Abdominal Pain in Special Populations
Keywords
• Abdominal pain • Immunocompromised • Immunosuppressed • Post-procedure
The immunocompromised patient
Patients with altered immunologic function comprise a heterogeneous population ranging from those who are mildly immunocompromised (eg, elderly, uremic, diabetic) to those who are moderately to severely immunocompromised (eg, current immunosuppressive therapy, post transplant, acquired immunodeficiency syndrome [AIDS], active malignancy undergoing chemotherapy). Compared with immunocompetent hosts, this population suffers from the same spectrum of diseases, but because of their blunted immune response they may present with atypical symptoms such as altered mental status and tachycardia, lack the classic signs of an acute abdomen, and/or seek medical attention later in their disease course.1 For example, transplanted organs lack native innervation, so even pain is an unreliable sign of underlying disease.2 In addition to the surgical conditions that must be considered in any patient with acute abdominal pain, the differential diagnosis should be expanded to include nonsurgical problems and infections that may be unique to this population. These diseases include cytomegalovirus (CMV) infection, neutropenic enterocolitis (typhlitis), and intra-abdominal abscesses. Because they are so challenging to diagnose and are more likely to harbor a potentially life-threatening disease, immunosuppressed patients will often receive diagnostic imaging with abdominal-pelvic computed tomography (CT) as part of their emergency department (ED) evaluation.
Human Immunodeficiency Virus/AIDS
The introduction of highly active antiretroviral therapy (HAART) has decreased the incidence of opportunistic infections (OIs) and gastrointestinal diseases. A retrospective study of 108 human immunodeficiency virus (HIV)-positive patients (84% on antiretroviral therapy or HAART, 44% with CD4 <200 cells/mm3) with undifferentiated abdominal pain3 showed that only 10% of patients had an OI as compared with pre-HAART reports of 41% to 86%.4–6 Disseminated mycobacterial disease was the most common diagnosis. Other OIs included Candida esophagitis, AIDS cholangiopathy, lymphoma, and intra-abdominal tuberculosis. Well-described causes of abdominal pain such as CMV colitis or peritonitis, cryptosporidiosis, and Kaposi sarcoma were not seen at all in this San Francisco population. Moreover, only 9 patients (8%) required surgical intervention, one of which was for drainage of a Mycobacterium abscess (the only HIV-associated OI). A more recent study of opportunistic gastrointestinal disorders in HIV patients corroborated these findings, showing that 26% of patients on HAART (vs 80% in the no-treatment group) had an OI, most commonly Candida esophagitis and CMV esophagitis or colitis.7 Kaposi sarcoma and lymphoma were less common.
The primary conditions requiring acute surgical intervention in HIV patients are appendicitis, cholecystitis, bowel obstruction, and intestinal perforation.8 These surgical conditions occur as frequently in HIV patients on HAART as in non-HIV patients,7,9 although the underlying pathology may be different. In immunocompromised patients, CMV infection can cause vasculitis in the gastrointestinal tract leading to ulcerations in the bowel wall, particularly in the terminal ileum and colon. CMV colitis may present acutely as gastrointestinal hemorrhage, perforation, or toxic megacolon.10 Appendicitis can also be caused by CMV infection and may present with more indolent symptoms. In addition, intestinal obstruction and intussusception may be caused by Kaposi sarcoma and lymphoma.8,9 Finally, although gallstone cholecystitis occurs equally in non-HIV and HIV patients, acalculous cholecystitis occurs more frequently in the HIV population. Moreover, these patients can develop cholangiopathy, both as an adverse effect of antiretroviral medications and from infiltration of opportunistic pathogens into the biliary ducts causing obstruction.9
In addition to the conditions already mentioned, abdominal pain in HIV patients may be an adverse effect of the same antiretroviral medications that have been shown to decrease gastrointestinal diseases. Patients will often present with associated symptoms of nausea, vomiting, and diarrhea, sometimes severe enough to cause dehydration or hemodynamic instability. Some medications are associated with specific conditions. For example, didanosine (Videx) can cause acute pancreatitis and indinavir (Crixivan) can precipitate in the kidneys, leading to nephrolithiasis.8 More often than not, the discomfort caused by these medications results in intermittent medication noncompliance and sometimes discontinuation of the treatment entirely.11 Lapses in treatment may increase a patient’s risk of developing OI.
Malignancy
Approximately 40% of the ED visits by cancer patients are for abdominal pain.12 The differential diagnosis of abdominal pain in this patient group should include conditions directly related to the malignancy itself in addition to complications of the malignancy treatment.13 Not surprisingly, intra-abdominal cancers increase a patient’s risk of developing a small bowel or large bowel obstruction,14 although non–intra-abdominal cancers (ie, breast, melanoma) can also cause bowel obstruction due to diffuse peritoneal carcinomatosis.15 A diagnosis of large bowel or gastric outlet obstruction in an otherwise healthy patient or someone with a history of prior cancer should increase the suspicion of an underlying or recurrent malignancy.13,14 Furthermore, solid tumors can become a lead point in causing intussusception, which is otherwise uncommon in healthy adults.16 Bowel perforation may be caused by transmural erosion of gastrointestinal cancers, intestinal metastatic lesions, and atypical infections. Pneumoperitoneum can be challenging to diagnose, as patients may have difficulty localizing the infection and therefore have a delayed presentation.1 In addition, patients with cancer can develop malignant ascites, causing abdominal distention and pain, and Budd-Chiari syndrome, a constellation of symptoms caused by hepatic venous outflow obstruction from thrombosis.13 In evaluating these patients, Abdominal CT is more clinically useful because it can detect closed-loop or strangulation obstruction, identify a transition point, and diagnose intestinal pneumatosis or vascular thrombosis,13 as compared with plain radiography, which has a sensitivity of only 66%17 for detecting small bowel obstruction and an inability to identify strangulated bowel or vascular thrombosis.18
Gastrointestinal symptoms can also be an adverse effect of the treatment of the underlying malignancy. Chemotherapeutic agents frequently cause abdominal pain associated with nausea, vomiting, and/or diarrhea. Radiation therapy to the abdomen or pelvis can lead to a progressive occlusive vasculitis and local narrowing of the intestinal lumen. Radiation enteritis is a spectrum of disease including acute bowel ulceration, intestinal perforation, and/or massive gastrointestinal hemorrhage, and may lead to chronic fistula formation and strictures.19
Cancer treatment with chemotherapy can also cause profound neutropenia. In neutropenic patients, the most common cause of an acute abdomen is neutropenic enterocolitis (NEC) or typhlitis.13 NEC is a necrotizing inflammation of the cecum and the adjacent small intestine in the setting of chemotherapy-induced neutropenia or bone marrow transplantation. Also known as necrotizing enterocolitis, it has been described in patients with aplastic anemia, AIDS, and organ transplantation. Cytotoxic agents such as the taxanes and vinorelbine (Navelbine) may also increase a patient’s risk of NEC.20
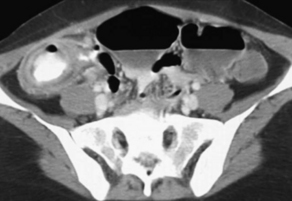
Symptoms of NEC include fever, nausea, vomiting, abdominal distension, diarrhea, and right lower quadrant pain, which may be initially thought to be due to appendicitis. Patients may also present with hypotension and other signs of sepsis. Although the symptoms of NEC may be difficult to distinguish from the routine side effects of the chemotherapeutic agents themselves, it is more likely to be associated with bowel wall thickening on abdominal CT of more than 4 mm, with a very poor prognosis if greater than 10 mm (Fig. 1).21,22 Treatment includes fluid resuscitation, broad-spectrum antibiotics effective for enteric gram-negative bacilli, Enterococcus spp, and anaerobes, bowel rest, and parenteral nutrition as needed.20 Surgical intervention is reserved for bowel ischemia and perforation.23 Mortality and morbidity rates are high so prompt recognition may be life-saving.24
Solid Organ Transplant Patients
Like many postsurgical patients, the two most common reasons for solid organ transplant patients to visit the ED are abdominal pain (31%), often associated with other gastrointestinal symptoms, and infectious symptoms (17%) such as fever or wound infection.2 In this ED study, infection (36%) and gastrointestinal or genitourinary pathology (20%) were the most common ED diagnoses, regardless of the time elapsed since transplantation. Moreover, the majority of transplant patients (61%) were hospitalized (compared with the 17% overall admission rate in the same institution during the study period), regardless of their diagnosis, presumably because a missed diagnosis may have serious consequences for both the graft and the patient. Any posttransplant patient with abdominal pain must be evaluated for organ rejection, systemic infection, and drug toxicity.
Specific conditions to consider in these patients will depend on the time elapsed since transplantation. In the early posttransplant period (<1 month), postsurgical complications and infections predominate.2 The surgical anastomosis may constrict or leak, causing bowel obstruction or peritonitis. Graft injuries, such as bile duct ischemia, can later become a liver abscess. Viral or candidal infections may be either donor-derived or a surgical complication. Clostridium difficile colitis is also common during this period, as opposed to OIs, which are typically absent.25 In addition, patients have a slightly higher risk of graft rejection within the first month than within the intermediate or late posttransplant period.2
During the intermediate posttransplant period (1–6 months), viral infections and graft rejection are the most common reasons for patients to develop a fever.25 Because the full effect of immunosuppressants is now present, OIs such as CMV colitis and intra-abdominal abscesses caused by fungal (eg, Candida, Cryptococcus) and bacterial infections (eg, Nocardia, Legionella) may develop. These patients should be considered severely immunocompromised and highly susceptible to infection.
In the late posttransplant period (>6 months), the risk of infection typically declines slightly as immunosuppressive therapy is tapered down in patients with good graft function. However, patients continue to be at risk for developing chronic rejection, often as a result of chronic viral infections.25 Acute diverticulitis is a common gastrointestinal infection seen during this period; patients may present with perforation due to a delay in diagnosis. In addition, this population may also develop posttransplantation lymphoproliferative disorder (PTLD), a lymphoproliferative disorder thought to be associated with Epstein-Barr virus infection. Patients with suspected PTLD may present with fever, a mononucleosis-like syndrome, gastrointestinal obstruction, bleeding, or perforation, and have significant hepatic or pancreatic dysfunction. Regression of PTLD may occur with a reduction in immunosuppressant therapy, although this disease often will require chemotherapy or immunotherapy.25
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree