Fig. 8.1
Intestinal sterol absorption and secretion. Sterols including free cholesterol (FC) and free plant sterols (PS) from diet and bile are mixed with phospholipids (PL) and bile acids (BA) to form micelles. FC and PS solubilized in mixed micelles are transported into absorptive enterocytes via an NPC1L1 -dependent and ezetimibe -inhibitable mechanism. FC is delivered to the ER for esterification by acyl-CoA: cholesterol acyltransferase-2 (ACAT2 ) to form cholesterol esters (CE) that is then packaged into nascent lipoprotein particles (nLP) and secreted as a constituent of chylomicrons . PS and FC that escapes ACAT2 esterification may be directly transported to nascent HDL (nHDL) through basolateral ABCA1 , or back to the gut lumen via ABCG5/G8 (Adapted from Brown and Yu [2])
The regulation of NPC1L1 is incompletely understood. Some data suggest that cellular cholesterol availability influences NPC1L1 expression, and that activation of many nuclear receptors play regulatory roles. Human sequence polymorphisms in NPC1L1 have been identified that affect cholesterol absorption efficiency, plasma LDL-cholesterol levels, and sensitivity to ezetimibe treatment. These polymorphisms alter NPC1L1 subcellular localization, glycosylation, or protein stability.
A second complex plays a key role in the transport of cholesterol and plant sterols from the enterocyte across the brush-border membrane into the intestinal lumen (Fig. 8.1). ABCG5 and ABCG8 are genes on chromosome 2 arranged in a head-to-head orientation with less than 400 base pairs between their respective start codons. The two genes code for two distinct proteins, sterolin-1 and sterolin-2 , that must heterodimerize to transport sterols. Both proteins contain an ATP-binding sequence near the N-terminus and six transmembrane domains, and the complex functions as an ATP-dependent cholesterol efflux pump delivering sterols from the enterocytes into the intestinal lumen for disposal in feces. The two proteins heterodimerize in the endoplasmic reticulum, traffic through the Golgi, and subsequently move to the apical plasma membrane. ABCG5 and ABCG8 undergo N-glycosylation, and glycosylation at Asn-619 in ABCG8 is critical for proper trafficking of the complex.
The ABCG5/ABCG8 complex is largely responsible for the greater intestinal absorption of cholesterol compared with a variety of dietary plant sterols. β-sitosterolemia is a genetic disease characterized by increased intestinal absorption and diminished biliary secretion of cholesterol and plant sterols such as β-sitosterol. Affected patients have accumulation of cholesterol and plant sterols in the plasma and various tissues and often suffer from premature atherosclerotic heart disease. β-sitosterolemia is caused by a mutation in either ABCG5 or ABCG8, with the majority of mutants causing impaired transport of the heterodimer from the endoplasmic reticulum to the plasma membrane.
ABCG5 and ABCG8 both appear to be regulated primarily at the level of gene transcription. The sterol-sensing transcription factors LXRα and LXRβ up-regulate expression of both ABCG5 and ABCG8, and effects of other transcription factors have been identified.
Scavenger receptor class B type 1 (SR-B1) is thought to play a role in cholesterol homeostasis mainly through its mediation of the selective uptake of HDL cholesteryl esters into the liver . SR-B1, however, is also abundantly expressed in the small intestine where it is found in both the brush-border and basolateral membranes (Fig. 8.1). SR-B1 is expressed more in the proximal than distal small bowel, paralleling the site of most cholesterol absorption. SR-B1 may be mediate some cholesterol uptake across the brush-border membrane, but may have more important roles in the intestinal uptake of circulating HDL-associated cholesterol esters and subsequent transport across the apical membrane for disposal in the feces, and/or the secretion of cholesterol-containing lipoproteins across the basolateral enterocyte membrane.
2.5 Cholesterol Esterification, Incorporation into Lipoproteins, and Secretion
Within the enterocyte, 70–90 % of the absorbed cholesterol is esterified with fatty acids. Various lipid binding proteins may be involved in directing the absorbed cholesterol to the endoplasmic reticulum. The principal enzyme responsible for cholesterol esterification is acyl CoA:cholesterol acyl transferase 2 (ACAT2 ) that is present in the endoplasmic reticulum. Sitosterol and other plant sterols are less effective substrates for ACAT2 than cholesterol. The microsomal triglyceride transfer protein (MTTP) transfers cholesteryl esters to the nascent chylomicron particle, and chylomicrons are the principal lipoprotein delivering cholesterol into the circulation. Within chylomicrons and other intestinal lipoproteins, cholesteryl esters are incorporated into the oily lipoprotein core, whereas free cholesterol is present on the surface of the particle.
Another route of cholesterol exit from the enterocyte may be via incorporation into HDL. A basolateral cholesterol efflux pump ABCA1 facilitates the transfer of cholesterol to an apolipoprotein A-I acceptor molecule forming discoidal HDL particles (Fig. 8.1). The amount of cholesterol absorbed via this pathway appears to be small, but ABCA1 may be a key role in HDL production. Patients with Tangier disease with mutations in ABCA1 have virtual absence of plasma HDL and increased enterocyte cholesterol content. ABCA1 is regulated by the transcriptional factor LXRα.
3 Phospholipid
Most of the phospholipid that the intestine absorbs originates from biliary secretion (10–20 g/day) rather than the diet (1–2 g/day). In bile, phospholipid is present in mixed micelles along with bile acids and cholesterol. Phosphatidylcholine is the major phospholipid in the diet and in bile along with small amounts of phosphatidylethanolamine , phosphatidylserine , and phosphatidylinositol . In the intestinal lumen, the phosphatidylcholine distributes between the lipid droplets and the micellar phase, but favors the micelles.
3.1 Digestion of Phosphatidylcholine
The major enzyme responsible for the digestion of phosphatidylcholine is pancreatic phospholipase A 2 (Fig. 8.2). This enzyme, which catalyzes the breakdown of phosphatidylcholine to lysophosphatidylcholine and fatty acid, is secreted as a zymogen and activated by tryptic cleavage of an N-terminal heptapeptide. Phospholipase A2 has a molecular weight of about 14 kd and is activated by calcium and by bile salts. Phospholipase A2 in the intestinal brush-border membrane and intracellular phospholipases within the enterocyte probably also participate in phospholipid digestion.
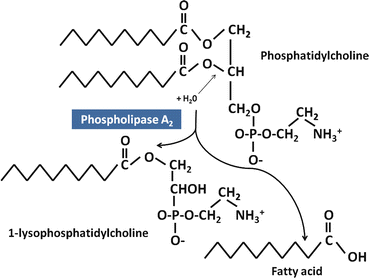

Fig. 8.2
Digestion of phosphatidylcholine by pancreatic phospholipase A2 . Phospholipids are substrates of pancreatic phospholipase A2
3.2 Absorption of Lysophosphatidylcholine
Lysophosphatidylcholine can be incorporated into both mixed micelles and liquid crystalline vesicles. Studies have also demonstrated that bile salts and lysophospholipids form submicellar aggregates than can transfer lysophospholipids between membranes. The relative contributions of these macromolecular structures to intestinal lysophospholipid absorption remain to be determined. It has been traditionally thought that lysophospholipids cross the enterocyte brush-border membrane by passive diffusion , but some data suggest a protein-mediated uptake mechanism.
3.3 Phospholipid Metabolism in the Enterocyte
The absorbed lysophosphatidylcholine can be metabolized by several pathways. Most of the lysophosphatidylcholine is reacylated to form phosphatidylcholine. Alternatively, the lysophosphatidylcholine can be hydrolyzed to fatty acid and 3-phosphorylcholine, or two molecules of lysophosphatidylcholine can react to form one molecule of phosphatidylcholine and one molecule of 3-phosphorylcholine. The fatty acids formed by these reactions can be used for triglyceride synthesis. In addition, the intestine is capable of synthesizing phosphatidylcholine from diglyceride (derived mainly from α-glycerophosphate) via the Kennedy pathway .
In the absence of absorbed lysophosphatidylcholine, there is a marked drop in the secretion of chylomicrons into lymph. The fatty acid composition of the phosphatidylcholine contained on the surface of chylomicrons is not greatly influenced by the dietary fatty acid content, but mainly reflects the fatty acid composition of biliary phosphatidylcholine. These data indicate that de novo phosphatidylcholine synthesis by the enterocyte is inadequate to provide the phospholipid needed for optimal packaging and secretion of chylomicrons, and that absorbed biliary phospholipid is preferentially utilized for chylomicron assembly.
4 Bile Acids
Bile acids are secreted by the liver almost exclusively as conjugated forms, mainly as conjugates with glycine (75 %) or taurine (25 %). A small fraction of these conjugated bile acids is absorbed in the proximal small intestine , mainly the glycine-conjugated dihydroxy bile acids, chenodeoxycholic acid -glycine and deoxycholic acid-glycine. These bile acids are likely protonated in the unstirred water layer of the jejunum, which is estimated to have a pH of approximately 5, and the protonated bile acids are then passively absorbed transcellularly. Glycine-conjugated trihydroxy bile acids and taurine-conjugated bile acids would be fully ionized at the small intestinal pH and therefore would not be absorbed by this mechanism. There is also the possibility of limited paracellular absorption of ionized and nonionized conjugated bile acids in the small bowel.
Most of the conjugated bile acids, however, remain in the lumen as the intestinal contents transit through the upper intestine, where they activate important enzymes and solubilize lipids to ensure efficient absorption. In the ileum , there is a high-capacity active transport mechanism for bile acids that results in the absorption of greater than 95 % of the secreted bile acids. The conserved bile acids are delivered via the portal blood to the liver , where they are taken up for secretion again into bile. This enterohepatic circulation of bile acids, which occurs several times during a meal, ensures an adequate delivery of bile acids into the intestine for highly efficient lipid absorption independent of the rate of hepatic bile acid synthesis from cholesterol.
4.1 Ileal Bile Acid Uptake
The active transport of bile acids across the ileal brush-border membrane is mediated by the transport protein ASBT (gene symbol SLC10A2) (Fig. 8.3). ASBT is a 348 amino acid membrane glycoprotein with a glycosylated extracellular amino terminus, a cytosolic carboxyl terminus, and 7 membrane spanning domains. ASBT is an electrogenic Na + -bile acid co-transporter, with two sodium ions transported per molecule of bile acid. The driving force for transport is the inwardly directed sodium gradient. Conjugated bile acids are transported more efficiency than unconjugated forms, and the affinity for dihydroxy bile acids is higher than for trihydroxy bile acids. ASBT is expressed mainly in the villus cells of ileal enterocytes, with small amounts of ASBT in the proximal intestine and colon. The GATA4 transcription factor is essential for silencing ASBT expression in the proximal intestine.
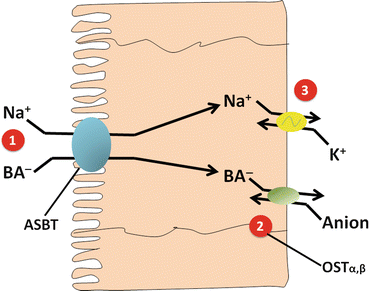

Fig. 8.3
Active transport of bile acids by the ileal enterocyte. (1) Apical Na+-conjugated bile acid carrier ASBT . (2) Basolateral bile-acid-anion exchanger. (3) Basolateral Na+/K+-ATPase (Adapted from Hofmann [7])
ASBT expression is reduced by the bile acid farnesoid X receptor (FXR) that has chenodeoxycholate and other bile acids as ligands. The uptake of bile acids, therefore, can be altered to meet physiological requirements. ASBT is also regulated by the PPARα receptor and by corticosteroids. ASBT and Na+-bile acid co-transport is developmentally regulated in mammals as they are absent at birth and develop in the first postnatal weeks at a variable, species-dependent rate.
Inactivating mutations in ASBT cause bile acid malabsorption . Beginning in early infancy these children have interruption of the enterohepatic circulation of bile acids, chronic diarrhea , and malabsorption of triglycerides and fat-soluble vitamins.
4.2 Intracellular Transport and Export from the Enterocyte
With the enterocyte bile salts associate with a 14 kDa protein called the ileal bile acid binding protein (IBABP) in a 2:1 stoichiometric complex. IBABP is postulated to facilitate transport of bile acids through the cell to the basolateral membrane. IBABP may also play a role in protecting the enterocyte from the toxic effects of high intracellular bile salt concentrations. IBABP expression is increased by FXR and is also developmentally regulated.
The movement of bile acids across the basolateral membrane occurs mainly via the OSTα/β hetereodimeric transporter (Fig. 8.3). OSTα is a 340 amino acid protein with an extracellular amino terminus, 7 transmembrane domains, and an intracellular carboxyl terminus. OSTβ is a 128 amino acid protein with an extracellular amino terminus and a cytoplasmic carboxyl terminus. Both subunits are required for trafficking to the plasma membrane and for bile acid transport. This transport complex appears to function by sodium-independent facilitated diffusion . OSTα/β can transport a number of compounds in addition to bile acids, including estrone-3-sulfate, digoxin, prostaglandin E2, and dehydroepiandstosterone-3-sulfate.
OSTα/β is also increased by FXR. Activation of FXR by bile salts decreases ASBT expression and increases IBABP and OSTα/β expression, thereby preventing intracellular bile acid accumulation.
The multidrug resistance protein 3 (MRP3) plays a minor role in basolateral bile acid export, but may be more important for the small amounts of modified (glucuronidated or sulfated) bile acids present in the intestinal lumen. Multidrug resistance protein 2 (MRP2) may mediate transport of modified bile acids across the brush-border membrane into the intestinal lumen.
4.3 Absorption of Unconjugated Bile Acids
In the intestine, particularly the colon, bacteria modify bile acids in various ways. They deconjugate bile acids by enzymatic hydrolysis of the bond linking bile acids to their amino acid (glycine or taurine ) conjugates. Further bacterial modifications include 7α-dehydroxylation and oxidation/epimerization of hydroxyl groups at various sites. These modifications increase the hydrophobicity and pKA of the bile acids, permitting some passive absorption across the intestinal epithelium. In addition, limited amounts of unconjugated bile acids may be absorbed via the small amounts of transport proteins expressed in the colon.
Bile acids in the distal intestine and colon cause fluid and electrolyte secretion by directly altering enterocyte and colonocyte ion transport and indirectly by stimulating neuroendocrine mechanisms. Excessive colonic bile salts from ileal resection or disease or other processes result in chronic diarrhea .
4.4 Ileal FGF19 Secretion and Regulation of Hepatic Bile Salt Synthesis
Ileal enterocytes synthesize and secrete fibroblast growth factor 19 (FGF19). FGF19 is responsive to FXR. FGF19 is released into the portal circulation, and in an endocrine manner actives fibroblast growth factor receptor 4 (FGF4) in hepatocytes , resulting in inhibition of bile acid synthesis. This pathway works in conjunction with the local intrahepatic regulation of bile acid synthesis by bile salts. A group of patients have been described who have low circulating FGF19 levels and chronic watery diarrhea . It appears that these patients have disordered regulation of hepatic bile acid synthesis and an expanded bile acid pool size that overwhelms a normally functioning ileal absorption mechanism. Excessive colonic bile acids cause fluid and electrolyte secretion and chronic watery diarrhea.
5 Vitamin A
5.1 Definitions
The term vitamin A denotes a family of compounds that are structurally related to all-trans-retinol and are required for vision, growth, cellular differentiation and proliferation, reproduction, and the integrity of the immune system. Naturally occurring compounds such as retinol, retinaldehyde (retinal), and retinoic acid and a large number of synthetic analogues with or without vitamin A biological activity are termed retinoids . Retinoids vary both qualitatively and quantitatively with respect to specific biological actions. For example, retinoic acid in contrast to retinol cannot effectively maintain normal vision or reproductive function.
5.2 Dietary Sources of Vitamin A
Dietary sources of vitamin A include preformed vitamin A, present in animal tissues largely as long-chain fatty acyl retinol esters, and certain carotenoid pigments present in fruits, vegetables, and some animal fats that are precursors of vitamin A. Of more than 500 carotenoids found naturally, only about 50 are precursors of retinol. Al1-trans-β-carotene is the most active on a weight basis and the most important vitamin A precursor for humans. Because the bioavailability of food carotenoids is less than that of retinol, due to poorer intestinal absorption and limited conversion of carotenoids to vitamin A, 12 μg of dietary β-carotene and 24 μg of three other dietary carotenoids are assumed to be nutritionally equivalent to 1 μg of retinol. The major sources of vitamin A or provitamin A in the American diet are liver , carrots, eggs, vegetable-based soups, whole-milk products, and fortified milk and other foods. It is estimated that on average less than one third of total vitamin A activity comes from carotenoids, although there is certainly considerable interindividual variation based on dietary habits and the efficiency of carotenoid absorption and conversion to vitamin A. The roles of β-carotene and other carotenoids in the prevention of cancers, heart disease, and other chronic degenerative diseases is a topic of intense current interest. Many of the physiological effects of carotenoids are not due to their function as vitamin A precursors, but reflect antioxidant and other properties of the carotenoids themselves.
