucin depletion, cytoplasmic basophilia, decreased cell height, nuclear enlargement and hyperchromasia and increased mitotic activity (Fig. 29.3). The villi demonstrate blunting and architectural distortion. The epithelial cell changes tend to be diffuse. The inflammatory infiltrate is mild to moderate with lymphocytes, including blastic or activated cells, eosinophils and occasional neutrophils. Oedema and vascular congestion are often present.
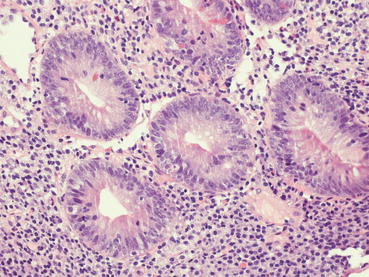

Fig. 29.3
In this biopsy, graded as acute cellular rejection, mild, here are shown crypt injuries: mucin depletion, cytoplasmic basophilia, decreased cell height, nuclear enlargement and hyperchromasia (H&E 20×)
Acute Cellular Rejection, Moderate: Grade 2/Score 3
Again six or more apoptotic bodies per ten crypt cross sections are present. In addition, single crypts may contain multiple apoptotic bodies with foci of “confluent apoptosis” causing erosions in crypt epithelium and focal crypt loss (Fig. 29.4). The inflammatory infiltrate (mononuclear cells with blasts and activated lymphocytes) is moderate to severe. Sometimes the superficial mucosa shows focal erosions, but this is not a prerequisite for the diagnosis.
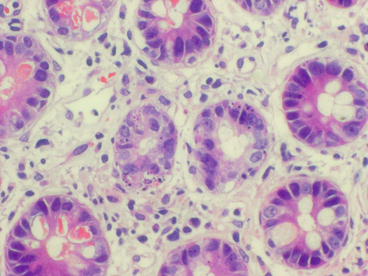

Fig. 29.4
Moderate acute cellular rejection. Single crypts may contain multiple apoptotic bodies with foci of “confluent apoptosis” causing erosions in crypt epithelium and focal crypt loss (H&E 20×)
Acute Cellular Rejection, Severe: Grade 3/Score 4
Crypt damage and destruction are prominent with crypt loss. Diffuse mucosal erosion and/or ulceration are present. Sometimes biopsy specimens show only granulation tissue and/or a pseudomembranous component (Fig. 29.5a, b). The number of epithelial apoptotic bodies varies and is not correlated to the severity of crypt loss: epithelial cell loss is frequently complete at the time of biopsy. When some epithelium is left adjacent to the ulceration, it usually exhibits rejection-associated changes, such as crypt epithelial damage [8].
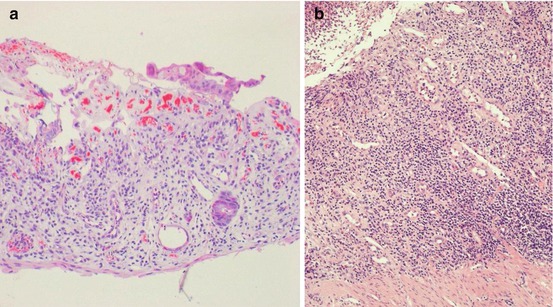

Fig. 29.5
(a, b). Severe acute cellular rejection. Crypt damage and destruction are prominent with crypt loss. Sometimes biopsy specimens show only granulation tissue and/or a pseudomembranous component (H&E 10×)
Apoptosis is characteristic of acute rejection although several inflammatory and immunologic processes (i.e. viral and autoimmune enteritis, GVHD) can be associated with an increase in apoptotic bodies. Investigation of both graft tissue and native mucosa can help pathologists and clinicians establish a correct diagnosis [9].
Top differential diagnoses:
Reperfusion injury (Fig. 29.6)
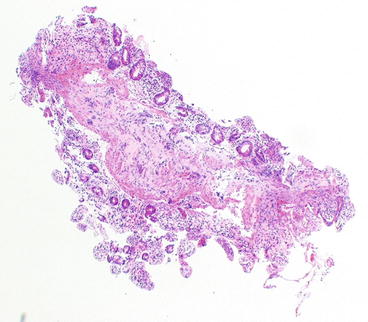
Fig. 29.6
Ischaemia-reperfusion injury. Mucosa shows blunting and denudation of the villi with a variable inflammatory infiltrate and oedema (H&E 5×)
Oedema of the villi
Surface denudation
Ischaemic injury with epithelial regeneration
Fewer apoptotic bodies
Viral infections [10–14]
(a)
Cytomegalovirus (CMV)
Nuclear inclusions with owl’s eye morphology (Fig. 29.7a)
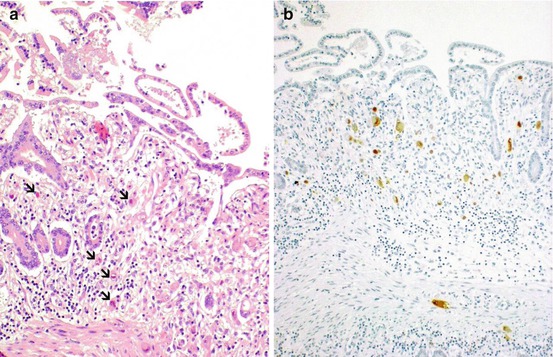
Fig. 29.7
(a, b) CMV infection. Nuclear inclusions in stromal cells in the mucosa (arrows, a H&E 10×) are immunoreactive for CMV antibody (b IHC staining 10×)
Neutrophils in the lamina propria along the luminal epithelial surface and in the crypts
Fewer apoptotic bodies
CMV immunohistochemistry (Fig. 29.7b)
(b)
Epstein-Barr virus (EBV)
Lymphocytic infiltrate in both the mucosa and epithelium (surface and crypts) (Fig. 29.8)
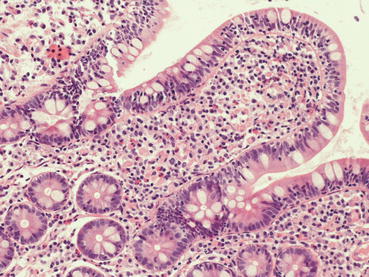
Fig. 29.8
EBV infection. Intraepithelial lymphocytic infiltrate in EBV positive patient (H&E 10×)
No evidence of apoptotic bodies
Lymphoid nodules extended to crypts, high risk of PTLD
(c)
Adenovirus
Surface epithelial degenerative changes
Characteristic inclusions most commonly in surface epithelium (round to crescent-shaped, “smudge” cells, eosinophilic inclusions)
Apoptotic bodies
(d)
Rotavirus
Mild to moderate villous blunting
Mixed inflammatory infiltrate (lymphocytes, plasma cells, eosinophils and neutrophil)
No significant increase in crypt apoptosis
29.1.1.2 Antibody-Mediated Rejection
ITx and MVTx can exhibit two types of AMR:
Hyperacute and accelerated acute rejection
Acute antibody-mediated rejection
Hyperacute and Accelerated Acute Rejection
This severe disease is the result of preformed donor-specific antibody (DSA) due to a previous allosensitization (pregnancy, blood transfusion or prior allogenic tissue exposure). The greater the allosensitization is, the more severe the clinical and pathological progression of the disease. The allograft organ can be rejected within minutes to hours (hyperacute rejection) or a few days (accelerated acute rejection) after transplantation [6, 15]. Cross-match tests in gastrointestinal transplantation have markedly reduced the incidence of these complications. The antibody response is directed at the endothelium of the donor microvasculature activating complement, innate immune response and the coagulation cascade [16]. The main morphological alterations are the result of a severe antibody-mediated response to the endothelia:
Dilated capillaries with prominent endothelial cells and neutrophils
Marked congestion
Capillary fibrin thrombi
Occasional mucosal ulceration
C4d staining in the endothelium of mucosal and submucosal capillaries
The resected specimens usually display arterial thrombosis and vasculitis [16].
Acute Antibody-Mediated (Humoral) Rejection (AMR)
The diagnosis of AMR is difficult and requires specific morphological and histochemical features and the knowledge of whether pre- and/or post-transplant alloantibodies are present. AMR can be isolated or associated with a T-cell-mediated acute rejection. The absence of previous allosensitization is a good prognostic factor, but allospecific HLA antibodies may arise de novo after transplantation and can trigger the onset of AMR [17].
The mucosa shows mild vascular congestion of the capillaries with red cell extravasation. These changes are not specific, being shared with other pathologies like bacterial and viral enteritis and ischaemia. In some cases, immunological endothelial damage can occur in the mesenteric arteries inducing progressive mucosal ischaemic damage and fibrosis. Vasculitis of the mesenteric arteries can lead to sclerosing mesenteritis; only surgical specimens can reveal these morphological changes [16].
Top differential diagnoses:
ACR (the two conditions can coexist)
Reperfusion injury
Surgical complications
Enteritis
29.1.1.3 Chronic Rejection
As with other solid organ transplants, CR is a progressive and chronic graft disease characterized by obliterative arteriopathy and graft loss. CR is insidious and lacks any morphological hallmarks in the mucosa. Clinical symptoms (watery diarrhoea, protein loss) are common to other diseases, but they are progressive and unresponsive to therapy. Submucosa and mesenteric obliterative arteriopathy is the main morphological change but is evident only in the surgical specimen of the failed graft or in full-thickness biopsy.
At endoscopy, the villi are flat and irregular, and ulcerative areas can be evident, while the bowel can be firm and fibrotic.
Mucosal biopsies show different degrees of fibrosis, low-grade apoptosis, crypt separation, mucin depletion and ischaemic damage. Not all these changes are diagnostic of CR, and clinical correlations are always required.
Surgical specimens often present serosal adhesion of adjacent bowel tracts and mesenteritis with firm, solid adipose tissue embracing the graft (Figs. 29.9 and 29.10). The wall is firm and thickened.
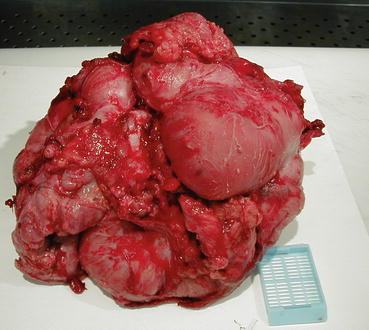
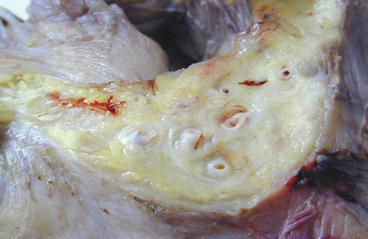

Fig. 29.9
Chronic rejection. Intestinal graft removed. Diffuse peritoneal fibrosis

Fig. 29.10
Chronic rejection. Intestinal graft removed. Perivascular fibrosis and mesenteritis in the adipose tissue
At histology the hallmark of CR is obliterative arteriopathy. Marked intimal hyperplasia, foam cells and a mild mononuclear inflammatory infiltrate are the main changes occurring in the medium-sized arteries in the submucosa and mesentery and mucosal ulcers (Figs. 29.11 and 29.12) [3, 6, 9].
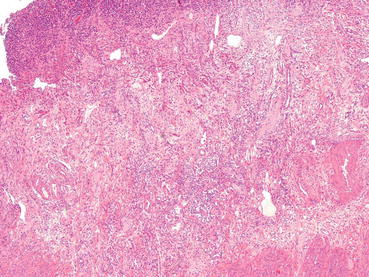
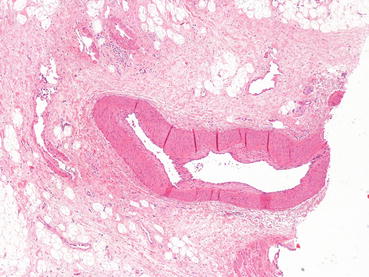

Fig. 29.11
Chronic rejection. Submucosal arteries with thickened wall and narrowed lumen, vasculitis and ulcerated mucosa (H&E 5×)

Fig. 29.12
Chronic rejection. Perivascular fibrosis and intimal thickening at histology (H&E 10×)
Top differential diagnoses:
Severe ACR
CMV chronic infection
Vascular diseases
29.1.2 Rejection in Other Organs in MVTx
In MVTx the different allografts can display varying forms and degrees of rejection. The first site to be involved is usually the distal part of the ileum, but the duodenum, stomach and colon may also be affected. Unlike the liver graft, MVTx seldom leads to rejection [19]. In addition, the criteria for the diagnosis of ACR and CR have to take into account organ-specific alterations.
29.1.2.1 Stomach Rejection
Stomach transplantation may be a part of a MVTx. The ACR changes in the grafted stomach are very similar to those encountered in small bowel allograft rejection [18].
The grading scheme proposed by Garcia et al. in 2004 suggests evaluating morphological changes in the different components of the mucosa: surface epithelium, lamina propria and glandular epithelium. The evaluation of apoptosis differs from bowel biopsies.
Morphological changes [19]:
Epithelial apoptosis, expressed as the number of apoptotic bodies/10 HPF
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






