5. PANCREAS
EXOCRINE FUNCTIONS
After studying this chapter you should be able to:
1. Describe the macroscopic and microscopic anatomy of the pancreas and relate these to its function.
2. Describe the components of exocrine function of the pancreas and apply this knowledge in understanding the pathological conditions of acute and chronic pancreatitis and cystic fibrosis.
Introduction
The pancreas contains exocrine tissue that secretes pancreatic juice, a major digestive secretion, and endocrine tissue that secretes the hormones insulin and glucagon. The hormones are important in the control of metabolism and their roles in the absorptive and postabsorptive metabolic states will be discussed in Chapter 9. This chapter will be mainly concerned with the exocrine secretions of the pancreas, their functions, and the mechanisms whereby the secretory processes are controlled.
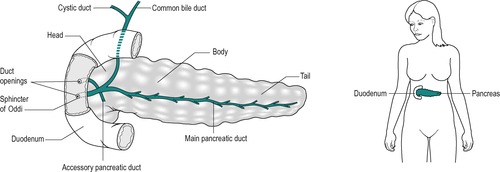
Pancreatic juice finds its way into the duodenum via the pancreatic duct that opens into the duodenum at the same location as the common bile duct (Fig. 5.1). Entry of both pancreatic juice and bile into the duodenum is controlled by the sphincter of Oddi. The smooth muscle of the sphincter is contracted between meals so that the junction is sealed. When a meal is being processed in the gastrointestinal tract, the sphincter muscle relaxes and allows the pancreatic juice and bile into the small intestine. The control of the sphincter of Oddi is discussed in Chapter 7. Pancreatic exocrine dysfunction may be due to disorders of the pancreas itself, or to blockage of the ducts which prevents the exocrine secretions reaching the duodenum. Duct blockage may also result in impaired bile flow from the liver, which can result in jaundice.
In the small intestine, pancreatic juice, bile and the juices secreted by the walls of the intestines, mix with the fluid (chyme) arriving from the stomach (see Fig. 7.1). Pancreatic juice provides most of the important digestive enzymes. In addition, by virtue of its bicarbonate content, it helps to provide an appropriate neutral or alkaline pH in the intestinal lumen for the enzymes to act on their nutrient substrates. The functional importance of the pancreas to the digestive processes can be illustrated by the problems arising in chronic pancreatitis (Case 5.1: 1) and cystic fibrosis (Case 5.2: 1) conditions in which pancreatic tissue is destroyed.
Case 5.1
A 40-year-old man who had been a heavy drinker for many years, went to see his general practitioner. He had made two previous visits over the past year complaining of recurrent episodes of abdominal pain. Although the pain had been intermittent at first, it was now continuous. The doctor ascertained that the pain originated in the epigastrium, and radiated through to the back. The patient had lost a considerable amount of weight since his last visit. The doctor noticed that he was mildly jaundiced. Arrangements were made for the patient to be admitted to hospital for tests.
An X-ray examination, and serum and urine analyses were performed. The patient’s stools were collected over 3 days. These were pale-coloured and bulky, indicating a high fat content (steatorrhoea). He was told to abstain from food the next morning so that a glucose tolerance test (see Chapter 9) could be performed.
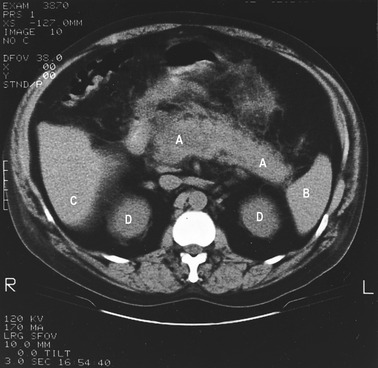
The blood tests showed increased bilirubin and alkaline phosphatase. The glucose tolerance test showed an abnormally high and prolonged rise in serum glucose, and urine analysis confirmed the presence of glucose (glycosuria), indicating that the patient was diabetic. The presumptive diagnosis was chronic pancreatitis. The patient was prescribed pethidine to control the pain. He was advised to abstain completely from alcohol, and to eat regular meals. Figure 5.2 shows a CT (computerized tomography) scan of the abdomen of an individual with chronic pancreatitis. The swollen pancreas can be clearly seen. Examination of the details of this case gives rise to the following questions:
• Is the primary defect in chronic pancreatitis known?
• What might the X-ray have revealed?
• What could be the cause of the condition in this patient?
• How are the exocrine and endocrine functions of the pancreas impaired in chronic pancreatitis?
• What did the high faecal fat content indicate?
• What is the basis of the glucose tolerance test and why has diabetes mellitus developed in this patient?
• Why did the patient appear jaundiced?
• Why were the patient’s serum bilirubin and alkaline phosphatase abnormally high?
• What are the main physiological consequences of this disease and how can the condition be treated or managed?
These questions will be addressed in this chapter.
Case 5.2
A 12-year-old boy who was suffering from cystic fibrosis was taken to the outpatient clinic for his regular check-up. Cystic fibrosis is an inherited autosomal recessive disorder. The primary defect is a mutation in the gene that encodes the cystic fibrosis transport regulator (CFTR). The CFTR is responsible for cyclic-AMP-regulated Cl− conductance in the membranes of secretory cells of epithelia. The defect causes impaired Cl− transport that is predominantly a problem in the wet surfaces of the respiratory tract, but also causes severe problems in the pancreas. It is also manifest in the reproductive tract and sweat glands. The boy’s condition had been diagnosed soon after birth and he had both respiratory tract and pancreatic involvement. He had been asked to bring a sample of his stool. This was pale-coloured, poorly formed and oily in appearance. It was sent to the laboratory for analysis to assess his pancreatic function. His exocrine pancreatic insufficiency was being treated with a pancreatic enzyme preparation and the anti-ulcer drug ranitidine.
After studying the above details, we can attempt to answer the following questions:
• How is the inherited defect manifest in the pancreas?
• Which abnormalities of pancreatic function result from this pathology?
• Why was the boy’s stool pale coloured?
• Which tests would be performed on the stool sample?
• Would there be any abnormalities in the acid–basestatus of this patient?
• Why was the child being treated with an enzyme preparation and would there be any problem with giving such a preparation by mouth?
• Why is the boy being treated with ranitidine?
Anatomy and morphology
The pancreas is an elongated gland that lies in the abdominal cavity. It can be divided into three regions: the head, the body and the tail (Fig. 5.1). The head is an expanded portion that lies adjacent to the C-shaped region of the duodenum to which it is intimately attached by connective tissue, and to which it is connected by a common blood supply. The body and tail extend across the midline of the body toward the hilum of the spleen. The pancreatic duct (duct of Wirsung) extends through the long axis of the gland to the duodenum. Pancreatic juice empties from this duct into the duodenum via the ampulla of Vater. In some individuals, there is also an accessory pancreatic duct. Bile in the common bile duct from the liver also enters the duodenum at the ampulla of Vater, by passing through the head of the pancreas. This is why inflammation (pancreatitis) and tumours (pancreatic cancer) involving the head of the pancreas are commonly associated with jaundice, as a consequence of blockage of the common bile duct.
Exocrine tissue
The exocrine units of the pancreas are tubuloacinar glands that are organized like bunches of grapes (Fig. 5.3), in a similar manner to the units in the salivary glands. The exocrine units surround the islets of Langerhans, the endocrine units of the pancreas. For this reason, destructive diseases such as chronic pancreatitis involve impairment of both exocrine and endocrine function. A thin layer of loose connective tissue surrounds the gland. Septa extend from this layer into the gland, dividing it into lobules and giving it an irregular surface. Larger areas of connective tissue surround the main ducts and the blood vessels and nerve fibres that penetrate the gland. Small mucous glands situated within the tissue surrounding the pancreatic duct secrete mucus into the duct.
Endocrine tissue
The endocrine units, or islets of Langerhans, are most numerous in the tail region of the pancreas. They consist of clusters of cells which are surrounded by the pancreatic acini (Fig. 5.3). The islets vary considerably in size. As with all endocrine tissue, the hormones they produce are secreted into the blood. The major endocrine cell types present are: α, β, D and PP cells, which secrete glucagon, insulin, somatostatin, and pancreatic polypeptide, respectively (for more information, see Ch. 9). Different types of endocrine cells can be distinguished under the electron microscope by the different appearance of the granules within them. The islet cells have the general features of APUD cells (see Ch. 1). In addition, there are a few (<5%) small ‘clear’ cells with as yet no clearly defined function.
Glucagon and insulin, the hormones produced by the α and β cells, respectively, are taken up by the local blood vessels to act systemically. Somatostatin acts locally in a paracrine manner to inhibit the secretion of the α and β cells, as well as the exocrine secretions of the acinar and duct cells. Pancreatic polypeptide acts in a paracrine manner to inhibit the exocrine secretions of the pancreas.
Oxygenated blood is supplied to the pancreas by branches of the coeliac and superior mesenteric arteries. The pancreatico-duodenal artery lies between the duodenum and the head of the pancreas. The close proximity of these structures means that it would be hazardous to remove the pancreas without the duodenum, because the main arterial supply would inevitably be damaged. The blood drains from the pancreas via the portal vein to the liver. This arrangement is important for the transport of pancreatic hormones to the liver where they contribute to the control of energy metabolism, a topic discussed in Chapter 9. The acini and ducts are surrounded by separate capillary beds. Some of the capillaries that supply the islets converge to form efferent arterioles which then enter further capillary networks around the acini. This arrangement is important for the paracrine control of pancreatic exocrine secretion.
Cholinergic preganglionic fibres of the vagus nerve enter the pancreas. These synapse with postganglionic cholinergic nerve fibres which lie within the pancreatic tissue and innervate both acinar and islet cells. Postganglionic sympathetic nerves from the coeliac and superior mesenteric plexi innervate the pancreatic blood vessels as well as the acinar and duct cells.
Histology of the exocrine tissue
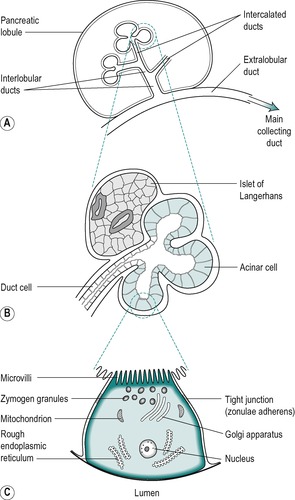
Figure 5.3 shows the structure of a pancreatic lobule. The exocrine units of the pancreas, or pancreatins, each consist of a terminal acinar portion and a duct (Fig. 5.4). The duct that drains the acinus is known as an intercalated duct. These empty into larger intralobular ducts. The intralobular ducts in each lobule drain into a larger extralobular duct that empties the secretions of that lobule into still larger ducts, and the latter converge into the main collecting duct, the pancreatic duct.
The acinus is a rounded structure consisting of mainly pyramidal epithelial cells (Fig. 5.4). These cells secrete the digestive enzymes of the pancreatic juice. They display polarized features that are common to secretory cells (Fig. 5.3). The nucleus of the acinar cell is situated at the base of the cell. The cytoplasm in the basal region can be stained with haematoxylin or basic dyes due to the presence of rough endoplasmic reticulum, the site of production of the digestive enzymes. Small mitochondria are situated throughout the cell. The apical portion of the cell contains the Golgi apparatus, and numerous zymogen granules that contain the pancreatic enzymes or their precursors. The apical region therefore stains with acid dyes such as eosin. Microvilli extend from the apical surface of the acinar cell into the lumen. The apical poles of neighbouring cells are joined by tight junctions, known as zonulae adherens. These junctions separate the fluid in the lumen of the acinus from the fluid in the intercellular spaces that bathes the basolateral surfaces of the cells. The tight junctions are impermeable to macromolecules, such as digestive enzymes, in the luminal fluid, but permit the exchange of water and ions between the interstitial spaces and the lumen of the acinus. Disruption of these junctions may be an aetiological factor in the development of chronic pancreatitis. Gap junctions between neighbouring cells allow rapid changes in membrane potential to be transmitted between the cells. They also permit the exchange of low molecular weight molecules (<1400kDa mass) between cells.
The intercalated duct begins within the acinus. This is a unique feature of secretory glands. The upper duct cells within the acinus are known as centroacinar cells (Fig. 5.4). These stain lightly with eosin. They are squamous cells with a centrally placed nucleus. These cells are continuous with those of the short intercalated duct that lies outside the acinus and drains it. The intercalated ducts are lined by flattened squamous epithelial cells. The neighbouring duct cells are joined by tight junctions, as in the acinus. These separate the duct lumen from the intercellular spaces and function to exclude large molecules from the spaces. They also have gap junctions that permit the transmission of membrane electrical changes between the cells. These ducts lead into the intralobular ducts that are lined with cuboidal or low columnar epithelium. Larger ducts contain interlobular connective tissue cells and APUD cells.
Pancreatic juice
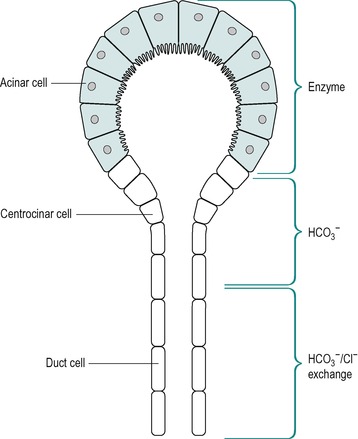
The pancreatic juice entering the duodenum is a mixture of two types of secretion: an enzyme-rich secretion from the acinar cells and an aqueous alkaline secretion from the ducts. Thus, if the ducts are ligated near the acini, which results in acinar cell degeneration, the secretion of the alkaline component of the juice is largely unaltered, but the secretion of enzymes is markedly reduced. The alkaline secretion originates largely from the centroacinar cells and the duct cells of the intralobular and small interlobular ducts. These relationships are illustrated in Figure 5.4.
Alkaline secretion
Composition
The cells of the upper ducts secrete an isotonic juice which is rich in bicarbonate but contains only traces of enzymes. There is a continuous resting secretion of this juice, but it can be stimulated up to 14-fold during a meal. It contains Na+, K+, HCO3−, Mg2+, Ca2+, Cl− and other ions present in concentrations similar to those of plasma. It therefore resembles an ultrafiltrate of plasma, but is alkaline by virtue of its high HCO3− content.
Functions
The pancreatic juice arriving in the duodenum is mixed with the chyme by contractions of the smooth muscle of the small intestine. The function of the alkaline pancreatic secretion, together with the other alkaline secretions (bile and intestinal juices) that act in the small intestine, is to neutralize the acid chyme arriving from the stomach. This is important for several reasons:
• The pancreatic enzymes require a neutral or slightly alkaline pH for their activity
• The absorption of fat depends on the formation in the intestinal lumen of micelles, a process which only takes place at neutral or slightly alkaline pH values
• It protects the intestinal mucosa from excess acid which can damage it, leading to the formation of ulcers. This (peptic) ulceration most commonly occurs in the first part of the duodenum, before the acidic chyme has mixed with the alkaline pancreatic juice.
Cellular mechanisms of secretion
The mechanisms involved in the production of intracellular HCO3− in the centroacinar and upper duct cells are illustrated in Figure 5.5. The initial intracellular step involves the reaction of CO2 with water. Secreted H+ ions react with HCO3− ions in the blood perfusing the gland and this generates CO2, some of which diffuses into the duct cell. More than 90% of the HCO3− in pancreatic juice is derived from blood CO2. In the cell, the CO2 combines with intracellular water to generate carbonic acid, in a reaction catalysed by carbonic anhydrase II, an enzyme present in the centroacinar and upper duct cells. The carbonic acid dissociates to give HCO3− and H+. Whilst bicarbonate is being secreted the partial pressure of CO2 (pCO2) in the cells is lower than in the blood as it is being used up in the production of HCO3− ions, and the higher the rate of secretion the greater the downhill gradient for diffusion of CO2 into the cell. The HCO3− ions are secreted across the luminal membrane by Cl−/HCO3− exchange, and the H+ ions are secreted into the blood. Thus for every HCO3− ion that is secreted into the duct lumen one H+ ion is secreted into the blood. Therefore the blood flowing through the pancreas becomes transiently acid when it is secreting HCO3−. The H+ ions in the blood help to neutralize the ‘alkaline tide’ produced during a meal by the secreting stomach (see Ch. 3), by combining with plasma HCO3− to produce CO2. In conditions where a patient is provided with a pancreatic fistula, loss of HCO3− must be carefully monitored (Box 5.1).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree