SRH: Stigmata of recent hemorrhage.
A number of other risk scoring systems have been developed of which the best known is that developed by Blatchford et al. [9]. The Blatchford system differs from the other scores in two ways. First, the system was designed to predict a need for intervention to treat bleeding, rather than to quantify the risk of re-bleeding or death. Second, the score does not include endoscopic findings as a component and is therefore potentially useful in patient triage at the time of admission. Data were obtained from 1748 patients admitted following upper gastrointestinal bleed-ing in the west of Scotland. After logistic regression analysis a risk score was developed based on the admission hemoglobin, blood urea, pulse, systolic blood pressure, presentation with syncope, presentation with melena, evidence of hepatic disease and evidence of cardiac failure. Intervention was defined as the requirement for a blood transfusion, endoscopic therapy or surgery to control bleeding. Increasing scores correlated with the need for intervention. A receiver operator curve was plotted for a subsequent internal validation sample of 197 patients, and the score discriminated well with an area under the curve of 0.92 (95% CI: 0.88–0.95). The number of patients requiring each intervention was not reported and this limits an assessment of its utility and clinical relevance. External validation of this scoring system is required before it can be recommended for clinical use.
Optimum management of patients with major upper GI hemorrhage relies very much on a team approach, with appropriate use of drug therapy, endoscopic intervention, radiological intervention and surgery. Despite much evidence from randomized trials, the management of an individual patient still depends on clinical judgment. Endoscopy should only be undertaken once appropriate resuscitation has been achieved in order to minimize cardiorespiratory complications of the procedure [10]. For the majority of patients with peptic ulcer bleeding an endoscopy within 24 hours of presentation is appropriate, and there is no evidence to support emergency endoscopy for all comers. However, in the small proportion of cases with severe, life-threatening bleeding resulting in a failure to respond to resuscitation efforts, an immediate endoscopy may be required.
Management may be best undertaken in a specialized “bleeding unit” in which the patient is treated using agreed protocols and guidelines and with management decisions based upon endoscopic and surgical opinions. Relatively weak evidence derived from comparison of results in case series with historical controls suggests that this approach may achieve lower hospital mortality and more efficient use of resources than management by generalists working in conventional medical or surgical units [11, 12]. More recently a study by Sanders et al. described the outcomes of 900 patients with significant bleeding who were managed over a three-year period in a dedicated bleeding unit [13]. Rockall scores were calculated for all patients and outcomes were analyzed prospectively. Patients were stratified into groups at low, medium and high risk of death according to their Rockall score and outcomes were compared with those of the patients in Rockall’s national audit [3] by calculation of standardized mortality ratios (SMR). Although there were more high-risk patients in the Sanders cohort, their overall mortality was significantly lower than that of the Rockall patients (SMR 0.63 (95% CI: 0.48–0.78)). The difference was most marked for patients at medium risk (Rockall score 4–6; SMR 0.56 (CI: 0.34–0.78)). However, it is worthy of note that if the SMRs had been calculated using the findings of the UK National Bleeding audit [1] the findings of this study may well have been negative. A trial in which patients with major bleeding have been randomized to management in either a specialized bleeding unit or a general medical or surgical ward has not been performed.
Specific therapy
For the 80% of patients who have relatively minor bleeding and who do not have major endoscopic stigmata of bleeding, supportive therapy including use of intravenous fluid and the management of co-morbidity (particularly cardi-orespiratory disease) is sufficient.
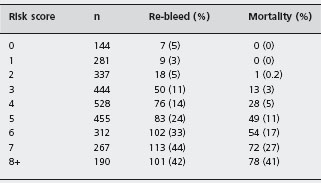
Patients who present with clinical shock and who at endoscopy have an actively bleeding peptic ulcer have an 80% risk of continuing to bleed or re-bleed in hospital [14]. Those who have a non-bleeding visible vessel have a 50% risk of further hemorrhage [15]. The “visible vessel” represents a pseudoaneurysm of the involved artery, or adherent blood clot, plugging the arterial defect [16]. Patients who are found to have a tightly adherent blood clot over the ulcer usually have an underlying high-risk lesion and should also be regarded as being at considerable risk of further hemorrhage in hospital. Patients who at endoscopy have a clean ulcer base or who have black or red spots are at very little risk of re-bleeding.
It follows from these observations that patients with major endoscopic stigmata should be considered for spe-cific hemostatic treatment and only such patients should be included in clinical trials of therapy for gastrointestinal bleeding. This review will only consider those studies that exclusively include patients having a non-bleeding visible vessel, active hemorrhage, or tightly adherent blood clot as entry criteria.
The specific non-surgical approaches to hemostasis are drug therapy, endoscopic therapy and transcatheter arterial embolization.
Drug therapy
There are three principles underlying the use of drugs as agents which might stop active hemorrhage and prevent re-bleeding. The first of these is that the stability of a blood clot is poor in an acid environment [17]. Thus, agents that suppress acid secretion, including H2-receptor antagonists (H2-RA) and proton pump inhibitor (PPI) drugs might reduce re-bleeding. The second is that a blood clot may be stabilized by decreasing fibrinolytic mechanisms using agents such as tranexamic acid. The third approach is that, since major gastrointestinal bleeding is due to arterial erosion, reduction of arterial blood flow by agents such as somatostatin and octreotide could achieve hemostasis and prevent re-bleeding.
Acid suppressing drugs
The efficacy of H2-RA in the management of acute upper gastrointestinal bleeding has been assessed in randomized trials [18, 19]. Unfortunately, no trial has shown benefit in terms of reduction of re-bleeding incidence or mortality. A1c
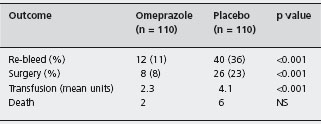
Experience involving the use of PPIs is inconsistent, but the evidence now supports their use in patients who have required endoscopic hemostasis. The largest trial involved 1147 patients who were randomized to receive omepra-zole, (initially intravenously, then orally) or placebo [20]. No significant differences in hospital mortality, operation rate or re-bleeding were demonstrated. The study was not restricted to the high-risk patients who had endoscopic stigmata of recent hemorrhage. Accordingly, event rates were rather low in the placebo group, and this may have limited the power of the study to show a difference. A1a
Khuroo et al. randomized 220 bleeding ulcer patients who had major endoscopic stigmata to receive high dose oral omeprazole or placebo [21]. Although all patients had major stigmata of hemorrhage, an adherent clot in the ulcer base was reported in 57% of patients. Re-bleeding, the need for urgent surgery, blood transfusion, and mortality were all reduced in the actively treated group of patients (Table 8.3). The number of patients needed to treat with omeprazole to prevent one death was 25, and to prevent one opera-tion was seven. A1c This trial has been criticized because it included relatively young patients with relatively little co-morbidity and because endoscopic therapy was not administered to any patient. The observation that omeprazole reduced re-bleeding and surgery rates when no endoscopic therapy was performed suggested a beneficial effect of the PPI. This effect might, however, have been exaggerated by the fact that the majority of patients in the trial were bleeding from ulcers in which an adherent clot was found at endoscopy.
Two trials published back to back in the Scandinavian Journal of Gastroenterology [22, 23] examined the use of high dose intravenous omeprazole after endoscopic hemostasis. All patients had major peptic ulcer bleeding, but as in the trial by Khuroo et al. half the patients had adherent clot as the reported stigma of hemorrhage. The conclusions were that intravenous omeprazole infusion for three days following endoscopic therapy improved outcome. A1d Both trials used composite endpoints which were complex and ill-defined and both were discontinued early due to an unexplained imbalance in mortality in one of the trials [23], factors that weaken the impact of these results. Villanueva et al. randomized 86 patients following successful endoscopic hemostasis for peptic ulcer bleeding to either intravenous omeprazole or ranitidine. There were no differences between the groups for the endpoints of re-bleeding, surgery or death [24]. A1d In contrast, a similar small trial by Lin and colleagues [25] concluded that intravenous omeprazole was superior to cimetidine in terms of reduction of re-bleeding rates, but not those of surgery or death.
