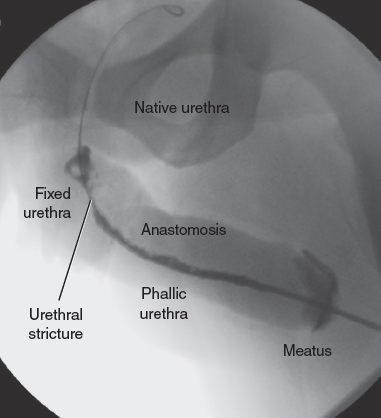
CHAPTER 12 One goal of neophallus construction in female-to-male transgender surgery is to give a patient the ability to void while standing. Although some patients use external “urinary assist” devices to facilitate urination from the native urethra while standing,1–3 many undergo neourethra construction, because the ability to stand to void is a high priority among female-to-male transgender individuals. More than 98% of patients reported a desire to stand to void.4 The management of urologic sequelae is important after neourethra construction, because fistulas and strictures are common complications. The urethra of the male-to-female transgender patient after phalloplasty can be divided into distinct segments3,5 from proximal to distal: native (female) urethra, fixed urethra, anastomotic urethra, phallic urethra, and meatus. The fixed urethra is the portion of the urethra formed after lengthening the native urethra through local vaginal or labial flaps, extragenital flaps, and grafts of skin or mucosa.3,6 The phallic urethra can be constructed by a variety of techniques, including prelamination, prefabrication, tube-in-tube techniques, and pedicled flaps.3,6 Urethrocutaneous fistula is the most common urethral complication. The fistula rate of radial forearm free flap phalloplasty ranges from 22% to 75%.7–10 Urethral fistulas commonly occur at points of anastomosis—between the phallic urethra and fixed urethra and between the fixed urethra and native urethra, although fistulas can occur anywhere along the neourethra.11 Fistulas occur most commonly at the anastomosis between the phallic urethra and fixed urethra8 as a result of vascular insufficiency of the flap and the decreased lumen of the phallic urethra. The change in caliber of the lumen from fixed to phallic urethra may cause a relative obstruction of the urinary stream distal to the site of the fistula.8 The smallcaliber lumen of the phallic urethra may be the result of tissue shrinkage or insufficient size of the urethra at the time of construction. Spontaneous closure of the fistula tract has been reported; Fang et al12 reported spontaneous closure of the fistula within 2 months in as many as 35.7% of patients. Urethral stricture is another common urologic complication, with reports of incidences varying from 25% to 58%.8,13,14 Although stricture can occur in any segment of the urethra, the most common location of stricture formation is at the anastomosis of the fixed and phallic portion of the neourethra (Fig. 12-1). Lumen et al5 characterized stricture formation after phalloplasty and determined that urethral stricture occurred at the anastomosis in 40.7%, phallic portion in 28%, the meatus in 15.3%, fixed segment in 12.7%, and multifocal in 7.6%. Ischemia is considered the cause of strictures at all levels. Fistula formation may also contribute to dense scar formation and kinking of the tissues, especially at the anastomosis of the phallic to fixed portions.5,13 At the meatus, contracture of the anastomosis between the skin of the glans and urethral tissue can lead to meatal stenosis. Mean stricture length in this series was 3.6 cm (range 0.5 to 15 cm).5 Fistula and urethral stricture may occur simultaneously. In a series of one-stage urethroplasty by Rohrmann and Jakse,8 40% of patients developed a fistula and strictures, with the fistula usually proximal to the stricture. Proper management of urologic sequelae of phalloplasty is mandatory, given that urinary fistulas and urinary obstruction caused by urethral stricture can have grave consequences, such as chronic infection, sepsis, and renal failure, as well as compromised quality of life. If a patient is in urinary retention, urinary drainage must be performed with the placement of a suprapubic catheter. The extent of subsequent urinary reconstruction will depend on the individual’s health and preferences. Fig. 12-1 Retrograde urethrogram of the neourethra, showing the urethral stricture at the anastomosis between the phallic and fixed urethra. A patient with urologic sequelae after phalloplasty will often present with voiding complaints. This may include increased difficulty with urination, whether with a decreased stream or an increased need to strain to void or a complete inability to void. If a urethrocutaneous fistula has formed, a patient may complain of urine or purulent drainage at a location other than the meatus. The drainage may occur at the time of micturition but may also occur afterward because of pooling of urine in the urinary tract. A patient may also complain of dysuria or suprapubic pain. The first step in patient evaluation is a careful physical examination. The suprapubic area and neophallus should be examined for evidence of infection such as erythema and induration. The areas are also palpated for fluctuance to determine if any fluid collections require drainage. Ultrasound examination may be performed to evaluate for the presence of an abscess. All areas are evaluated for fistulous openings, and the urethral meatus itself is examined for patency. Treatment of infections should be performed before surgery. Cellulitis should be treated. If there is concern about a urinary tract infection, a urine sample may be sent for urinalysis and urine culture. If a patient has a suprapubic tube, the urine sample may be sent from the tube. However, indwelling catheters are often colonized, and organisms grown from these cultures may not be representive of infections. If the urine culture findings return positive with a clinical indication of infection, culture-specific antibiotics should be given. Further anatomic evaluation can be performed with a retrograde urethrogram or voiding cystourethrogram to help to delineate the location of the stricture or fistula. Examination under anesthesia is frequently useful given the complex urinary anatomy of a patient’s status after phalloplasty. A suprapubic tube can be placed at that time of this examination if a patient does not already have one in place. A notation should be made regarding a patient’s previous surgeries and what potential flaps and grafts are available for use for urologic reconstruction. The first step in managing a urinary fistula or stricture causing urinary obstruction is to ensure that a patient’s urine is adequately diverted with a suprapubic catheter of adequate size. The suprapubic catheter should be placed 2 to 4 cm above the pubis. Placement of a 16 Fr or larger catheter facilitates the use of the suprapubic channel for antegrade cystourethroscopy to delineate the proximal urethra. Antegrade cystourethroscopy is easiest when the suprapubic tube is at the midline. The surgeon must be careful to avoid the vascular anastomosis to the neophallus. The standard preoperative evaluation for urethral strictures and fistulas includes a retrograde urethrogram combined with endoscopic evaluation to determine the extent of stricture and level of fistulization if present. The patient is brought to the operating room and is placed in the low lithotomy position and prepped and draped to allow access to the perineum and suprapubic region. A layer of complexity is added to the patient’s status after phalloplasty, because the neourethra created may have a smaller caliber than the native urethra and may be unable to accommodate a standard 16 Fr flexible cystourethroscope. At our institution, we use a flexible ureteroscope to navigate the neourethra. The retrograde urethrogram can be performed by injecting contrast through the ureteroscope. This technique has the advantage of allowing direct visualization of the neourethra rather than attempting to blindly pass a catheter into a possibly tortuous neourethra. A guidewire is used to gradually advance the ureteroscope. After contrast is injected, fluoroscopic images are obtained to delineate the anatomy of the stricture and fistulas. If the suprapubic tract is mature, antegrade cystoscopy may be performed to delineate the anatomy proximal to the stricture point. Location, length, and caliber of the stricture, as well as location of any fistulas, are key factors to determine. Any fluid collections or abscesses, which have formed as a result of obstruction, should be adequately drained. If a fistula does not heal on its own, excision of the fistula tract with closure and coverage with a flap is often required. The patient is placed under general anesthesia. The surgeon carefully positions the endotracheal tube to the side opposite the potential side of graft harvest if a buccal graft is planned. Positioning a patient in the lithotomy position allows access to the genitalia, suprapubic area, and thighs as needed for flap harvest.
Management of Unfavorable Urologic Sequelae After Phalloplasty in Transgender Patients
Key Points
 Urinary fistula and urethral stricture are common after neophallus reconstruction.
Urinary fistula and urethral stricture are common after neophallus reconstruction.
 Fistulas commonly occur at sites of anastomosis.
Fistulas commonly occur at sites of anastomosis.
 Reconstruction must be tailored to the patient’s anatomy.
Reconstruction must be tailored to the patient’s anatomy.
Indications and Contraindications
Patient Evaluation
Preoperative Planning and Preparation
Surgical Technique
Abdominal Key
Fastest Abdominal Insight Engine