5.3 Digestion of Dietary Retinyl Esters
As with other esterified lipids, such as cholesteryl esters, retinyl esters must be hydrolyzed before intestinal absorption can occur (Fig. 8.4). Several pancreatic enzymes, including pancreatic lipase , pancreatic lipase-related protein 2, and pancreatic bile salt-activated lipase can hydrolyze retinyl esters, with pancreatic lipase appearing to be most important. Brush-border membrane phospholipase B (PLB) also participates in retinyl ester hydrolysis. The relative contributions of pancreatic and small intestinal enzymes to retinyl ester hydrolysis are currently uncertain.
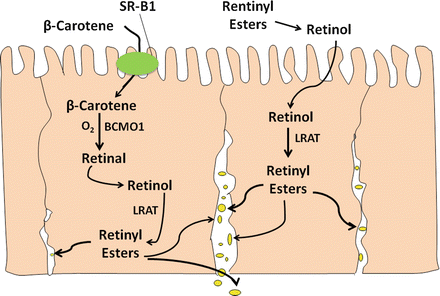
5.4 Uptake of Retinol and β-Carotene by the Enterocyte
Both retinol (formed by hydrolysis of retinyl esters) and β-carotene are solubilized in mixed micelles within the luminal contents. At low concentrations, retinol is taken up by a saturable, energy-independent process that is consistent with carrier-mediated facilitated diffusion (Fig. 8.4). A specific carrier has not yet been identified. At higher pharmacological retinol concentrations, uptake is not saturable and likely occurs via simple, passive diffusion . Retinol uptake is greater in the jejunum than ileum and is greater in neonatal animals than adults, suggesting developmental regulation of a carrier. Uptake of β-carotene and other carotenoids across the brush-border membrane is mediated by the scavenger receptor class B, type 1 (SR-B1) (Fig. 8.4).
5.5 Carotene Cleavage and Retinal Reduction
Within the enterocyte, carotenes are cleaved, forming retinal. Two enzymes, β-carotene -15,15′-monooxygenase (BCM01) and β-carotene-9′,10′-monooxygenase (BCM02) can cleave carotenoids (Fig. 8.5). BCM01 cleaves carotene centrally forming two molecules of retinal, whereas BCM02 cleaves asymmetrically forming apocarotenals that are subsequently shortened to produce retinal. Some intact β-carotene is absorbed and circulates in plasma. Liver and other tissues have the capacity to form vitamin A from carotene via BCM01. The retinal produced from carotenes is efficiently reduced to retinol in the enterocyte (Fig. 8.4). Both soluble and microsomal reductases have been identified. Within the enterocyte, most of the retinal is probably bound to a specific binding protein, cellular retinol-binding protein, type 2 or CRBP(II) (see below). The microsomal reductase effectively utilizes retinal bound to CRBP(II) as a substrate, whereas the soluble reductase appears to prefer free retinal.
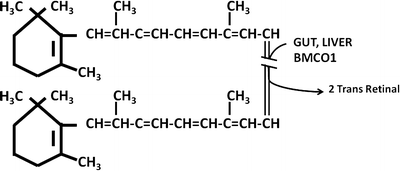

Fig. 8.5
Conversion of β-carotene to vitamin A
Intestinal BCM01 mRNA expression is increased when animals are fed a retinoid-deficient diet. Polymorphisms have been identified in the human BCM01 gene that decrease the conversion of β-carotene to vitamin A, and may explain some of the inter-individual variability in the ability to absorb and convert proretinoid carotenoids to retinoids .
5.6 Cellular Retinol-Binding Protein, Type 2
Several retinoid-binding proteins play crucial roles in the transport, metabolism, and cellular actions of vitamin A. In the enterocyte, one of these proteins, CRBP(II), is key in the regulation of vitamin A absorption and metabolism. CRBP(II) is a 133-amino-acid protein with one retinoid-binding site that constitutes approximately 1 % of the soluble protein in the jejunum. CRBP(II) belongs to a large superfamily of lipid-binding proteins that includes the fatty acid-binding proteins and another retinoid-binding protein, CRBP(I). CRBP(I) is found at low levels in many tissues including the small intestine , but it is not thought to play a role in vitamin A absorption. In adults CRBP(II) is expressed only in the small intestinal villus epithelial cells. CRBP(II) mRNA and protein are greater in the jejunum than ileum and increase during pregnancy and lactation. In addition to its role in the reduction of retinal derived from carotenes, CRBP(II) also appears to regulate the esterification of retinol within the enterocyte.
5.7 Retinol Esterification
Both absorbed retinol and retinol generated from carotenes are esterified in the enterocyte with long-chain fatty acids. Palmitate , stearate , oleate , and linoleate account for most of the esterified fatty acids in an approximate ratio of 8:4:2:1, and this pattern is not significantly altered by changes in dietary fatty acid composition. Two microsomal enzymes capable of esterifying retinol have been identified. Lecithin-retinol acyltransferase (LRAT) effectively esterifies CRBP(II)-bound retinol (Fig. 8.4). This enzyme uses phosphatidylcholine as the fatty acid donor, and the fatty acid composition of the product retinyl esters is similar to that of position 1 of intestinal lymph phosphatidylcholine. LRAT has a low Km for CRBP(II)bound retinol that would be appropriate for activity at a low dietary vitamin A intake, and LRAT accounts for about 90 % of the esterification of physiological amounts of retinol. LRAT is greater in the proximal than the distal small bowel and is developmentally regulated in a pattern similar to CRBP(II). Levels of these proteins increase prior to birth, remain high during suckling, and then decline with weaning to the adult level. Diacylglycerol acyltransferase 1 (DGAT1), a key enzyme in triglyceride absorption, also can esterify retinol, and plays an important role at high levels of retinol intake.
5.8 Export of Vitamin A and Carotenoids from the Enterocyte
Retinyl esters synthesized in the enterocyte and carotenoids that escape cleavage are incorporated into the lipid core of chylomicrons and released into the intestinal lymph. Retinyl esters and carotenoids appear to be inserted into chylomicrons by the MMTP at a late state of chylomicron production. Some retinol is released from the enterocyte into portal blood, perhaps mediated by the ABCA1 transporter in the basolateral membrane.
5.9 Hepatic Metabolism and Storage of Vitamin A
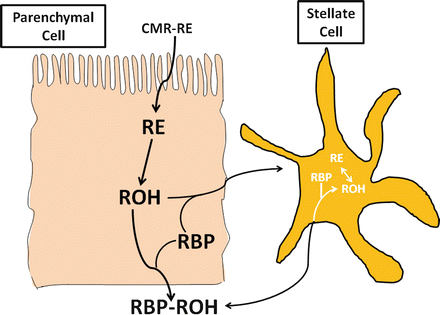
Most (65–75 %) of absorbed vitamin A stays associated with the lipid core of chylomicrons as the triglyceride in these lipoproteins is hydrolyzed, forming chylomicron remnants . Extrahepatic uptake of retinyl esters from chylomicrons and chylomicron remnants in certain tissues is important in supplying vitamin A to these cells. Chylomicron remnants are taken up by hepatocytes using several receptor-mediated mechanisms. Once remnant-associated retinyl esters have been taken up by hepatocytes, rapid hydrolysis occurs; multiple hepatic enzymes can hydrolyze retinyl esters (Fig. 8.6). Retinol is transferred from hepatocytes to peri-sinusoidal hepatic stellate cells. Cellular retinol binding protein 1 [CRBP1] is important in the transfer of retinol to hepatic stellate cells and its subsequent esterification. Some, but not all data, suggests the involvement of the 21 kDa serum retinol binding protein (RBP) in this process. CRBP(I)-bound retinol is esterified by LRAT in the hepatic stellate cells. Normally, stellate cells contain about 90 % of liver vitamin A, and 98 % is in the form of retinyl esters. Retinyl esters are stored within the hepatic stellate cells along with other lipids in the form of lipid droplets.
 < div class='tao-gold-member'>
< div class='tao-gold-member'>





Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree