Fig. 19.1
Extraperitoneal approach to external iliac vessels: left eternal iliac vein encircled with blue Silastic rubber; left external iliac artery encircled with red Silastic rubber
Anastomosis was performed using 6-0 Prolene running stitches in the end-to-side fashion (Fig. 19.2).
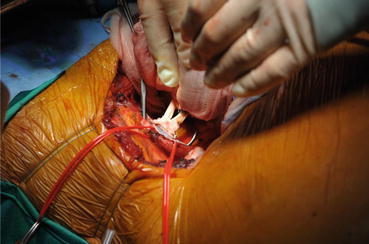

Fig. 19.2
Graft renal vein anastomosis with recipient’s external iliac vein clamped
In cases of external iliac vein thrombosis, the common iliac vein can be useful to perform the anastomosis, and only in rare cases is the inferior vena cava of the recipients anastomosed with the graft renal vein with an intraperitoneal approach.
A short or damaged right renal vein (SRRV) can make renal transplantation very difficult [19–22]. The right renal hilum has a single long artery and a short vein that causes difficulties while performing a venous anastomosis either from a living or cadaveric kidney and especially when the right renal artery has an aortic patch in the case of a cadaveric donor. It is more complicated and takes more time to perform the transplant especially with either deep iliac vessels or in obese patients. Anastomosis of an SRRV to the common or external iliac vein has been reported to be associated with technical problems such as angulation or tension of the venous anastomosis, reduced mobility, limited placement and inspection of the graft for hemostasis, and possible kinking of the donor artery. One technical solution consists of a more extensive mobilization of the recipient’s vessels, with the increased risk of lymphocele formation. A number of surgical approaches have been described to solve the problem of an SRRV [21]. Several techniques have been used to overcome this technical challenge, such as renal vein extension using an autologous saphenous graft, bovine arterial heterograft, or polytetrafluoroethylene (PTFE) vascular prostheses [21–24]. A technique of extending the right renal vein using the contiguous inferior vena cava was introduced to make vascular anastomosis feasible [25–27]. Using this technique, a portion of the contiguous inferior vena cava is removed during organ harvesting to extend very short right renal veins from cadaveric kidneys [27]. The technique for obtaining an appropriate length of the right renal vein using the vena cava is simple, physiological, and feasible and does not interfere with multiorgan procurement [19–27].
Short vessels can take up more time and extend the length of the warm ischemia during renal vessel anastomoses. In renal transplantation, when the vein is slightly longer than the corresponding artery, it allows easier venous and arterial anastomoses. Renal vein thrombosis is a serious complication leading to graft nephrectomy in many cases, despite medical or surgical therapy [21, 22]. In addition to this risk, there is concern that continuous thrombosis to the recipient vein may lead to a pulmonary embolus [21, 26, 27]. Right renal vein elongation with the inferior vena cava seems to be a much better approach than a venous saphenous autograft; spiral gonadal veins, bovine arterial heterografts, and vascular prostheses have also been used for these surgical challenges [21–24].
Dissection of the hilum of the donor kidney to lengthen the right renal vein is not recommended due to the risk of parenchyma hemorrhage and injury of the blood supply of the pelvis and ureter and can consequently cause necrosis to both. Faulty surgical techniques may be the cause for the majority of late ureteral stenosis due to ureteral devascularization and ischemia [19]. Right renal vein extension is particularly important in kidneys with multiple vessels, in order to avoid lesions to these venous and arterial variations, and to ensure easier anastomosis and possibly better positioning of the kidney. There is no increased risk with the use of the vena cava extension, and we recommend that the donor team routinely provide the right kidney with the vena cava attached. This allows the recipient team to determine whether an extension is appropriate for the particular recipient. This technique eliminates the need to mobilize the recipient’s vessels by a more extensive dissection, and the internal iliac vein division is not required. The kidney graft can easily be placed above the iliac vessels. This technique also preserves a patch of cadaveric aortic wall along with the right renal artery, thereby minimizing the risk of arterial graft kinking, renal transplant artery stenosis, and thrombosis.
Performance of this technique depends upon the vena cava being left intact and attached to the right renal vein when the organs are separated [25, 27]. Multiple right renal veins can also be elongated with the inferior vena cava.
19.1.3 Graft Renal Artery Anastomosis
There are few data available comparing vascular anastomosis techniques, and while there is no difference in the incidence of renal artery stenosis following end-to-end (hypogastric artery) or end-to-side (common or external iliac artery) arterial anastomosis, most of the data come from retrospective studies [28]. A recent prospective study by Matheus et al. [29] showed similar short and long results with both arterial anastomosis techniques. Although some doubts persist about what the best technique for arterial anastomosis is, end-to-side anastomosis to the external iliac artery is the preferred technique in deceased donors, because of the large Carrel patch obtained from the aorta [30]. Due to similar postoperative results in both arterial anastomosis groups, the choice of anastomosis technique in cadaver grafts still depends on surgical circumstances such as arteriosclerosis involving internal or external iliac arteries, multiple renal arteries, kidney position, and surgical team preferences [31, 32]. All the surgical procedures were performed by unilateral extraperitoneal approach to the iliac vessels. Anastomosis was performed using 7-0 Prolene running stitches in the end-to-side fashion (Fig. 19.3).
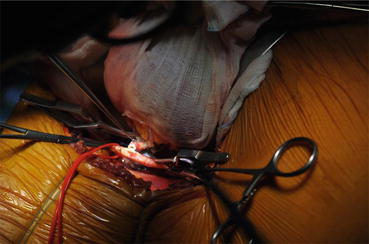

Fig. 19.3
Graft renal artery anastomosis with recipient’s external iliac artery clamped superiorly and inferiorly and sectioned with an aortic punch
However, some authors described the possibility of the occurrence of erectile dysfunction and renal artery stenosis with end-to-end internal iliac artery anastomosis [33, 34]. Arterial stenosis is a challenging issue, with a high incidence of complications during arterial stenosis correction, either by surgery or by stent insertion using percutaneous methods, due to the angle of the arterial anastomosis point [28]. Arterial anastomosis to the external iliac artery could reduce the incidence of erectile dysfunction, mainly in cases of a second transplant, in which the first kidney graft had been anastomosed to the contralateral internal iliac artery [31, 32, 35].
19.1.4 Graft Ureteral Anastomosis with the Recipient’s Bladder
In 1954, Muray et al. [36] performed the first successful renal transplant between identical twins using a Politano-Leadbetter (PL) intravesical technique to reimplant the transplanted ureter [37]. The PL technique [37] was subsequently used by most centers in North America owing to its high success rate in correcting vesicoureteral reflux in children. One source of complications with the PL technique was the cystostomy itself, with the risk of postoperative urine leakage. As a result, many centers began to use an anterior extravesical ureteral anastomosis. The extravesical ureteroneocystostomy was first described by Witzel in 1896 [38] and was popularized by Lich et al. [39]. Herein called the Lich-Gregoir, it is characterized by extravesicular access, the formation of an antireflux tunnel, and an urothelial anastomosis. An incision is made in the bladder wall musculature at the dome for 2–3 cm to expose the mucosa of the bladder wall. The mucosa of the bladder is then continuously sutured to the ureteral end with absorbable sutures (5-0 polydioxanone). The detrusor muscle is then closed over the anastomosis to create a submucosal tunnel approximately 2–3 cm long using 4-0 nonabsorbable polypropylene suture. Several operative techniques for reimplantation of the transplant ureter into the bladder have been used successfully. The anterior, extravesical approach described by Lich and associates [39] has been widely used, as has the Politano-Leadbetter intravesical technique [37]. Modifications of these methods have been described [40]. Each of these methods has advantages but also drawbacks; the Lich-Gregoir (LG) technique saves time but is not particularly effective in preventing reflux; the Politano-Leadbetter approach effectively prevents reflux but requires more difficult exposure and a longer operating time. The LG technique significantly lowers the risk of ureteral leakage when compared with the PL technique and significantly lowers the risk of hematuria when compared with the PL technique in kidney transplantation.
There were no differences in the prevalence of ureteral strictures and vesicoureteral reflux between the various techniques [41]. The higher risk of urinary leakage in the PL group might be the result of the second cystotomy, which creates a potential extra leakage site. It has also been hypothesized that the use of a shorter segment of the ureter in the LG technique decreases the risk of distal ureteral necrosis and therefore results in a lower risk of urine leakage at the ureterovesical junction [42, 43]. The higher rate of hematuria in the PL group might also be explained by the extra cystotomy, from which bleeding can arise. The Lich-Gregoir and Politano-Leadbetter approaches are used either with or without a temporary ureteral stent; however, ureteral stenting seems to have a significant protective effect against the development of urological complications after renal transplantation, as described in a meta-analysis by Mangus et al. and a Cochrane review by Wilson et al. [44, 45] (Fig. 19.4).
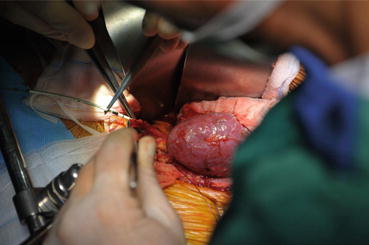

Fig. 19.4
Donor kidney ureteral anastomosis with the recipient’s bladder: ureteral stenting with double J stent 6–12 French
However, whether ureteral stenting is preferably performed by routine or selective approach in the case of problematic anastomoses has still to be clarified [46–48]. Some studies have shown an increase in urinary tract infections (UTIs) with ureteral stenting [45, 49–51]. Wilson et al. describe a relative risk of 1.49 (95 % CI 1.04–2.15) for UTIs with ureteral stenting, unless the patients were given prophylactic antibiotics, in which case the prevalence was equal to the non-stented group (RR 0.97, 95 % CI 0.71–1.33) [45]. There were not enough data in the included studies to assess the effect of ureteral stenting on UTIs.
19.2 Double Kidney Transplantation with “Suboptimal” Donors
The shortage of kidney donors is a rate-limiting step in renal transplantation programs in many transplant centers. To overcome the disparity between supply and demand of organs, various strategies such as the increased use of organs from extended criteria donors (ECD) have been proposed. Double kidney transplantation (DKT) is another approach for expansion of the existing deceased donor pool. The number of functioning nephrons is the most important determinant of kidney function; therefore, the higher number of functioning nephrons supplied by dual marginal kidneys should slow down or even prevent progressive deterioration of graft function [52]. Transplantation of two marginal kidneys rather than one suboptimal kidney to one recipient would result in more functioning nephrons that ultimately may improve the patient and graft outcome [52]. The decision to perform DKT is based on gross characteristics of the kidneys and results of the renal biopsy. Small kidneys or those with extensive surface scarring or cystic lesions are discarded. In addition to the clinical evaluation of the donor, the selection criteria of deceased donor (DD) kidneys are based on a macroscopic and histological assessment. Marginal DD kidneys are classified as low or high risk, based on the donor’s age, renal function, and comorbidities; in particular, high-risk marginal kidneys are considered those from donors aged >70 years or 60–69 years with at least two of the following conditions: serum creatinine >1.5 mg/dL, calculated creatinine clearance <60 ml/min, history of hypertension or diabetes, proteinuria >1 g/day, and cause of death is cerebrovascular. High-risk donors are subjected to a renal biopsy, and, based on the Karpinski [53] and Remuzzi [52] histological scores, the kidneys are allocated for single kidney transplantation (SKT) or DKT. Using a histological scoring system (4–18), a score between 0 and 12 is attributed to the kidneys, depending on the percentage of glomerulosclerosis, tubular atrophy, interstitial fibrosis, and arterial and arteriolar narrowing. Kidneys scoring 0–3 are used for SKT, those scoring 4–6 are used for DKT, and those scoring >6 are considered inadequate for transplantation. Recipients are required to sign an informed consent form for DKT before placement on the waiting list. Potential recipients of DKT (mainly older than 60 years) are also placed on an additional, but separate, DKT waiting list, which facilitates the allocation of ECD kidneys in an “old-for-old” fashion, i.e., kidneys from older donors are allocated to older recipients. Patients are considered suitable for DKT listing when they satisfy the standard criteria for eligibility for kidney transplantation, but are excluded if highly sensitized (PRA >50 %) or if undergoing retransplantation, because allocation of DKT is not based on HLA-matching. In addition, patients affected by polycystic kidney disease are excluded unless they have been nephrectomized or clearly have enough space in the iliac fossa for the implantation of two kidneys. DKT is considered contraindicated in the presence of severe bilateral atherosclerosis of the iliac vessels. The first DKT from an adult deceased donor was reported in 1996 by Johnson et al. [54]. The classic surgical technique for DKT included bilateral placement of the two kidneys extraperitoneally through two separate Gibson incisions or intraperitoneally through a midline incision. Unilateral DKT was first reported by Masson et al. in 1998 [55]. There are various surgical techniques for unilateral DKT [56–58]. Ekser et al. proposed sequential DKT: one kidney with its vessels anastomosed end-to-side to recipient vessels (external iliac artery and vein) and then another kidney with its vessels anastomosed to the external iliac artery and vein distal to the site of the first anastomosis [59]. After creating an adequate extraperitoneal space, the right donor kidney is preferably placed superiorly because its renal vein can be lengthened by a segment of inferior vena cava. Another reason to position the right kidney superolaterally in the right flank is because the right kidney has a longer artery. If necessary, the internal iliac (hypogastric) vein is dissected to mobilize the external iliac vein and thus facilitate renal vein anastomoses to the external iliac vein of the recipient. After revascularization of the right kidney, vascular clamps are placed immediately below the venous and arterial anastomoses. The left donor kidney is transplanted distally, allowing the transplanted right kidney to continue to be perfused. Extravesical ureteroneocystostomies are performed separately, according to the Lich-Gregoir technique, with a double J stent for each ureter, leaving the ureter of the upper transplanted kidney lateral to the lower one. Veroux et al. proposed another technique for DKT. They joined the arteries and veins of the two kidneys at bench surgery. The newly joined artery and vein of the two kidneys were then anastomosed end-to-side to the common iliac artery and external iliac vein, respectively [60]. Although unilateral DKT is a complex surgical procedure, it can reduce the cold ischemia time and the operating time, leaving the contralateral side intact for further transplant. On the other hand, kidneys with multiple arteries and veins can be transplanted like a single kidney.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






