Bowel Obstruction and Hernia
Keywords
• Obstruction • Ileus • Hernia
Bowel obstruction
Bowel obstruction accounts for more than 15% of admissions from the emergency department (ED) for abdominal pain.1 Intestinal obstructions (85% small bowel, 15% large bowel obstructions) generated more than 320,000 hospitalizations in the United States in 2007 alone, with 3.4 billion dollars spent on diagnosis and management.2,3 At least 30,000 deaths per year are attributed to bowel obstruction. Delayed diagnosis and misdiagnosis of both small bowel and colonic obstruction are common and lead to increased morbidity and mortality.4 The elderly are particularly at risk, because bowel obstruction is a common cause of abdominal pain (12%–25%), with a mortality of 7% to 14%.5,6 A thorough understanding of these complex gastrointestinal pathologies is essential for the emergency physician. This article focuses on bowel obstruction in the adult patient. Pediatric gastrointestinal disorders are addressed in a separate article by Marin and Alpern elsewhere in this issue.
Small bowel obstruction
Background
Small bowel obstructions (SBO) are responsible for approximately 4% of all ED visits for abdominal pain. These cases account for around 20% of all surgical admissions.7,8 Although hernias were historically the most common cause for SBO, and continue to be a primary cause in some developing countries, postoperative adhesions now account for more than 75% of all obstructive events in the United States.9,10 There are a variety of other causes of SBO (Table 1). Although the overall mortality for patients with SBO is less than 3%, it is much higher in the elderly.11 When strangulation complicates SBO, mortality increases to 30%.12
Pathophysiology
In general, SBOs are categorized on the ability of fluid or gas to travel through the site of obstruction. From a physiologic perspective, there is either no passage (complete obstruction) or some passage (partial obstruction) of enteric contents past an obstruction. More precise classification is typically based on radiologic criteria. In complete obstruction, there is no passage of contrast. In low-grade partial SBO, there is minimal delay, whereas in high-grade partial SBO there is stasis or moderate delay of contrast passage.13 In simple terms, an occlusion of the small bowel results in the accumulation of bowel fluids and the production of gas from bacterial overgrowth. As the disease process progresses, bowel dilatation leads to increased intraluminal pressure. The feared consequences of this sequence are mucosal ischemia and, eventually, necrosis and perforation. Closed-loop obstructions, commonly caused by an incarcerated hernia or twisting of a loop of bowel, are particularly at risk. Strangulation denotes compromise in the vascular supply, either because of twisting of the mesentery or bowel, or simply from increased bowel wall transmural pressures.13
Clinical Features
Although the classic presentation of a complete SBO may be quickly recognized, partial SBO remains a diagnostic challenge. Decreased passage of flatus and/or stool, nausea and vomiting, abdominal distention with tympany to percussion, paroxysms of abdominal pain occurring every 4 to 5 minutes, dehydration, and tachycardia are all commonly described. Patients may initially experience colicky pain and high-pitched, hyperactive bowel sounds, but the clinical presentation may then progress to more continuous pain and hypoactive bowel sounds because of bowel fatigue. However, these various signs and symptoms are nonspecific and not reliably observed.14 Patients with obstruction may even continue to pass stool. Classically, distention tends to be more pronounced in distal, compared with proximal, obstructions. Moreover, complete obstruction is often more sudden in onset, with a more severe and rapid course than a partial obstruction. Mild, generalized tenderness is common in SBO. It may be alleviated by vomiting. In patients with a suspicion for obstruction, localized tenderness or peritoneal signs indicate intestinal ischemia or necrosis until proved otherwise. Pain that is constant and out of proportion to findings on physical examination is a commonly described feature of both closed-loop obstructions and bowel ischemia.
Laboratory Tests
Laboratory tests are both insensitive and nonspecific, and they do not improve diagnosis or predict the need for an operation.15 Leukocytosis is common and electrolyte abnormalities are sometimes observed. However, no laboratory test reliably detects obstruction, intestinal ischemia, or the need for operative management.4,15,16 Serum creatinine kinase, amylase, and lactate are generally increased only late in the course of bowel obstruction.
Imaging
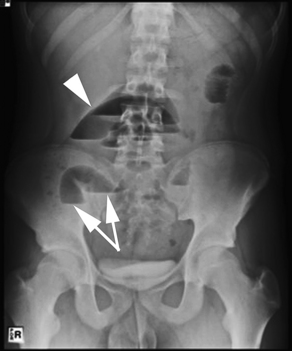
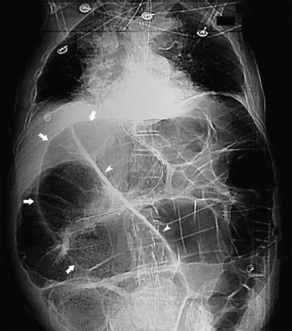
The classic findings on AXR are distended small bowel (>3 cm in diameter), air-fluid levels, and a paucity of colonic or rectal gas (Table 2, Fig. 1). Differential air-fluid levels are more specific for SBO than for other types of obstruction. Dilated bowel loops that are stacked one under the other are described as having a stepladder appearance. Overall, AXR are diagnostic in 30% to 70% of cases of SBO, with a specificity of approximately 50%.17,18 The cause of obstruction is rarely shown. There tends to be poor interobserver agreement in terms of the degree and location of obstruction, and discrepancies commonly exist on radiographic interpretations between emergency physicians and radiologists.19,20 In cases of proven SBO, AXR are interpreted as definite SBO in 50% of cases, probable SBO in 30%, and normal or nonspecific in 20%.15 AXR may also appear normal in cases of closed-loop or strangulated obstructions.21 Conversely, functional obstructions (eg, adynamic ileus) can have similar radiographic findings to SBO. Multiple gas-filled or fluid-filled loops of dilated small bowel, with little colonic gas, may also be observed in appendicitis, diverticulitis, or even mesenteric ischemia.22
Table 2 Radiographic findings for abdominal disorders
| Abdominal Radiographs | Computed Tomography | |
|---|---|---|
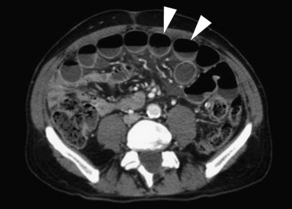
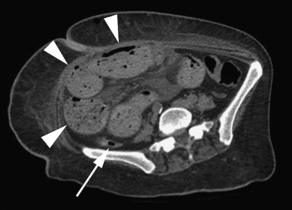
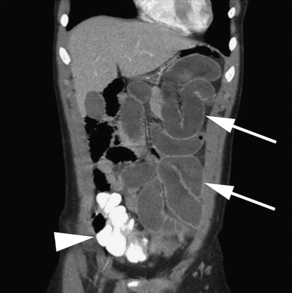
| Small bowel obstruction | Distended small bowel (>3 cm in diameter), air-fluid levels (differential or step-laddering early, string of pearls late), paucity of colonic or rectal gas | Dilated proximal small bowel loops (>3 cm) and collapsed distal small bowel ± a transition zone; small bowel feces sign; absence of distal contrast; bowel wall thickening (late) |
| Large bowel obstruction | Dilated colon (>5 cm), disproportionate distention of the cecum (>10 cm) if competent ileocecal valve | Dilated colon and disproportionate cecal distention, with collapsed distal colon or rectum |
| Colonic volvulus | Sigmoid: coffee bean appearance to dilated, doubled-back bowel segment, proximal large bowel dilated Cecal: kidney bean shape of single dilated segment of cecum, distal large bowel collapsed, small bowel dilatation | Sigmoid: entire colon (especially sigmoid) markedly dilated, whirl sign at site of torsion Cecal: very dilated cecum with bird’s beak or whirl sign (from twisted bowel and mesentery) |
| Ischemia/infarction | May see ileus or obstruction; thumbprinting or thickening of bowel wall, air in bowel wall (pneumatosis) or portal venous gas | Bowel wall thickening >5 mm, diminished or absent bowel wall enhancement, mesenteric fluid, air in bowel wall (pneumatosis) or portal venous gas; bowel wall edema (target sign) |
| Adynamic ileus | Distention of both small and large bowel loops, without a transition zone | Distention of both small and large bowel loops, without a transition zone |
| Acute colonic pseudo-obstruction | Extensive colonic distention (± small bowel), especially in the transverse colon, without a transition point (gas and stool in rectum) | Extensive colonic distention (± small bowel), especially in the transverse colon, without a transition point (gas and stool in rectum) |
Data from Schwartz DT. Abdominal radiology. In: Schwartz D, editor. Emergency radiology: case studies. New York: McGraw-Hill; 2008. p. 147–87; and Stoker J, van Randen A, Lameris W, et al. Imaging patients with acute abdominal pain. Radiology 2009;253(1):31–46.
Because of the low sensitivity of plain films in partial SBO, as well as a need for more accurate information regarding the level and cause of the obstruction, CT of the abdomen and pelvis plays an increasingly central role in diagnosis (see Table 2). Some investigators advocate omitting AXR and using CT as the first-line imaging modality. When the clinical picture is highly suggestive of complete SBO, AXR have comparable sensitivity with CT17 and may quickly confirm the diagnosis and expedite surgical disposition. However, if the clinical picture is equivocal and the AXR is likely to be nondiagnostic, CT may be a reasonable initial choice. The American College of Radiology (ACR) has proposed that CT with intravenous (IV) contrast is the most appropriate imaging modality even when complete or high-grade SBO is suspected.23 However, oral contrast material is not required in these cases because the intraluminal fluid in distended bowel loops acts as a natural contrast agent.24 Intravenous contrast improves the diagnosis of strangulation, ischemia, and other vascular pathologies such as superior mesenteric artery occlusion or superior mesenteric vein thrombus.4 Positive oral contrast (standard water-soluble contrast) or neutral (water) contrast may play a role in the evaluation of partial small bowel obstructions and is recommended by the ACR. The decision to add oral contrast should be determined on a case-by-case basis, with due consideration of ED throughput, the patient’s clinical condition, patient tolerance of the contrast, and the potential for added benefit with an oral contrast agent in identifying alternative diagnoses. A recent study, which addressed the value of nonenhanced (no IV, no oral contrast) CT in evaluating SBO, found a comparable accuracy between nonenhanced and enhanced CT.25
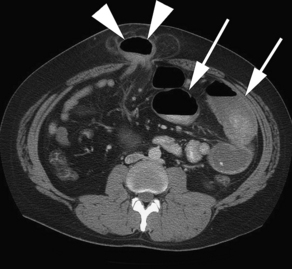
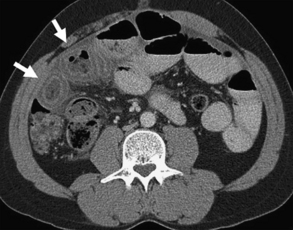
Findings on CT that are consistent with SBO include dilated small bowel loops proximal to collapsed loops distally (see Table 2). A transition zone is often identified, and a small bowel feces sign is highly specific (Figs. 2–5). The small bowel feces sign derives from the inspissated debris in the dilated, obstructed small bowel; it has the appearance of colonic fecal material. Overall, CT has a sensitivity of 64% to 94% and specificity of 79% to 95% in the detection of SBO, with an accuracy of 67% to 95%.17,26,27 It provides information regarding the degree of obstruction (low grade vs high grade), proximal or distal location, and the specific cause of the obstruction. In particular, CT identifies closed-loop obstructions that have a high risk of ischemia and mandate surgery. CT is also beneficial in distinguishing SBO from other abdominal disorders such as inflammatory bowel disease, appendicitis, diverticulitis, and ileus.28 Although CT is accurate in the diagnosis of complete and high-grade obstruction, it is only around 50% sensitive for low-grade partial obstructions.29
Regarding CT detection of ischemia and strangulation, a meta-analysis reported 83% sensitivity (63%–100%) and 92% specificity (61%–100%) in detecting intestinal ischemia.30 However, one prospective study by Sheedy and colleagues31 reported a much lower sensitivity (52%) in the detection of ischemia; the cause of this discrepancy is unclear. The most common CT findings suggestive of ischemia and/or infarction include bowel wall thickening and diminished or absent bowel wall enhancement by IV contrast (see Table 2; Fig. 6). It is possible that modern multidetector CT (MDCT) with coronal reformations may significantly improve diagnostic certainty in the identification or exclusion of SBO, as well as the specific cause and level of the obstruction.32
Abdominal ultrasound is commonly performed outside the United States for assessment of SBO. It is also recommended for this purpose by the ACR. When studied, ultrasound has been shown to have similar sensitivity and greater specificity compared with plain films.33–35 Several factors argue for the superiority of CT, including the operator dependency of ultrasound, the logistical challenges of around-the-clock sonographic assessment, limited experience with abdominal sonography in bowel evaluation, and interobserver variability in the interpretation of ultrasound images of the small bowel.22
The literature regarding MRI in the diagnosis of SBO is limited. One study reported a high sensitivity (95%), specificity (100%), and accuracy (71%).36 In general, there is increasing evidence to suggest that MRI can reliably identify SBO and provide information regarding location and cause. The ACR recommends MRI only in particular circumstances, including pregnant patients with concern for SBO.23
SBFT with barium or water-soluble contrast allows visualization of dilated loops of bowel, while identifying a transition point as defined by barium. Because SBFT is time consuming and the volume of contrast is not well tolerated by patients, it has been largely supplanted by CT in the United States.7 Enteroclysis involves intubation of the small bowel through the stomach (often under conscious sedation), allowing infusion of enteral contrast directly into the jejunum. This technique is only applicable to the subacute setting, because it is time-intensive and resource-intensive, although when used appropriately has been reported by 1 author to have 100% sensitivity, 88% specificity, and 86% accuracy in determining the cause of obstruction.18 With the advent of MDCT, CT enteroclysis is increasingly used for the nonemergent evaluation of SBO. It is both more sensitive (89%) and more specific (100%) than CT regarding partial SBO.37 Similar to conventional enteroclysis, this modality necessitates intubation of the descending duodenum under fluoroscopy, infusion of enteric contrast material, and an abdominopelvic CT.38
Management and Disposition
Most surgeons support conservative therapy for SBO in the absence of peritonitis or strangulation. Success rates with nonoperative management range from 43% to 73%.39–41 Of those successfully treated, more than 80% have substantial improvement within 48 hours. In partial SBO, strangulation only occurs in a minority of cases (3%–5%) that are managed conservatively.40
Conservative management includes intravenous resuscitation, decompression, and bowel rest. Rehydration is essential, because liters of gastrointestinal secretions can accumulate during an obstructive episode. Nasogastric tube decompression remains a mainstay of therapy, with no added benefit from long intestinal tubes.42 There are no data to support or refute the administration of antibiotics in SBO, although they are often given before surgery.
Although conservative management is common practice, several studies do suggest that a nonsurgical approach renders a greater risk of recurrent obstruction.10,41,43 Nonetheless, recurrence is common with or without surgical intervention, and further surgeries lead to additional adhesion formation. Adhesive SBO has been shown to recur in 19% to 53% of cases.43,44 Recurrence also becomes more likely with each subsequent SBO episode.43
With complete obstruction, the failure rate of conservative management is higher, and subsequent complications more severe. The dictum “never let the sun rise or set on a small bowel obstruction” continues to have some validity. Although 35% to 50% of those with high-grade and complete obstruction may resolve with fluid resuscitation and bowel decompression alone, many advocate early surgical intervention owing to the high failure rate of conservative management.45,46 Around 30% of patients with complete SBO require bowel resection because of compromise of the small intestine.46 Of all patients admitted to the ED with SBO, an estimated one-quarter undergo surgery.47 Other causes of SBO, including impacted gallstones, bezoars, food, or other foreign bodies, are generally managed endoscopically or surgically.
The frequency of strangulation is largely dependent on the degree of obstruction. Strangulation is less often observed in nonoperative, partial obstructions (typically ≤10%). Conversely, retrospective studies of obstructions requiring surgery, in particular high-grade or complete obstructions, report strangulated bowel in 25% to 45% of cases. When looking at all surgeries for SBO, nonviable strangulation (necrotic bowel) is found in 13% to 16% of cases. Strangulation risk is also known to increase with patient age.40,43,48,49 The rapid identification of strangulation is essential, because delayed surgery (>24 hours after the onset of symptoms of strangulation) increases mortality threefold.48 Clearly, patients with fever and peritonitis require early operative intervention, because this is suggestive of ischemia and strangulation. In cases in which the CT is negative but the clinical examination is concerning, surgical consultation is warranted because of the false-negative rate of CT.
The admission service is also important in the management of SBO. One study showed a higher mortality when patients with SBO were admitted to a medical, rather than a surgical, service. This increased mortality was attributed to delays in surgical disposition.50 Bowel obstructions are most appropriately admitted to a surgical service.
Large bowel obstruction
Background
Large bowel obstruction (LBO) occurs much less frequently than SBO, although it is disproportionately more common in the elderly. Around 60% of LBOs are caused by neoplasm. Of all patients diagnosed with primary colorectal cancer, 15% to 20% initially present with a malignant bowel obstruction (MBO).51–53 Colonic volvulus, involving either the sigmoid or cecum, is responsible for an additional 5% to 15% of cases of LBO.54,55 Volvulus, which most commonly occurs in the sigmoid or cecum, is typically a disease of older patients, although it may affect all ages. Sigmoid volvulus is often observed in debilitated, institutionalized patients; diet and constipation may contribute. Cecal volvulus is attributed to a congenital defect in the mesentery that allows for increased bowel motility and occasionally torsion.56 Another 10% of LBO cases are related to strictures from chronic diverticular disease (see Table 1). LBO carries a higher morbidity and mortality than SBO because of an older patient population and a variety of underlying medical conditions. Overall in-hospital mortality is approximately 10%, with poorer outcomes observed in those with renal failure, peritonitis, proximal colonic ischemia, and infarction.57
Pathophysiology
The ileocecal valve plays an important role in colonic obstruction. If the valve is competent, then fluid and gas necessarily accumulate in the cecum, resulting in a functional closed-loop obstruction. The cecum rapidly dilates, producing further wall tension because of a larger radius (Laplace law).4 Eventually, acute dilatation may result in wall ischemia and perforation. Regardless of the ileocecal valve, volvulus is of particular concern because it creates a closed-loop segment of bowel that can quickly become ischemic and perforate.
MBO may result from external, intramural, or intraluminal compression. Tumor infiltration of the mesentery, muscle, and nerves may cause bowel dysmotility, further contributing to obstructive findings.58
Clinical Features
LBO commonly presents with abdominal pain, distention, and progressive obstipation. However, these features are nonspecific, and clinical diagnosis is difficult because the onset of symptoms is frequently gradual, especially with MBO. With continued colonic absorption of nutrients, water, and electrolytes, LBO may be tolerated for days to weeks before the patient seeks medical care. Because of vascular effects, volvulus may present more acutely with pain, abdominal distention, and occasionally shock. Important elements in the history include the patient’s bowel habits, recent weight loss, rectal bleeding, history of inflammatory bowel disease, and prior obstruction.59 Diarrhea is common, because there may be overflow from bacterial liquefaction of blocked fecal material. In contrast with the early onset of vomiting after oral intake in SBO, patients with LBO may never develop vomiting if the ileocecal valve is competent.58 Marked abdominal pain or fever is suggestive of underlying ischemia, perforation, or peritonitis. Rectal examination may identify bleeding, a mass (malignancy), or a large volume of stool suggesting fecal impaction.
Laboratory Studies
Similar to small bowel obstruction, laboratory studies in LBO are generally unhelpful. A significantly increased white blood cell count may be concerning for ischemia or perforation, and electrolyte abnormalities may be present because of vomiting or poor oral intake.56
Imaging
The reliability of abdominal radiographs in the evaluation LBO is poorly studied. One retrospective study described abdominal plain films as 84% sensitive and 72% specific in the diagnosis of LBO.60 However, another study reported that one-third of patients diagnosed with LBO based on clinical assessment and AXR had no obstruction. Conversely, up to 20% of patients initially believed to have pseudo-obstruction were then shown to have a mechanical LBO.61 There is also poor interobserver agreement in AXR of large bowel obstructions.19 Radiographs provide limited information regarding the underlying disease process. Moreover, the sensitivity of AXR for volvulus has been reported as less than 50%.62 LBO presents radiographically as marked colonic dilatation, with disproportionate distention of the cecum (>10 cm diameter) if the ileocecal valve is competent. Volvulus has several unique radiographic findings, as described in Table 2 and Fig. 7.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree