Fig. 54.1
T2W MRI image of a patient with BASM. The liver is cirrhotic with a large central regenerative nodule and atrophied left lobe and posterior segments of the right lobe. There is polysplenia and interrupted IVC with hemi-azygous continuation ( arrow)
When dilated ducts are detected, the differential will include choledochal malformation , inspissated bile syndrome and Caroli with congenital hepatic fibrosis . In these instances, the patient needs further evaluation to define the anatomy; historically, this was achieved by means of direct percutaneous cholangiography (PTC) or endoscopic retrograde cholangiopancreatography (ERCP), but the implementation of magnetic resonance imaging (MRI) with heavily T2-weighted cholangiography sequences has allowed the radiologist to achieve the diagnosis confidently and in a noninvasive way [4].
Magnetic resonance cholangiopancreatography (MRCP) can in fact classify the different types of choledochal malformations and reveal anomalies of the pancreatico-biliary junction (more often associated with type I cysts) with possible complications, such as calculi within a long common channel (Fig. 54.2).
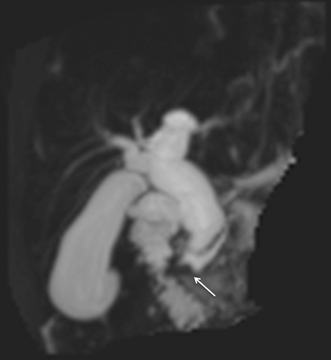

Fig. 54.2
MRCP MIP reconstruction of the biliary tree showing a type of choledocal malformation with an impacted calculus within a long common channel ( arrow)
In case of Caroli , MRCP will show multifocal cystic dilatation of the intrahepatic bile ducts, possibly containing filling defects, representing calculi; these cysts communicate with the biliary tree, thus excluding autosomal dominant cystic liver disease and biliary hamartomas. A specific MR finding of Caroli is the “central dot sign,” a portal vein branch protruding into the lumen of a dilated duct, which enhances with gadolinium [5].
ERCP is not routinely indicated but may still be required in doubtful cases or as a therapeutic option in those patents where MRCP has identified an obstructed biliary system due to inspissated bile or choledocholithiasis (shown as signal voids) .
Older Children
Jaundice in older children can be caused by hepatocellular disease (acute or chronic) and obstructive causes and imaging investigations can distinguish these entities and often establish the etiology of many chronic conditions and acquired or developmental biliary disorders.
Ultrasound findings in acute hepatitis are normally nonspecific, with US demonstrating hypoechoic parenchyma, increased periportal reflectivity due to edema, and thickened gallbladder walls.
In the context of chronic liver disease, radiology can confirm the clinical diagnosis demonstrating coarse, heterogeneous liver parenchyma and abnormal liver architecture as well as signs of portal hypertension (Fig. 54.3).
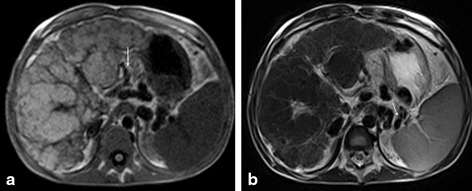

Fig. 54.3
MRI T1W (a) and T2W (b) images of a cirrhotic liver with markedly abnormal contour and varying size-regenerative nodules throughout. The portal vein is occluded and there are numerous short gastric and splenic varices ( arrows)
Medical causes of jaundice in older children include Wilson’s disease , cystic fibrosis, glycogen storage disorder, tyrosinemia, and alpha1 antitripsin deficiency. In all these conditions, US will show nonspecific changes with hyperechoic liver parenchyma; however, these patients are prone to develop focal liver lesions (such as adenomas and, importantly, hepatocellular carcinomas) and in this instance, US is extremely useful as a noninvasive, radiation-free surveillance test. In addition, some of these conditions have specific imaging features, such as multiple small nodules with low signal intensity in T2 on MR in Wilson’s disease and focal biliary cirrhosis with periportal fibrosis seen on US and MR in cystic fibrosis.
Calculi, benign strictures (as seen in primary sclerosing cholangitis, PSC), and neoplasms (which will be discussed separately) are all obstructive causes of jaundice in older children.
Calculi are seen in US as hyperechoic foci with posterior acoustic shadowing, mobile when detected within the gallbladder; these are seen as signal voids on MR. Inspissated bile is again hyperechoic in US but does not cause posterior shadowing.
Primary sclerosing cholangitis (PSC) is characterized by inflammation and fibrosis of the biliary tree, which results in cholestasis with progression to secondary biliary cirrhosis and hepatic failure. Histologically, the intrahepatic bile ducts are surrounded by cuffs of inflammatory cells and fibrosis, which results in segmental dilatation of the peripheral ducts alternated with narrow or obliterated segments, reflected in the imaging findings. The radiological diagnosis of PSC is often challenging, and needs confirmatory clinical, biochemical, and histologic findings. Traditionally, ERCP and PTC have been used as standard imaging procedures for the radiological diagnosis of PSC, however, MRCP has nowadays replaced these techniques.
Ultrasound remains the first-line investigation in these patients and shows segmental duct dilatation, irregular thickened walls of the CBD (Fig. 54.4), lymph nodes at the porta hepatis and heterogeneous reflectivity of the liver parenchyma (depending on the stage of disease). In established chronic liver disease, there is coarse echotexture with nodularity and relative atrophy of the right lobe with hypertrophy of the caudate and lateral segment of the left lobe.
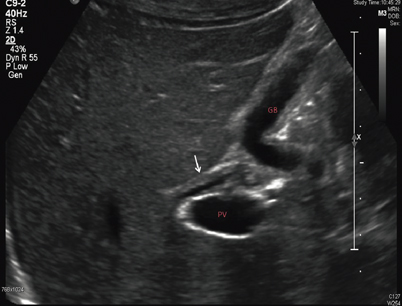

Fig. 54.4
US of the liver of a patient with PSC shows thickened wall of the common bile duct ( arrow). PV portal vein, GB gallbladder
MRCP can depict ductal irregularity, stricturing, focal dilatation, beading, and pruning affecting the intra- or extrahepatic biliary tree (Fig. 54.5). Post-contrast dynamic MR sequences should also be included to look for enhancement of thickened walls of the extrahepatic ducts and cholangiocarcinoma.
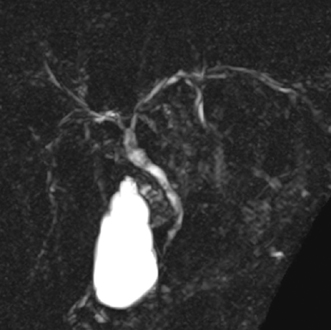

Fig. 54.5
MRCP MIP reconstruction of the biliary tree showing irregularity of the walls of the intra- and extrahepatic bile ducts, with focal structuring, in keeping with PSC. MIP maximum-intensity projection
Congenital and Acquired Vascular Disorders
Portal Hypertension
The etiology of portal hypertension can be classified as cirrhotic and non-cirrhotic. The latter can be prehepatic (portal vein thrombosis), intrahepatic (portal vein sclerosis, congenital hepatic fibrosis , steatosis, nodular regenerative hyperplasia, veno-occlusive disease), or suprahepatic (Budd–Chiari). Depending on the etiology, different radiological findings will be present. Liver function tests may be normal, and the diagnosis of portal hypertension often relies on demonstration of splenomegaly and formation of portosystemic collaterals.
In cirrhosis, prehepatic and pre-sinusoidal portal hypertension, the role of radiology is to determine the extent of involvement of the portal venous system, as this would influence the therapeutical options, which include surgical portosystemic shunt, Rex bypass, transjugular intrahepatic portosystemic shunt (TIPS), and ultimately liver transplantation .
The radiological approach to children with portal hypertension starts again from US with Doppler, which can detect low velocity, retrograde, or absent flow in the main portal vein and intrahepatic divisions. A patent umbilical vein, splenomegaly with varices, and ascites can also be easily seen on US; however, cross-sectional imaging is used as a “roadmap” for the evaluation of the portal system and collaterals pathways; CT and MR venography (MRV) can also demonstrate cavernous transformation of the portal vein, seen as a typical “beaded” appearance at the porta hepatis, which consists of venous channels within and around a previously stenosed or occluded portal vein [6]. This can occur within a few weeks after a thrombotic event, even in case of partial recanalization of the thrombosed segment, and can extend to the intrahepatic branches. Cystic and pericholecystic dilated veins can also be seen. A computed tomography (CT) performed in the arterial phase often shows patchy areas of high density at the periphery of the liver parenchyma, and this is due to a peripheral increase of the arterial inflow to supply the areas not reached by the cavernous portal vein.
Intra- and Extrahepatic Vascular Shunts
Intrahepatic communications between portal vein, hepatic artery, and hepatic veins are rare, and the most common, although often difficult to demonstrate, are small arterioportal shunts seen in cirrhotic livers. Large connections include portosystemic shunts (intra- and extrahepatic), arterioportal shunts, and arteriovenous shunts/malformations.
The origin of portosystemic shunts is still controversial and congenital, genetic, (such as trisomy 21) and acquired causes (such as in cirrhosis and post-traumatic) have been postulated [7]. These shunts have been historically classified into intrahepatic and extrahepatic, however, some authors discourage this classification and suggest to use an anatomical classification, which is aimed to establish the origin of the shunt (main portal vein, its tributaries or intrahepatic branches) and its systemic termination, the type of communication (end-to-end or side-to-side) and the number of connections. A distinction should be made between these shunts and a persistence of the ductus venosus [8].
The aim of radiology is to detect the shunts, delineate the anatomy , assist planning, and monitoring of interventional/surgical corrections and follow-up. Doppler US is the modality of choice to achieve this: direct communication between a portal branch and hepatic vein or the doctus venosus can be readily demonstrated, often due to enlargement and tortuosity of the vessels involved; the flow is usually continuous but triphasic flow can be seen in the portal branch. In case of main portal vein IVC connections, there is low velocity flow or non-visible intrahepatic portal vein, and the liver is usually decreased in size. Cross-sectional imaging is performed routinely to confirm the diagnosis and define the anatomy prior to intervention or surgery; in addition, shunts between the splenic vein or superior mesenteric vein and the IVC may be easily missed on US. CT and MR are also useful to characterize focal liver lesions, often found in associations with these shunts.
Angiography is often part of the workup of these children, either as a therapeutic option or prior to surgery to detect non-visible intrahepatic portal vein branches; a balloon occlusion catheter is placed in the shunt and pressures are measured before and after occlusion to evaluate how closure will be tolerated.
Arterioportal shunts may be congenital (in Rendu–Osler disease) or acquired (posttraumatic, post-liver biopsy, cirrhosis).
On CT, small arterioportal shunts in cirrhosis appear as small, wedge-shaped, peripheral areas of increased attenuation with early portal venous filling in the arterial phase and uniform attenuation in the portal venous phase. In presence of large arterioportal shunts or fistulas, there is early and marked enhancement of the main portal vein or segmental branches during the arterial phase. At Doppler US, large shunts will manifest with pulsatility of the portal vein flow [9].
Connections between the hepatic artery and systemic veins are rare, and can be seen in congenital arteriovenous malformations (AVM), hepatocellular carcinoma (HCC) and large hemangioendotheliomas; on Doppler US, altered waveforms of the hepatic vein can be seen in severe AVM, whereas CT will show asymmetrical, early filling of the hepatic vein.
Budd–Chiari Syndrome
Budd–Chiari syndrome is the clinical manifestation of hepatic venous outflow obstruction at any level of the hepatic veins, IVC or right atrium; this could be primary, caused by an endoluminal thrombus or membrane or secondary, when the occlusion is due to nonvascular material or from extrinsic compression. On US, there is narrowing, lack of visualization or thrombosis of the hepatic veins/IVC, with absent or monophasic flow at color Doppler. There is enlargement of the caudate lobe, ascites , and signs of portal hypertension with retrograde flow in the portal vein and splenomegaly .
On cross-sectional imaging, in the acute setting, occlusion of the hepatic veins with severe ascites is the typical finding. There is patchy, decreased peripheral enhancement of the liver due to portal and sinusoidal stasis and higher enhancement of the central parenchyma and caudate lobe, which compresses the IVC (Fig. 54.6). In subacute Budd–Chiari syndrome, portal vein thrombosis can develop as the result of reduced portal flow caused by blockage of the outflow. Finally, in chronic stages, multiple regenerative nodules can be seen, and on MR, these are bright on T1-weighted images and strongly hypervascular on post-gadolinium sequences. The nodules are predominantly isointense or hypointense relative to the liver on T2-weighted images [10] .
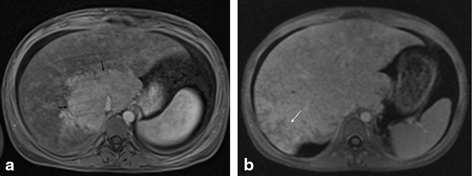

Fig. 54.6
MRI post-gadolinium T1W images of a patient with Budd–Chiari. In the acute phase a, there is significant enlargement of the caudate lobe ( black arrows), which enhances normally versus the poorly vascularized peripheral parenchyma. The IVC is compressed by the caudate lobe. In the subacute phase b, there are signs of paraseptal necrosis ( white arrow) with preserved central enhancement
Imaging of Transplant Liver
Liver transplantation is the treatment of choice for children with end-stage acute or chronic liver failure in which other therapies have failed or are not available. The successful development of novel surgical techniques of segmental, split, auxiliary, and living donor transplantation, together with advances in organ preservation, immunosuppressive therapy and adequate choice of donor organ, have led to a reduced rate of complications. However, these new surgical techniques have also brought with them new potential complications: in living-related transplants, for example, when the vascular pedicle may be too short, an autologous iliac artery conduit can be used, leading to potential higher risk of vascular complications.
Early recognition is crucial to the successful management of these complications, and a multimodality imaging approach is most effective to achieve the diagnosis, except in case of graft rejection, in which radiology has no role.
Serial ultrasound is the screening modality of choice immediately postoperatively and can be easily performed at the patient’s bedside; however, ultrasound has inherent limitations and, even though the use of ultrasound contrast agents can improve its sensitivity, it depends on the expertise of the operator, and cross-sectional imaging is often necessary in inconclusive cases. Conventional angiography is now limited to those cases in which endovascular treatment is required.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