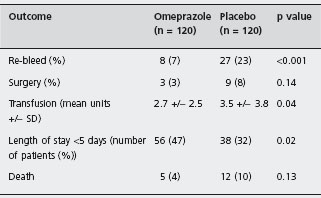
The most important recent trial was performed by Lau et al. [26]. Two hundred and forty patients in whom endoscopic therapy for major ulcer bleeding had been successful were randomized. All patients had high-risk ulcers with active bleeding or non-bleeding visible vessels, and were treated by adrenaline injection followed by heater probe thermocoagulation. Adherent clots were removed to allow therapy to the underlying vessel. The patients then received either an 80 mg bolus dose of intravenous omeprazole followed by an infusion of 8mg per hour for 72 hours or placebo. Re-bleeding rates, blood transfusion requirements and length of hospital stay were significantly reduced in the omeprazole group compared to placebo. A1a There was a trend toward fewer operations and deaths in the omeprazole group, but these differences were not statistically significant (Table 8.4). These results have been closely replicated in a similar sized trial by Zargar et al. in which an almost identical design to that of Lau et al. was used, except that pantoprazole was substituted for omeprazole [27]. This supports the likelihood that the observed outcomes of therapy with PPIs following endoscopic hemostasis are the result of a class effect of PPIs, rather than a specific response to omeprazole.
A subsequent trial by the Hong Kong group included 156 patients with peptic ulcers containing non-bleeding visible vessels or adherent clot in the ulcer base [28]. Patients were randomized to endoscopic therapy using adrenaline injection and heater probe thermocoagulation plus the previously published high dose intravenous PPI regimen, or to the PPI regimen alone. The probability of re-bleeding within 30 days of the index episode was significantly reduced in the combination therapy group, suggesting that PPI infusion in combination with endoscopic therapy is superior to PPI infusion alone. A1c Seventeen percent of patients with non-bleeding visible vessels re-bled in the PPI only group, and although a control group receiving no treatment was not included for ethical reasons, this represents a substantial improvement over the expected re-bleeding rate of 30–50% based on previous studies.
Following the publication of the trial by Lau et al. [26] the use of high dose intravenous PPI after successful endoscopic therapy for bleeding ulcer has become standard management in many centers in the UK and Europe. The 80 mg bolus and 8mg per hour infusion regimen consistently raises intragastric pH above six for the majority of a 24-hour period [29]. It is not known, however, whether this optimum regimen is actually necessary following endo-scopic therapy, and whether bolus intravenous or even oral PPI would suffice. A recent study has suggested that frequent oral PPI therapy with lansoprazole could raise intra-gastric pH to similar levels to those achieved by lansoprazole infusion. However, the confidence intervals were wide and the conclusion can be questioned [30]. Two small clinical studies have attempted to answer these questions.
Udd et al. [31] randomized 142 patients with ulcer bleeding to the high dose three-day intravenous omeprazole regimen or to a single daily bolus dose of 20 mg for three days. Rates of re-bleeding (8% for high dose vs 12% for standard dose), surgery (4% vs 7%) and death (6% vs 3%) were comparable between the groups. Only 102 patients had required endoscopic therapy, and approximately 30% of patients had an ulcer with a black base only. Thus, the number of high-risk ulcers in the trial was small, the event rates were low and the study may have lacked power to demonstrate a difference between the effects of the two treatments.
The effect of oral omeprazole following endoscopic therapy for bleeding peptic ulcer was studied by Javid et al. [32]. One hundred and sixty six patients with ulcers which were actively bleeding, and had non-bleeding visible vessels or adherent clots were treated with a combination of 1:10,000 adrenaline plus 1% polidocanol injection. They were then randomized to receive oral omeprazole 40 mg bd or placebo. Six (7%) of the 82 patients in the omeprazole group re-bled compared with 18 (21%) of the 84 patients in the placebo group (p = 0.02). A1c Surgery was required in two patients in the omeprazole group and seven patients in the placebo group (p = 0.17). One death occurred in the omeprazole group compared to two in the placebo group. The results are comparable to those achieved by Lau et al. with the high dose intravenous regimen [26]. It should be noted, however, that 40% of patients in this trial had ulcers with adherent clot, and the number of patients with high-risk lesions was therefore correspondingly lower than that in the Hong Kong study. A further high quality trial comparing the use of intravenous and oral omeprazole in high-risk ulcer bleeding patients is required.
The trials involving PPI therapy in ulcer bleeding are heterogeneous but have been subjected to several meta-analyses. The most comprehensive is the recently updated report from the Cochrane group [33]. This systematic review of 24 trials included 4373 patients with peptic ulcer bleeding who had been treated with PPIs compared with H2-receptor antagonists or placebo. Seven trials included high-risk patients following endoscopic therapy, and four of these used high dose PPI infusion in the active therapy arm. Overall results demonstrated a reduction in re-bleeding (OR 0.49; 95% CI: 0.37–0.65, NNT 13), surgery (OR 0.61; 95% CI: 0.48–0.78, NNT 34) and requirement for further endoscopic hemostatic therapy (OR 0.32; 95% CI: 0.20–0.51, NNT 10). Mortality was not reduced (OR 1.01; 95% CI: 0.74-1.40). However, in a subgroup analysis, mortality in PPI groups was significantly reduced in Asian patients (OR 0.35; 95% CI: 0.16–0.74, NNT 34) and in the high-risk patients entered into the endoscopic therapy trials (OR 0.53; 95% CI: 0.31–0.91, NNT 50).
The more pronounced PPI treatment effect observed in Asian patients is likely to be the result of a combination of lower mean age, reduced parietal cell mass and a greater proportion of slow metabolisers of PPIs. There is a need to further evaluate the use of PPI therapy in non-Asian populations, but in our view the available evidence supports the routine use high dose intravenous PPI therapy for 72 hours following endoscopy in those patients who have required hemostatic therapy. A1a
As PPI therapy has become more widely used in acute peptic ulcer bleeding it has become common practice to institute intravenous PPI therapy before endoscopy is undertaken. This approach is not evidence based and to try to determine whether PPI prior to endoscopy was beneficial, Lau et al. randomized 638 patients with overt clinical signs of upper GI bleeding to high dose IV PPI or to placebo infusion before endoscopy was performed [34]. Patients who were taking aspirin were excluded as they were randomized into a concurrent study. One hundred and eighty seven patients in the omeprazole arm and 190 patients in the placebo arm were found to be bleeding from peptic ulcers. Fewer patients pre-treated with omeprazole were actively bleeding at endoscopy, resulting in a reduced need for endoscopic therapy (22.5% vs 36.8%, RR 0.61; 95% CI: 0.44–0.84) and a shorter hospital stay (median 3 days (range 1–43) vs 3 days (1–54), p = 0.003). Despite these findings, the important outcomes of re-bleeding, surgery and mortality were unchanged. The conclusion was that PPI therapy prior to endoscopy reduced the proportion of patients found to have stigmata of hemorrhage, with consequent resource savings. These results are encouraging, but do not show a benefit in terms of clinical outcomes and may not be applicable generally. The principal criticism of this trial is the exclusion of patients requiring long-term aspirin therapy. In addition, 60% of patients presenting with upper GI bleeding in Hong Kong are found to have peptic ulcer compared to 50% or lower in Western populations, in whom the effect of general prescription of PPIs before endoscopy is likely to be significantly less apparent. Further studies in non-Asian populations are required.
Tranexamic acid
Two meta-analyses have been published examining the role of tranexamic acid for gastrointestinal bleeding [35, 36]. The most recent by Gluud et al. [36] included 1754 patients from seven placebo controlled trials. Pooled analysis demonstrated a reduction in all cause mortality for patients treated with tranexamic acid (5% vs 8%, RR 0.61; 95% CI: 0.42–0.89). Rates of further bleeding, surgery and blood transfusion were unaffected.
The largest study included in the meta-analysis was undertaken by the Nottingham group [37]. Seven hundred and seventy-five patients presenting to hospital because of acute gastrointestinal bleeding were randomized to receive oral cimetidine, tranexamic acid or placebo. No significant difference in bleeding or operation rates was demonstrated, but there was a rather surprising large difference in mortality. Mortality was 7.7% in cimetidine treated patients, 6.3% in tranexamic acid treated patients and 13.5% in those treated with placebo. The mortality rate of 13.5% in the placebo treated group is greater than that expected for conservatively treated patients based on the results of other studies. Furthermore, other studies do not demonstrate benefit from the use of cimetidine. It is possible that more high-risk patients were inadvertently randomized to the placebo group in this study.
The meta-analyses included trials in which many patients did not have major endoscopic stigmata of bleeding. In addition, there were significant differences in methodology, doses and routes of administration of tranexamic acid and adjunctive therapy, and few patients received endoscopic therapy and PPI administration. Therefore, it is unlikely that the patients studied can be seen as representative of the current population, and further randomized trials in high-risk patients are required before tranexamic acid can be recommended as a standard therapy for peptic ulcer bleeding.
Somatostatin and octreotide
Somatostatin and its analogs have two actions which are theoretically valuable in the management of ulcer bleeding, namely inhibition of acid secretion and reduction of splanchnic blood flow. Mesenteric blood flow falls dramatically during infusions of somatostatin but it is not clear whether this is principally due to vasoconstriction of major blood vessels or peripheral arterioles.
There have been 14 controlled trials of somatostatin versus other therapy in the management of patients presenting with acute gastrointestinal bleeding [38–51]. Two meta-analyses suggest that somatostatin but not octreotide has a primary hemostatic role and reduces the need for surgical intervention [52, 53]. However, scrutiny of the relevant trials reveals many problems. Many of the studies were small and inclusion criteria varied widely from gastritis to major active bleeding. In common with the studies of tranexamic acid, the somatostatin trials were conducted before the widespread use of endoscopic therapy and PPI administration.
Currently, the evidence for routine use of somatostatin is weak and further studies are needed before this agent can be advocated as therapy for acute non-variceal gastrointestinal bleeding.
Endoscopic therapy
Many therapeutic endoscopic treatments have been used to try to stop active ulcer bleeding and prevent re-bleeding. These can be classified into three basic endoscopic approaches (Table 8.5).
Thermal approaches involving laser, the heater probe and electrocoagulation by monopolar or bipolar probes attempt to induce thermocoagulation with thrombosis of the bleeding point. In experimental bleeding ulcers these approaches are more effective than injection treatments [54]. However, there is currently no good model of acute peptic ulcer bleeding. Experiments in animals were historically based upon observation or intervention following superficial mucosal injury, which is different from erosion of arteries by chronic or acute peptic ulceration. A novel pig model has recently been developed, which appears to resemble the real life situation of an actively bleeding peptic ulcer more closely, although the element of chronic inflammation cannot be reproduced [55]. This model could facilitate the development of new endoscopic therapeutic methods. Injection therapy may produce tamponade by the injection of a relatively large volume of fluid into a rigid compartment, compressing the bleeding artery. Vasoconstriction induced by dilute adrenaline, endarteritis induced by sclerosants, dehydration following absolute alcohol injection or a direct effect upon blood clot formation following injection of thrombin or fibrin glue are other putative mechanisms. Mechanical clips, staples and sewing attempt to produce hemostasis by clamping the bleeding arterial lesion. Many clinical trials of endoscopic therapy for non-variceal bleeding have been published. The quality of these trials varies greatly. In general, the number of patients randomized in any one study is small and clinicians managing the patients have not been blinded to the type of endoscopic therapy. Only one trial has included a placebo control intervention for endoscopic therapy [56].
Table 8.5 Endoscopic therapy.
| Modality | Type |
| Thermal | Argon laser |
| Nd-YAG laser | |
| Heater probe | |
| Electrocoagulation | |
| Argon plasma coagulation | |
| Injection | Adrenaline |
| Sclerosants | |
| Alcohol | |
| Thrombin | |
| Fibrin glue | |
| Mechanical | Hemoclips |
| Staples | |
| Sutures |
Thermal methods
Laser photocoagulation
Lasers were the first endoscopic therapeutic modality shown to be effective in managing acute non-variceal gastrointestinal bleeding. Initial experience involved the use of argon lasers but it subsequently became clear that the tissue characteristics of thermal injury achieved by Nd-YAG were more appropriate. There have been a number of trials involving lasers for peptic ulcer bleeding [57–68]. Most of these studies show that laser treatment significantly reduced the rates of re-bleeding, transfusion requirement and operation rate. One trial showed significant improvement in hospital mortality [60].
Endoscopic laser therapy was found to be relatively safe with few complications; in particular, gastrointestinal perforation was rare. However, since the technique is difficult, relatively expensive and because other approaches are at least as effective, laser therapy for peptic ulcer bleeding is no longer used.
Heater probe
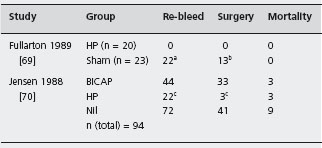
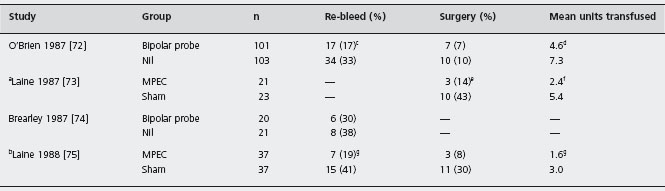
The heater probe transmits preset amounts of energy to the bleeding point via a Teflon tipped catheter. A powerful water jet is used to clean the ulcer base, help visualize the bleeding point and also to prevent the probe sticking to the bleeding point. Hemostasis is achieved by coaptive coagulation, using both tamponade and the application of heat. Best results are achieved using large sized probes. There have been two trials in which the heater probe has been compared to conservative therapy [69, 70]. Both showed benefit in terms of further bleeding, and surgery, and one published only in abstract form [71] demonstrated a trend towards reduction in mortality (Table 8.6). A1d
The heater probe is “user friendly”. Its capacity to apply thermal energy by tangential application and its powerful water jet are particular advantages. Perforations have occurred following treatment, although these are unusual, and are of the order of 1% [71]. In general, medium power settings (20–30 joules) are used, but it is not possible to be prescriptive concerning the total amount of energy that should be applied. Most authorities consider that treatment should be continued until active hemorrhage is stopped and until the treated area is blackened and cavitated.
ap = 0.05.
bp = 0.23.
cp < 0.05.
HP: heater probe; BICAP: bipolar electrocoagulation.
Electrocoagulation
Monopolar electrocoagulation uses a metal ball-tipped probe. An electrical circuit is completed by a plate attached to the patient. Application of energy is rather haphazard and perforations and a death were reported in early series. Consequently this device is no longer used. Multipolar electrocoagulation is based upon transmission of electrical energy between adjacent electrodes. The BICAP has eight separate electrodes over its surface. Early studies from the UK involving small numbers of patients showed no benefit for active treatment compared to conservative therapy. Subsequently, however, trials from the UK and the USA showed improved outcomes including primary hemostasis, re-bleeding, the need for surgery and transfusion requirements with bipolar electrocoagulation compared to conventionally treated patients (Table 8.7) [72–75]. The efficacy of the heater probe and BICAP appear to be comparable with similar low complication rates [71, 76]. A1d
Argon plasma coagulation
This procedure is based upon coagulation through a jet of argon gas. Relatively superficial thermal damage is achieved. The method is particularly applicable to mucosal and superficial bleeding lesions and its final role may be in dealing with vascular malformations such as gastric antral vascular ectasia. One small trial has shown that argon plasma coagulation is comparable in efficacy to heater probe therapy for ulcer hemostasis [77]. A second trial compared the argon plasma coagulator with combination injection of adrenaline and polidocanol [78]. Again the two approaches were equally effective. Nevertheless the tissue damage characteristics of argon plasma coagulation are less than ideal for managing arterial bleeding, and it will probably prove to be less appropriate for managing peptic ulcer bleeding than contact methods.
aStudy included ulcers, Mallory – Weiss tears and vascular malformations.
bStudy was restricted to ulcers with non – bleeding visible vessels. See also Jensen [70] , Table 8.6 .
cp = 0.01.
dp = 0.13.
ep = 0.049.
fp = 0.002.
gp < 0.05.
MPEC: multipolar electrocoagulation.
Conclusions
Thermal methods of hemostasis were shown to be superior to conservative management in two meta-analyses. In the study of Cook et al. [79] the odds ratio for prevention of re-bleeding was 0.48 (95% CI: 0.32–0.76); and for avoidance of surgery was 0.47 (95% CI: 0.27–0.80). Similarly, in the study of Henry and White [80], the odds ratio for prevention of bleeding was 0.32 (95% CI: 0.22–0.41) and for the avoidance of surgery was 0.31 (95% CI: 0.19–0.43). A1d Thermal contact methods (heater probe and bipolar coagulation) are technically easier to undertake than laser techniques. There are insufficient data to determine whether the heater probe is better than the BICAP.
The safety profile of thermal modalities is generally very good. Perforations are unusual and treatment induced exacerbation of bleeding is not usually clinically important.
Injection therapy
Injection treatment is simple to perform and is the cheapest available hemostatic modality. A large range of injection materials have been studied and it is difficult to prove that any one of these is superior to the others.
Dilute adrenaline
In 1988 Chung et al. reported a controlled trial in which patients with active ulcer bleeding were randomized to receive endoscopic injection with 1:10,000 adrenaline or were treated conservatively [81]. Primary hemostasis was achieved in all injected patients and the need for subsequent urgent surgery was significantly reduced (Table 8.8). A1d Re-bleeding occurred in 24% of injected patients, suggesting that although dilute adrenaline did stop active bleeding, its effects were temporary.
Table 8.8 Adrenaline for gastrointestinal bleeding
Source: Chung et al. [81]
| Outcome | Adrenaline n = 34 | Conservative n = 34 |
| Primary hemostasis (%) | 34 (100) | —a |
| Surgery (%) | 5(15) | 14 (41) |
| Mortality (%) | 3(9) E | 2(6) |
a Twenty patients stopped bleeding spontaneously.
It seemed logical to combine an injection of adrenaline with that of an agent which might cause permanent sealing of the bleeding arterial defect. For this reason a series of trials were undertaken in which adrenaline injection was combined with a range of sclerosants.
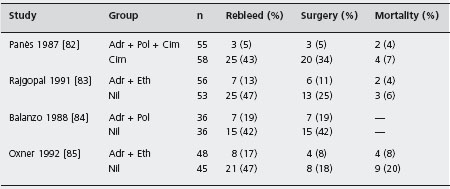
The results of trials in which a combination of adrenaline plus sclerosants were compared to conservative therapy are summarized in Table 8.9 [82–85]. All showed that active bleeding stopped more rapidly in treated patients, that re-bleeding rates were less, and that the need for surgery was reduced. No single trial, however, was powerful enough to determine whether mortality was affected. A subsequent meta-analysis, involving thermal contact devices, laser and injection therapy performed by Cook et al. [79] did show a modest reduction in mortality, although this was statistically significant only for laser therapy. A1a
Adr: adrenaline; Pol: polidocanol; Eth: ethanolamine; Cim: cimetidine.
Sclerosants
The sclerosants that have been studied are polidocanol, 5% ethanolamine oleate and 3% sodium tetradecyl sulphate. There are no controlled trials in which outcome has been assessed in patients randomized to sclerosants versus conservative (no injection) therapy. Several trials compared the efficacy of sclerosants with other endoscopic therapies. Benedetti et al. [86] showed similar efficacy for polidocanol and thrombin injection in patients presenting with a range of bleeding lesions. Strohm et al. [87] randomized patients to one of four treatment arms (fibrin glue, 1% polidocanol, dilute adrenaline or adrenaline plus polidocanol) and showed no advantage for any one approach. Rutgeerts et al. showed no difference in outcome for patients treated by polidocanol or Nd-YAG laser therapy [88]. In general these studies suffer from the problem of small sample size and they probably lacked statistical power.
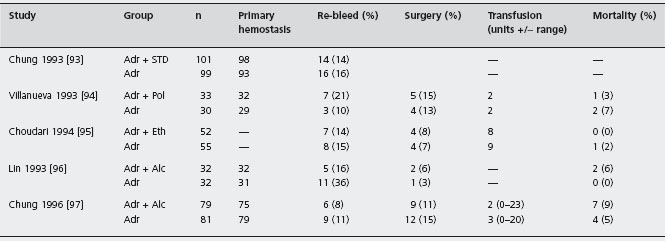
A series of case reports documented complications of injection by sclerosant [89,90], particularly perforation and necrosis of the upper gastrointestinal tract. These complications did not occur following adrenaline injection and indeed the latter seems remarkably safe. Fears concerning the possible systemic affects of circulating adrenaline have not translated into cardiovascular mishaps [91]. Since complications are mainly due to sclerosant injection, it was important to confirm the importance of combining the sclerosant with the adrenaline injection. Whilst the logic of attempting to induce endarteritis using sclerosants was reasonable, experiments in animals did not demonstrate that this could be achieved by injection using ethanolamine or absolute alcohol [92]. C Three trials have compared the efficacy of injection by adrenaline alone versus a combination of adrenaline plus a sclerosant [93–95], and a further two have compared adrenaline to adrenaline plus alcohol [96, 97]. As shown in Table 8.10, these five studies did not show that combination treatment was superior to injection by adrenaline alone. A1c No study has directly compared outcome in patients randomized to dilute adrenaline or to a sclerosant.
Since the addition of sclerosants to an injection of adrenaline offers no proven advantage over injecting adrenaline alone, and because sclerosants have the potential to cause significant local complications following injection, they should no longer be employed as part of the injection treatment regimen.
Alcohol
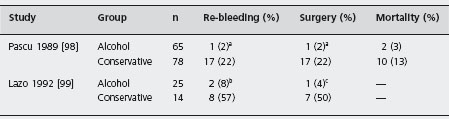
The efficacy of injecting absolute alcohol into bleeding ulcers has been examined in several clinical trials. Two of these (Table 8.11) [98, 99] randomized patients to alcohol injection or to conservative therapy and showed benefit in terms of reduction in re-bleeding rates and need for surgical intervention. A1d
In a randomized controlled trial, Lin et al. [100] reported that alcohol injection stopped active bleeding and prevented re-bleeding in 86% of patients whose ulcers were injected, and this result was similar to the proportion of bleeding ulcers responding to injection with 3% sodium chloride, 50% dextrose, or normal saline. Only one small study [101 ] has attempted to compare the efficacy of alcohol with dilute adrenaline injection, but this study lacked statistical power to demonstrate differences in the effects of these interventions, should any exist.
Adr: adrenaline; STD: sodium tetradecyl sulphate; Alc: alcohol; Eth: ethanolamine; Pol: polidocanol.
a p = 0.0007.
b p < 0.001.
c p < 0.05.
The evidence that alcohol stops active bleeding and prevents re-bleeding is stronger than that for the sclerosants. Unfortunately, the potential for adverse effects is higher for alcohol than for adrenaline. Deep ulcers commonly follow alcohol injection and perforations have occurred [102]. Two trials have examined the need to combine alcohol injection with adrenaline and found no beneficial effect over the injection of adrenaline alone (Table 8.10) [96, 97].
Whilst alcohol injection is an effective hemostatic therapy, current evidence suggests that the magnitude of its effect is probably similar to that achieved by injection with adrenaline alone. Because of its propensity for causing adverse effects, alcohol injection is not recommended as treatment for ulcer bleeding.
Thrombin and fibrin glue
The most attractive endoscopic approach is to directly cause blood clot formation by injecting thrombogenic substances. In the 1980s small trials examined the efficacy of bovine thrombin and showed little benefit compared to other modalities.
In 1996 Kubba et al. [103] reported a comparison of endoscopic injection therapy using a combination of adrenaline plus human thrombin with dilute adrenaline injection alone (Table 8.12). A proportion of randomized patients had active bleeding at the time of randomization, while the remainder had non-bleeding visible vessels. Re-bleeding and mortality were significantly reduced in the group receiving combination therapy compared to patients receiving adrenaline alone. The number of patients needed to be treated with combination therapy rather than adrenaline alone to prevent one death is approximately 14. Paradoxically, no statistically significant differences in the need for surgical operation and the overall rate of hemos-tasis were demonstrated. Indeed, deaths in this study all occurred, as is usually the case, in patients who had significant co-morbidity. Complications in this study were minimal. Although this was not a direct comparison of adrenaline versus thrombin, it did strongly suggest that human thrombin is an effective modality.
Table 8.12 Adrenaline plus thrombin vs adrenaline alone for gastrointestinal bleeding 1.
Source : Kubba et al. [103]
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree