Ischemic Colitis
General Comments
Acute ischemia results from an acute reduction in the blood flow resulting from an acute occlusive event or as a result of a decreased blood flow. It can result from either arterial or venous impairment. Ischemic colitis affects patients of all ages including infants, but it preferentially affects elderly patients with a history of arteriosclerosis, diabetes, hypertension, renal insufficiency, and/or cardiovascular disease. The mesenteric arteries become atherosclerotic with either gradual arterial occlusion or acute blockage by a thrombus. It is also more common in patients with a thrombophilic state including the presence of factor V Leiden mutations (96). Ischemia developing in a younger person without a history of drug use and in a patient without apparent predisposing factors, such as cardiac failure or arrhythmia, should be investigated for one of the vascular lesions listed in Table 6.8 or for a primary clotting abnormality. In women, the use of hormones or oral contraceptives should be investigated. Ischemia also complicates many other disorders, including some infections, especially cytomegalovirus (CMV). Common causes of low flow states include cardiac failure, arrhythmias, digitalis toxicity, shock resulting from a marked reduction in left ventricular output, and sepsis. Occlusive causes of acute ischemia include arteriolar or venous obstruction
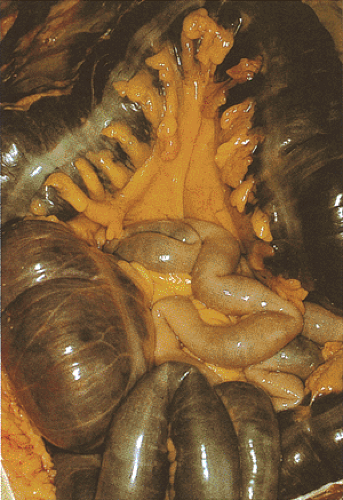
(Table 13.5) as the consequence of thromboembolism (Fig. 13.66), dissecting aneurysm, vascular spasm, or vascular compression during prolapse, intussusception, etc. Colonic ischemia also complicates major surgery, infections, drug use (see below), retroperitoneal fibrosis (97), and cocaine abuse. Marathon runners may also develop colonic ischemia (98). Large intestinal ischemia exhibits a spectrum of changes ranging from fatal infarctions with gangrene (Fig. 13.67) to reversible ischemia.
(Table 13.5) as the consequence of thromboembolism (Fig. 13.66), dissecting aneurysm, vascular spasm, or vascular compression during prolapse, intussusception, etc. Colonic ischemia also complicates major surgery, infections, drug use (see below), retroperitoneal fibrosis (97), and cocaine abuse. Marathon runners may also develop colonic ischemia (98). Large intestinal ischemia exhibits a spectrum of changes ranging from fatal infarctions with gangrene (Fig. 13.67) to reversible ischemia.
Clinical Features
The clinical signs and symptoms vary according to the underlying etiology, duration and linear extent, and depth of mural involvement. In elderly individuals with generalized cardiovascular disease, a rapidly developing syndrome that starts with mild to moderate abdominal pain and quickly evolves with nausea, vomiting, diarrhea, rectal bleeding, and abdominal distention suggests the presence of ischemia (99). Such patients often have a predisposing hypotensive period. Reversible colonic ischemia manifests as crampy, usually left-sided, lower abdominal pain, associated with tenesmus, fever, and leukocytosis. Severe pain, fever, leukocytosis, peritoneal irritation, and/or profound ileus indicates extensive, and probably irreversible, ischemic damage (100). Anorexia, nausea or vomiting, abdominal distention from ileus, an altered sensorium, and clinical signs or symptoms of sepsis or shock can also develop. When ischemia extends to the muscularis propria, motility problems develop.
 FIG. 13.65. Biopsy artifact. The glands have intussuscepted into one another, appearing as glands lying within the lumen of other glands. |
TABLE 13.5 Causes of Intestinal Venous Compromise | |
|---|---|
|
Three types of colonic ischemia develop: (a) a transient reversible form that usually remains confined to the mucosa or submucosa; (b) a chronic form that extends into the bowel wall and lasts for months; and (c) an acute fulminant form that progressively destroys all layers of the bowel wall, usually with catastrophic consequences, including transmural necrosis and perforation. Complete recovery often follows mild ischemia. More severe disease undergoes partial resolution complicated by fibrosis and stricture formation.
The most severe form of injury, transmural infarction, requires resection of the affected segment; otherwise, perforation or death will ensue. Almost half of patients with colonic ischemia have a transient, reversible form of the disease; 20% to 25% have chronic colitis or deep colonic infarction; and more than 10% to 15% develop late ischemic strictures (100,101,102). Approximately 15% to 20% of patients develop gangrene or perforation, either as an initial presentation of the disease or within a variable time following symptom onset. Sigmoidoscopic findings can include striking mucosal edema, ulceration, nodularity, hypervascularity, friability, and pseudomembranes. Ulcers may or may not be present. These features evolve rapidly from one examination to the next.
The most severe form of injury, transmural infarction, requires resection of the affected segment; otherwise, perforation or death will ensue. Almost half of patients with colonic ischemia have a transient, reversible form of the disease; 20% to 25% have chronic colitis or deep colonic infarction; and more than 10% to 15% develop late ischemic strictures (100,101,102). Approximately 15% to 20% of patients develop gangrene or perforation, either as an initial presentation of the disease or within a variable time following symptom onset. Sigmoidoscopic findings can include striking mucosal edema, ulceration, nodularity, hypervascularity, friability, and pseudomembranes. Ulcers may or may not be present. These features evolve rapidly from one examination to the next.
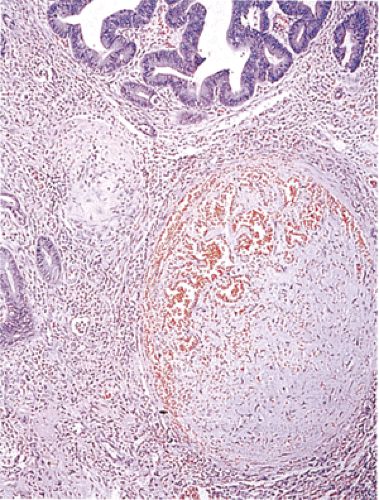 FIG. 13.66. A recanalized thrombus is present in a submucosal vessel. The overlying mucosa is neoplastic. |
Pathophysiology of Ischemic Injury
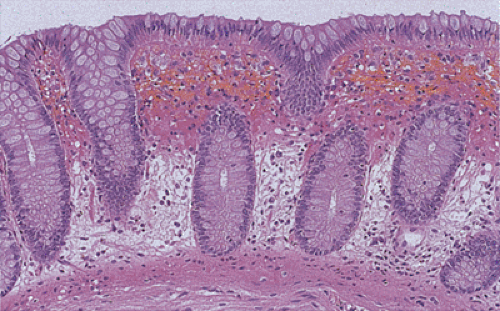
As in the small intestine, the physiology of the circulation is such that a countercurrent exchange mechanism shunts oxygen into the lower mucosa at the expense of its upper portion (103). Consequently, the normally relatively hypoxic upper mucosa, which is particularly vulnerable to the effects of hypoperfusion, becomes necrotic, while the lower mucosa remains intact (Fig. 13.68). The degree of ischemic damage depends on the adequacy of the collateral circulation, on vascular autoregulatory mechanisms, and on tissue resistance to hypoxia. The extent and pattern of disease also depends on the anatomy of the blood supply, duration of the hypoxic episode, and bacterial population within the bowel lumen. The mildest lesions affect only the mucosa, and one sees superficial necrosis and hemorrhage. If reflow occurs, neutrophils infiltrate the tissues (see Chapter 6). There are three phases in the colonic reaction to ischemia: (a) acute with hemorrhage and necrosis, (b) repair with the formation of granulation tissue and fibrosis, and (c) the formation of structures and other complications. A number of disorders and damaging agents cause colonic injury via an ischemic mechanism (Table 13.6).
Large Vessel Occlusion Versus Small Vessel Occlusion
The severity of the lesions varies from microscopic focal damage to involvement of the entire colon. The size of the lesion correlates directly with the size of the affected vessels. Colonic ischemia results from occlusion of large vessels such as the mesenteric arteries or diseases or thromboemboli, which affect the intramural circulation with its smaller vasculature. Obstruction or diffuse vasospasm involving the major arterial supply of the large intestine results in severe ischemia to extensive regions of the colon or rectum. More distal obstruction of the colonic blood supply results in segmental ischemia or infarction. Only rarely does one see complete vascular occlusion. The superior branch of the mesenteric artery is more prone to undergo embolization than the inferior mesenteric artery due to the smaller caliber and more acute angle of takeoff of the inferior mesenteric artery (103). The ostium may undergo thrombosis at its origin from the aorta due to aortic atherosclerosis, but the presence of a collateral circulation often maintains an adequate perfusion (104). Small vessel disease results from microangiopathies as seen in patients with hypertension, arteriolosclerosis, diabetes, vasculitis, radiation injury, or coagulation diatheses. Vascular obstruction also results from nonocclusive factors or obstruction of the vessels by external forces, as discussed in Chapter 6.
Sites of Injury
Ischemia affects the colon more commonly than it affects the small bowel, probably because of the vast collateral vascular network that exists in the small bowel (105). Colonic regions with poor collateral circulation, particularly around the splenic flexure and the rectosigmoid, are especially vulnerable (106). The superior and inferior mesenteric arteries communicate via the left branch of the middle colic artery and the ascending branch of the left colic artery at the arc of Riolan (marginal artery of Drummond). Approximately 5% of individuals have underdeveloped or absent connections between the two arterial systems at the splenic flexure, making them susceptible to ischemic damage (107).
Gross Features
The gross features of large intestinal ischemia resemble those of small intestinal ischemia. The changes vary depending on
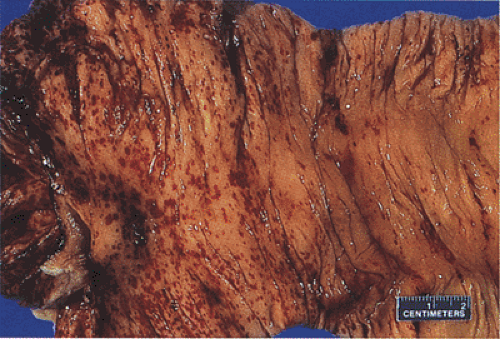
the severity and depth of the ischemic process, whether the event is acute or chronic, and the stage of healing that may have occurred (Figs. 13.69, 13.70, and 13.71). In early ischemia (usually seen during endoscopic procedures), the mucosa appears paler than normal but an increased vascular pattern with patchy, granular, swollen areas may be present. Bowels with transmural infarction may either appear pale or hemorrhagic depending on the circumstances. If hemorrhage has occurred, the bowel appears darkish blue or purple with blood in the bowel lumen. The wall may appear thinner than normal. As the disease becomes more severe, it extends into the submucosa causing edema and hemorrhage. Eventually,
the mucosa sloughs and areas of grayish green necrosis develop, producing pseudomembranes. Ulcers may be present. These may be only superficial or they may be deep and linear. Occasionally the ulceration is extensive and confluent. Perforation may occur. The features may mimic fulminant UC or toxic megacolon. The ischemic changes exhibit a geographic distribution, often with a sharp line of demarcation between the involved and uninvolved mucosa (Fig. 13.70).
the severity and depth of the ischemic process, whether the event is acute or chronic, and the stage of healing that may have occurred (Figs. 13.69, 13.70, and 13.71). In early ischemia (usually seen during endoscopic procedures), the mucosa appears paler than normal but an increased vascular pattern with patchy, granular, swollen areas may be present. Bowels with transmural infarction may either appear pale or hemorrhagic depending on the circumstances. If hemorrhage has occurred, the bowel appears darkish blue or purple with blood in the bowel lumen. The wall may appear thinner than normal. As the disease becomes more severe, it extends into the submucosa causing edema and hemorrhage. Eventually,
the mucosa sloughs and areas of grayish green necrosis develop, producing pseudomembranes. Ulcers may be present. These may be only superficial or they may be deep and linear. Occasionally the ulceration is extensive and confluent. Perforation may occur. The features may mimic fulminant UC or toxic megacolon. The ischemic changes exhibit a geographic distribution, often with a sharp line of demarcation between the involved and uninvolved mucosa (Fig. 13.70).
TABLE 13.6 Disorders with an Ischemic Basis | |
|---|---|
|
In patients who survive the acute event, the wall will appear thickened, fibrotic, and contracted. Strictures covered either by an ulcerated granular-appearing mucosa or by a re-epithelialized mucosa may be present. The gross features of chronic disease may mimic Crohn disease or a neoplasm.
Resection specimens for ischemia should be handled in a regular way so as to evaluate the extent of the ischemic damage, the viability of the margins, and, if possible, the etiology. The vessels should be examined both macroscopically and microscopically to detect evidence of thrombosis or vascular occlusion. Handling an intestinal resection specimen is discussed in detail in Chapter 6.
Transitory Ischemia
There is a form of ischemia that is transient and reversible. Usually the patients present with mild GI complaints and no obvious cardiovascular event precedes or appears to precipitate the ischemia. The patients may also have crampy abdominal pain and rectal bleeding. The diagnosis is usually a clinical one. Therefore, the lesion is not typically resected or biopsied because the bowel will heal if the damage is minimal. However, sometimes biopsies are performed to elucidate the cause of the pain or the bleeding. The biopsies may show the acute ischemic changes described below or they may show evidence of mucosal regeneration.
Acute Ischemia
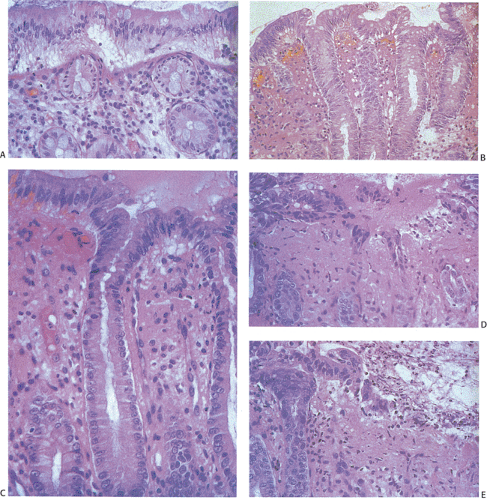
The earliest ischemic lesions vary in their histologic appearance depending on the size of the occluded vessel. If the vessel is small and the degree of ischemia is minimal, the first changes affect the superficial parts of crypts and spare all but the superficial lamina propria. With continuing injury the damage progressively involves more of the crypts, eventually reaching the base. Biopsies in the acute stage show variable mucosal necrosis with loss of surface epithelium (Figs. 13.68 and 13.72, 13.73, 13.74), dilated capillaries and lymphatics, mucosal edema with hemorrhage and fibrin deposition (Fig. 13.72), and crypt dropout. In severe cases, the crypts appear to be bursting, dilated, and lined by an attenuated epithelium (Fig. 13.73). The crypts become filled with mucus and inflammatory debris. One may also see crypt abscesses. The epithelial loss leaves dilated, ghostlike, bare crypts supported by a congested lamina propria (Fig. 13.73). Goblet cells become mucin depleted and neutrophils infiltrate the mucosa. The muscularis mucosae becomes frayed. Early in the process neutrophils are relatively sparse.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree