Chapter 28 Endoscopic Palliation of Malignant Dysphagia and Esophageal Fistulas
![]() Video related to this chapter’s topics: Esophageal Stent Placement
Video related to this chapter’s topics: Esophageal Stent Placement
Introduction
Annually, cancer of the esophagus and gastroesophageal junction (GEJ) is diagnosed worldwide in more than 500,000 patients, which makes it the eighth most common malignancy and sixth most common cause of cancer mortality.1 It is difficult to determine the true incidence because cancer of the GEJ is classified sometimes as gastric cancer and sometimes as esophageal cancer. In clinical practice, this distinction is unimportant because the curative and palliative options for treatment are the same for both adenocarcinoma of the esophagus and adenocarcinoma of the GEJ.
Overall, cancer of the esophagus and GEJ has a poor prognosis with a 5-year survival rate of less than 20% in the Western world.2 This poor prognosis is at least partly due to the fact that more than 50% of patients with carcinoma of the esophagus or GEJ already have inoperable disease at presentation.3 Most of these patients require palliative treatment to relieve progressive dysphagia or to treat associated problems such as the presence of a fistula.
Epidemiology
Squamous Cell Carcinoma
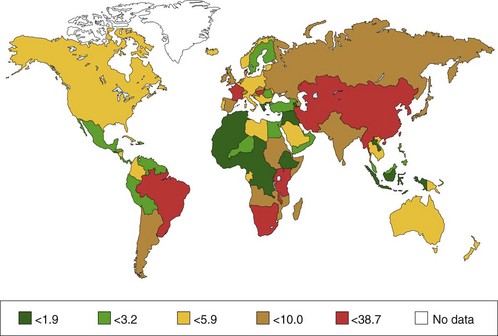
The incidence of squamous cell carcinoma (SCC) varies from country to country; also, it may occur more often in certain regions within a country. About two-thirds of new cases of SCC are detected in China (47%) and Central Asia (19%); this is known as the Central Asia Esophageal Cancer Belt. The incidence of SCC in this area ranges from 19 per 100,000 in Azerbaijan to 340 per 100,000 in northern China. Other areas of relatively high risk are southern and eastern Africa, south central Asia, and (in men only) Japan. The incidence of SCC in Western Europe and the United States is much lower (i.e., 53 to 86 per 100,000). In Western countries, SCC of the esophagus is mainly found in older people with the highest incidence between 50 and 70 years of age. Esophageal cancer is more common in men in most areas—the sex ratio is 7 : 1 in Eastern Europe—although in the high-risk areas of Asia and Africa, the sex ratio is much closer to unity.1 The distribution between men and women is 3 : 1 to 4 : 1.3
Adenocarcinoma
Until about 1970, more than 90% of esophageal cancers were SCCs. However, population-based studies have shown a large increase in the incidence of adenocarcinoma of the esophagus and GEJ over the last 30 years in North America and Western Europe, especially among white men but also in white women to a lesser degree.4,5 In men, the incidence of adenocarcinoma of the esophagus and GEJ has surpassed SCC.6 In the United States, the annual rates of esophageal adenocarcinoma per 100,000 population are 7.8% for white men and 6.5% for women, increasing from 0.7 during 1974–1976 to 3.2 during 1992–1994, an increase of more than 350%.5 The same trend, although occurring less rapidly, has been reported in other areas, including Australia, New Zealand, and Western Europe.7
It is generally believed that the increase in esophageal adenocarcinoma is related to an increase in the incidence of Barrett’s esophagus. In a report from The Netherlands, van Soestal and colleagues8 found that the incidence of new diagnoses of Barrett’s esophagus increased from 14.3 per 100,000 person-years in 1997 to 23.1 per 100,000 person-years in 2002. The number of upper gastrointestinal endoscopies decreased from 7.2 per 1000 person-years to 5.7 per 1000 person-years over the same time period. The rate of detection of Barrett’s esophagus increased over the same years from 1.4 to 42.7 (16.5 if only cases with histologic confirmation were included) per 1000 endoscopic procedures.
Multiple reports confirm that adenocarcinoma of the esophagus and GEJ occurs more frequently in white men. The distribution between men and women is 4 : 1. Most patients with esophageal adenocarcinoma are older individuals with a peak incidence around age 65 years.9 The worldwide distribution of esophageal cancer (for men) is shown in Fig. 28.1.
Pathogenesis
Squamous Cell Carcinoma
Smoking and Alcohol
The most important risk factors for SCC in Western Europe and the United States are smoking and alcohol intake. Risk of SCC is increased by a factor of 5 for moderate smokers and a factor of 10 for heavy smokers. It has been shown that alcohol intake and smoking are independent risk factors for the development of esophageal SCC.10
Food
In Hong Kong, a correlation has been established between the use of pickled vegetables and the development of SCC. This correlation was found to be caused by herbs that were used for these vegetables, which were often contaminated with toxic fungi.11
Other Factors
Prior radiation therapy has been associated with an increased risk of SCC. A study showed that patients who underwent radiation therapy for breast cancer more than 10 years ago had an increased risk of developing SCC in the esophagus.12
Hot drinks, particularly tea in certain areas in Asia, such as the Golestan province in northern Iran, are associated with an increased risk of developing SCC. The suggested mechanism is chronic irritation of the esophageal mucosa caused by the hot drinks.13
The role played by human papillomavirus (HPV) is unclear. In South Africa, where the incidence of SCC is high, HPV DNA was detected in more than 50% of cancers.14 In contrast, in the Netherlands, the presence of HPV in SCC is rare.15
Disorders Associated with Increased Risk of Squamous Cell Carcinoma
Achalasia
In a cohort study from Sweden, in which 1062 patients with achalasia were followed, the risk of SCC was increased by a factor of 16 after a follow-up of 9864 patient-years.16 Because most tumors were detected at an advanced stage, a curative resection was possible in only a few patients. Nonetheless, follow-up with endoscopic surveillance in patients with long-standing achalasia has been suggested. It needs to be determined whether this approach is cost-effective.
Caustic Ingestion
The incidence of esophageal SCC is increased by a factor of 1000 to 3000 in patients with a stricture in the esophagus caused by a caustic ingestion. The risk of developing a malignancy is probably highest after the ingestion of lye.17 The mean time between ingestion of a corrosive agent and the development of SCC is 30 to 40 years.
Head and Neck Cancer
SCC of the esophagus and the hypopharynx is associated with smoking and alcohol intake. Of patients with head and neck cancer, 1% to 8% also have esophageal cancer or develop it later on.18 The risk of esophageal cancer is increased by a factor of 3 to 10 in patients with head and neck cancer.
Adenocarcinoma
Gastroesophageal Reflux Disease
Lagergren and coworkers19 found a direct association between reflux and adenocarcinomas, rather than the presumed sequence of reflux disease leading to Barrett’s esophagus and this condition leading to adenocarcinoma. The esophageal adenocarcinoma risk was 7.7 times increased in individuals with heartburn and acid reflux occurring at least once a week. For people with severe symptoms for 20 years or longer, the risk was 43.5 times increased for esophageal adenocarcinoma but only 4.4 times increased for adenocarcinoma of the gastric cardia. There was no correlation with SCC.
Barrett’s Esophagus
Barrett’s esophagus is a disorder of the distal esophagus in which the squamous epithelium is replaced by metaplastic columnar epithelium. Barrett’s esophagus is a complication of long-standing gastroesophageal reflux disease.20 A causal relationship between Barrett’s esophagus and the development of esophageal adenocarcinoma has been established.
In older reports, the risk of esophageal adenocarcinoma in long-segment Barrett’s esophagus was 30 to 52 times greater than the normal population. Cancer was diagnosed at a median rate of about 1 per 100 patient-years of follow-up. These reports were often based on a short period of follow-up, however, with the possibility of including prevalent cancers as incidence cases, and may have overestimated the cancer risk. More recent reports with longer follow-up times found 1 cancer per 180 to 2200 patient-years of follow-up.21 The prevalence of Barrett’s esophagus in consecutive patients undergoing endoscopy for any clinical indication ranges from 0.3% to 2%.22 Several studies have shown that Barrett’s esophagus is a disorder of white patients and is mainly found in Western Europe. The distribution between men and women is 2.5 to 4 : 1.9
Clinical Features
Local effects of esophageal carcinoma include dysphagia, odynophagia, coughing, regurgitation, vomiting, or a vague discomfort in the back of the throat. At the time of diagnosis, tumor length is mostly more than 4 cm, and patients often already have 6 weeks to 4 months of dysphagia with accompanying substantial weight loss.23 Dysphagia is not diagnostic of an esophageal malignancy because nonmalignant diseases also manifest with dysphagia such as achalasia or peptic strictures caused by reflux esophagitis. In cases of rapidly progressive dysphagia and weight loss, however, the suspicion of a malignant tumor of the esophagus or GEJ is high. Dysphagia is a late symptom of an esophageal malignancy. Only when a mass lesion has come to a critical size does it impair the passage of food. At this time, the tumor has usually invaded the deeper layers of the esophageal wall, making the prognosis poor.
Patients with esophageal cancer may develop iron deficiency anemia. Bleeding from the tumor is usually a slow, occult process. Sometimes patients experience frank hemorrhage. Rarely, when the tumor invades the aorta or another major vessel, a patient may experience exsanguination, which is a frequent cause of death.23
Pathology
Squamous Cell Carcinoma
Of SCCs, 24% occur in the upper third, 47% occur in the middle third, and 29% occur in the lower third of the esophagus.24 It has been shown that SCC develops from low-grade or high-grade dysplasia to intraepithelial carcinoma and finally invasive esophageal carcinoma. Endoscopic follow-up in 327 Chinese patients with high-grade dysplasia showed that SCC was diagnosed at a median rate of 4 cases per 100 patient-years of follow-up.25 In the Western world, less than 10% of patients with SCC are diagnosed at an early stage.24
Adenocarcinoma
Adenocarcinomas of the esophagus and GEJ are located in the distal esophagus and proximal stomach. There is also clear evidence for a dysplasia-carcinoma sequence in Barrett’s esophagus, whereby Barrett’s esophagus without dysplasia progresses to low-grade dysplasia, high-grade dysplasia, and ultimately carcinoma.26 During a mean follow-up of 3 to 5.2 years, progression from Barrett’s esophagus without dysplasia to low-grade dysplasia occurred in 12% to 18% of patients, and progression from low-grade to high-grade dysplasia or adenocarcinoma occurred in 10% to 25% of patients.27,28 Progression from high-grade dysplasia to carcinoma occurs in 17% to 66% of patients over 0.75 to 9 years.29,30 The distribution of the grade of dysplasia in transversal studies of patients with Barrett’s esophagus is 80% no dysplasia, 18% low-grade dysplasia, and 2% high-grade dysplasia or adenocarcinoma.31,32
Given the dismal prognosis among patients with symptomatic esophageal cancer, guidelines from the American College of Gastroenterology33 recommend endoscopic surveillance of patients with Barrett’s esophagus in an attempt to prevent death from adenocarcinoma. Retrospective studies have found that patients whose esophageal adenocarcinoma was detected in a surveillance program presented at an earlier stage and had better 5-year survival rates than patients without surveillance who presented with cancer.34 A more recent study showed that less than 5% of patients who presented with esophageal adenocarcinoma actually underwent endoscopic surveillance.35
Esophageal cancer grows by intraesophageal spread, direct extension, and lymphatic and hematogenous metastases. The tumor typically invades adjacent structures, and lymph node metastases range from 40% to 70%. Because esophageal lymph node flow is bidirectional, sites of nodal metastases are many. Distant metastases, particularly to liver, lung, and bone, are present in 25% to 30% of patients at diagnosis.36
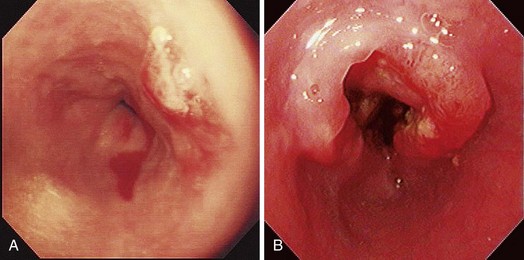
Early esophageal carcinomas are usually slightly elevated, coarse, or polypoid with denuded epithelium at endoscopy (Fig. 28.2A). The gross appearance of SCCs and adenocarcinomas is practically indistinguishable. Adenocarcinoma of the esophagus, especially in its early stage, can be distinguished from SCC by the presence of Barrett’s esophagus. If esophageal adenocarcinoma is advanced, however, it is often impossible to detect Barrett’s esophagus because the tumor has presumably overgrown its precursor. The macroscopic features of advanced esophageal cancers can be ulcerative, stenotic, polypoid, or a combination of these (Fig. 28.2B).
Treatment
The preferred treatment for esophageal cancer is surgical resection. Resection of the esophagus with a gastric pull-up or a colonic interposition is an invasive procedure, however, with significant morbidity and mortality.36 A discussion of the different surgical techniques, long-term results, and complications after surgery is beyond the scope of this chapter. In the past decade, endoscopic methods have been developed to remove early cancers in the esophagus nonsurgically. Indications and contraindications for endoscopic treatment of early esophageal cancer are discussed in Chapter 27.
Various palliative techniques are currently available (Table 28.1). The main options can be divided into nonendoscopic modalities, of which chemoradiation therapy is most commonly used, and endoscopic procedures, of which placement of a self-expanding metal stent to relieve obstruction resulting from a malignant stricture in the esophagus is the most frequently used technique. Some of the endoscopic procedures for palliation of malignant dysphagia are discussed.
Table 28.1 Palliative Modalities for Esophageal Carcinoma
| NONENDOSCOPIC TECHNIQUES |
| ENDOSCOPIC TECHNIQUES |
Self-Expanding Metal Stents
Placement of a self-expanding metal or plastic stent is a frequently used method for palliation of malignant dysphagia. Since 1990, more than 130 studies have been published on the outcome of metal stent placement for palliation of malignant dysphagia and esophageal fistulas.37–39
Metal Stents versus Rigid Plastic Endoprosthetics
Several randomized trials have compared metal stents with prosthetic tubes.40–45 These studies have shown that placement of a metal stent is associated with fewer procedure-related complications than placement of a prosthetic tube.41,42,44,45 In one study, metal stents were also more effective in improving dysphagia.43 Studies on cost-effectiveness have shown that, despite the high initial purchase cost, metal stents were more cost-effective than prosthetic tubes because of a shorter hospital stay for procedures for stent-related complications.40,41,43,46
Covered versus Uncovered Metal Stents
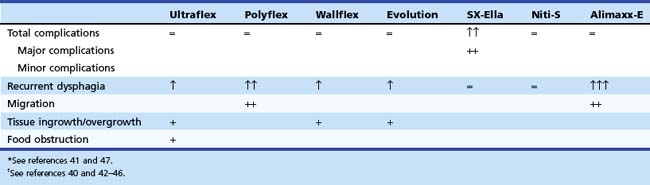
In a prospective randomized trial by Vakil and coworkers,47 covered and uncovered Ultraflex (Boston Scientific, Natick, MA) stents were compared in 62 patients with obstructing tumors at the GEJ (Figs. 28.3 and 28.4). Tumor ingrowth or overgrowth was significantly more common in the uncovered stent group (9 of 30 [30%]) than in the covered stent group (1 of 32 [3%]). Stent migration was not different between the two treatment groups (uncovered stent, 2 of 30 [7%], vs. covered stent, 4 of 32 [12%]). Covered stents apparently give better long-term palliation of malignant dysphagia than uncovered stents.
Currently Available Covered Metal Stents
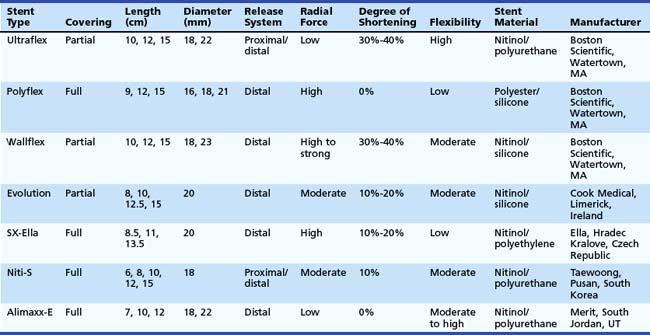
An ideal stent does not exist. However, all available covered metal stents meet some of these criteria (Table 28.2).
The Ultraflex stent consists of a knitted nitinol wire tube, and the covered version has a polyurethane layer that covers the midsection of the stent extending to within 1.5 cm of either end of the stent (see Figs. 28.3 and 28.4). The stent has a proximal flare with two sizes: 28 mm (distal diameter 23 mm) and 23 mm (distal diameter 18 mm). The Ultraflex stent has an easy-to-use delivery system of the stent and can be deployed gradually from the proximal to the distal end or vice versa. The degree of shortening after stent placement is 30% to 40%. The radial force of the Ultraflex stent is the lowest among currently available metal stents. Partial obstruction of the stent can occur in stents that are sharply angulated after passing across the GEJ.
The Polyflex stent (Boston Scientific) is a silicone device with an encapsulated monofilament braid made of polyester. The meshes are completely covered by a silicone layer with a smooth inner surface and a more structured outer surface (see Fig. 28.4). The edges of the monofilaments are protected with silicone to avoid impaction or tissue damage at the proximal and distal ends. The stent has a proximal flare of 25 mm, 23 mm, and 21 mm and body diameter of 21 mm, 18 mm, and 16 mm. It is available in three lengths: 9 cm, 12 cm, and 15 cm. The stent needs to be loaded in the introducer sheath before placement. This introduction device has a diameter of 14 mm, 13 mm, and 12 mm. This is the largest stent system compared with other systems and the system is rigid; investigators have suggested that the relatively high occurrence of perforations after Polyflex stent placement is at least partly due to these characteristics.48 In addition, the stent is less suitable for angulated strictures because the distal dilator is short. The inappropriate forced transmission of such an introduction sheath may complicate its passage across angulated strictures. Finally, because the stent has only a mesh on the outside and no other antimigration properties, it is associated with an increased risk of stent migration.48 The Polyflex stent is more frequently used for benign strictures in the esophagus than for malignant strictures.49
The Wallflex stent (Boston Scientific) consists of a wire braided construction and is partially covered with nitinol extending to within 1.5 cm of either end of the stent (see Fig. 28.4). The stent has a proximal and distal flare with two sizes: 28 mm (body diameter 23 mm) and 23 mm (body diameter 18 mm). The degree of shortening after Wallflex stent placement is considerable (i.e., 30% to 40%). The radial force of the Wallflex stent is one of the highest among currently available metal stents. Clinical experience is limited to one prospective follow-up study showing favorable results with the partially covered Wallflex stent.50
The Evolution stent (Cook, Limerick, Ireland) is constructed of a single woven nitinol wire. The stent has an internal and external silicone coating and uncoated flanges on both ends (see Fig. 28.4). The body diameter of the stent is 20 mm, and the flange diameter is 25 mm on both ends. The stent is available in four lengths: 8 cm, 10 cm, 12.5 cm, and 15 cm. The stent slightly foreshortens because of its design. A pistol-grip delivery system handle allows step-by-step stent deployment or recapturing. Initial studies have shown that this stent type is safe and effective in treating malignant esophageal strictures.51
The SX-Ella stent (Ella, Hradec Kralove, Czech Republic) is made of nitinol and a single braided wire. To decrease the risk of migration, the SX-Ella stent has a flip-flop type of antimigration ring that is circumferentially attached to the proximal stent portion (see Fig. 28.4). This ring functions as a circular hook that prevents migration; however, the ring is flexible and everts when the traction force is too strong. The stent flares to 25 mm at its proximal and distal ends with a body diameter of 20 mm. It is available in lengths of 85 mm, 110 mm, and 135 mm. Initial studies showed that the antimigration ring did not reduce stent migration, but the stent was associated with an increased risk of hemorrhage. In addition to hemorrhage, severe pain and fistula formation at the upper end of the SX-Ella stent were frequently observed. In normal circumstances, stents exert some pressure on the tumor and the normal mucosa of the esophagus to fixate the stent to the esophageal wall to reduce migration risk. In case of the SX-Ella stent, this effect may be more pronounced because of the pressure effect of the antimigration ring, particularly when it is flipping in and out for its antimigration effect.52
The Niti-S stent (Taewoong, Pusan, South Korea) has a double-layer configuration over its entire length, consisting of an inner polyurethane layer and an outer uncovered nitinol wire (see Fig. 28.4). This double layer was designed to reduce the risk of stent migration. The stent flares to 26 mm at its proximal and distal ends and has a body diameter of 18 mm. It is available in five lengths: 6 cm, 8 cm, 10 cm, 12 cm, and 15 cm. It has both a proximal and a distal release system.48,53
The Alimaxx-E stent (Merit, South Jordan, UT) is also made of nitinol, and it is fully covered with polyurethane to resist tissue ingrowth (see Fig. 28.4). This stent has a proximal flare of 27 mm and 23 mm, a luminal diameter of 22 mm and 18 mm, and a distal flare of 25 mm and 21 mm. The outward force of the stent is most pronounced at the body. The stent also has a pistol type of release system, similar to the Evolution stent (see earlier). In the initial versions of this stent, it could be introduced over a guidewire (Alimaxx-E GW system) but also under direct vision using a delivery system in which the delivery catheter fitted over a small-caliber endoscope (Alimaxx-E DV system). The latter system featured a window at the end of the introduction sheath to view stent deployment. The size of the introduction catheter of the Alimaxx-E DV was 30 Fr, whereas the size of the introduction catheter of the Alimaxx-E GW was 22 Fr. Because the DV system was difficult to manipulate owing to its size and stiffness, and perforations were noted with this system, this system was withdrawn from the market. Another issue was that the initial version of the Alimaxx-E stent had 20 antimigration struts to prevent stent migration; this was increased to 45 antimigration struts, however, because the migration rate was more than 30% with the original stent design.54
Comparison of Different Types of Metal Stents
Four prospective randomized trials48,55–57 have compared the outcome of currently available stent designs of different types of metal stents. In one study, 101 patients with unresectable esophageal carcinoma were randomly assigned to placement of a Polyflex (n = 47) or a partially covered Ultraflex (n = 54) stent.55 Patients with GEJ malignancy were excluded. Placement was equally successful with both stent designs: A Polyflex stent was placed in 46 (98%) patients, and an Ultraflex stent was placed in 54 (100%) patients. There were no significant differences in dysphagia improvement between the two stent designs (after 1 week improvement by at least one grade in 100% of the Polyflex group and in 94% of the Ultraflex group). Major complications were observed in 48% of the Polyflex group and in 33% of the Ultraflex group. Intraprocedural perforation occurred in one Polyflex patient and one Ultraflex patient. Two Polyflex patients had postprocedural hemorrhage. Recurrent dysphagia occurred in 20 (44%) patients with a Polyflex stent and 18 (33%) patients with an Ultraflex stent because of tumor overgrowth, stent migration, hyperplastic granulomatous reaction, or food bolus impaction. Multivariate analysis showed a significantly higher complication rate with Polyflex stents than with Ultraflex stents (odds ratio 2.3, 95% confidence interval 1.2 to 4.4). Median survival was similar: 134 days with Polyflex stents and 122 days with Ultraflex stents. The authors concluded that palliation of dysphagia was not different between the two stents. Significantly more complications, especially late stent migration, were observed in the Polyflex group.
In another prospective study, 125 patients with dysphagia from inoperable carcinoma of the esophagus or gastric cardia were randomly assigned to placement of an Ultraflex stent (n = 42), Polyflex stent (n = 41), or Niti-S stent (n = 42).48 Stent placement was technically successful in all patients with an Ultraflex stent, in 34 of 41 (83%) patients with a Polyflex stent, and in 40 of 42 (95%) patients treated with a Niti-S stent (P = .008). The dysphagia score improved in all patients. There were no differences in major complications among the three stent types. Recurrent dysphagia, caused by tissue ingrowth or overgrowth, migration, or food obstruction, was significantly different between patients with an Ultraflex stent and patients with a Polyflex stent or Niti-S stent (22 [52%] vs. 15 [37%] vs. 13 [31%]; P = .03). Stent migration occurred more frequently with Polyflex stents, whereas tissue ingrowth or overgrowth was seen more frequently with partially covered Ultraflex stents and, to a lesser degree, Niti-S stents. No differences were found in survival (median survival Ultraflex stent, 132 days, vs. Polyflex stent, 102 days, vs. Niti-S stent, 159 days) among the three stent types. It was concluded that all three stents were safe and offered adequate palliation of dysphagia from esophageal or gastric cardia cancer. Nonetheless, Polyflex stents seemed the least preferable stent type in this patient group because placement of this device is technically demanding and associated with a high rate of stent migrations.
In a prospective trial, 100 patients were randomly assigned to one of three types of covered metal stents: the Ultraflex stent, the Flamingo Wallstent, and the Z-stent.56 There were no significant differences in dysphagia improvement and the occurrence of complications or recurrent dysphagia, although there was a trend toward more complications with the Z-stent (Ultraflex stent, 8 of 34 [24%]; Flamingo Wallstent, 6 of 33 [18%]; and Z-stent, 12 of 33 [36%]; P = .23). In another prospective trial, the Ultraflex stent and the Flamingo Wallstent were compared in patients with distal esophageal cancer.57 The two stent types were equally effective in the palliation of dysphagia in this patient group, and the complication rate associated with their use was comparable (Ultraflex stent, 7 of 31 [23%], and Flamingo Wallstent, 5 of 22 [23%]).
From these data, it can be concluded that there are only minor differences between the most commonly used stent types. The choice of stent in patients with a malignant stricture in the esophagus or gastric cardia should be determined by the location and the anatomy of the malignant stricture on the one hand and the specific characteristics of the stent on the other hand (Table 28.3; see Table 28.2). Based on the currently available results, caution is needed when using Polyflex stents (migration, robust introduction device), SX-Ella stents (complications owing to the antimigration ring), or Alimaxx-E stents (migration) for this indication.
Metal Stents for Tumors of the Gastroesophageal Junction
Because the incidence of adenocarcinoma of the distal esophagus is increasing rapidly,4,5 the deployment of metal stents across the GEJ is likely to increase. However, stent placement for tumors of the distal esophagus and GEJ constitutes a particular problem. Compared with stents placed for more proximally located esophageal tumors, these procedures provide inferior palliation and have higher complication rates.58 Migration is more likely with stents placed across the GEJ than with stents placed for more proximally located tumors because the distal part of the stent projects freely into the fundus of the stomach, and this part cannot fix itself to the wall.
How can migration of stents be prevented? Following are some considerations:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree