Imaging and Laboratory Testing in Acute Abdominal Pain
Keywords
• Abdominal pain • Computed tomography • Ultrasonography
Overview of laboratory testing
Acute abdominal pain was the leading symptom-related cause for visiting an emergency department (ED), at 6.7% or 8 million visits, in 2006.1 Laboratory work in the patient with abdominal pain can serve to make a diagnosis, indicate the severity of disease, or direct attention toward coexisting medical problems. However, tests may result in incidental findings that may confound the clinical picture. For this reason, tests should be ordered with a specific clinical question in mind and, when possible, with a clear sense of the pretest probability and likelihood ratios engendered by a positive or negative outcome of the test.
Determining the value of an individual laboratory test for a patient with abdominal pain is difficult. In fact, the 1994 American College of Emergency Physicians (ACEP) policy statement recommends laboratory analysis for very few clinical diagnoses in patients with abdominal pain. However, clinical practice certainly varies significantly from this policy. Some studies have looked at the diagnostic importance of individual laboratory tests in the evaluation of specific conditions or suspected conditions (eg, suspicion of appendicitis). While review of data shows that individual laboratory studies taken in isolation infrequently “make” a diagnosis, results of laboratory studies do frequently affect disposition or treatment in the ED. In addition, many studies investigating laboratory testing in abdominal pain involve protocols in which laboratory tests are ordered for all patients and then tailored to the individual case after history and physical examination.2–5 Most abdominal labs test general physiology, for example the complete blood count (CBC). Few tests are as specific as the lipase and urinalysis. In addition, even tests that are relatively specific, such as aspartate transaminase (AST) and alanine aminotransferase (ALT), may be abnormal in many different conditions.
Overview of diagnostic imaging
Imaging plays an important role in the evaluation of patients with acute abdominal pain. In 2005, 44% of ED visits included imaging; 35% of visits included plain radiography, 11% had CT, 3% had ultrasonography (US), and 0.5% magnetic resonance imaging (MRI).6 While these statistics are for all complaints, not just for abdominal pain, it reflects the reliance on ancillary testing in the ED. When evaluating a patient with acute abdominal pain, the decision to order an imaging study, just as with laboratory testing, should come from information gleaned from a comprehensive yet focused history and physical examination. It is important to remember that all diagnostic testing has significant false-positive and false-negative results, and in the setting of a high pretest suspicion for disease, a negative test does not rule out disease. In addition, radiographic imaging may carry risks such as contrast and radiation exposure, and as such it is important to consider risks and benefits of an imaging modality when evaluating a patient with acute abdominal pain.
Plain Abdominal Radiography
Plain radiography has historically been the initial imaging modality used for the evaluation of abdominal pain, due to its ease of acquisition and cost. With the increased availability and technological advances of other imaging modalities such as CT and US, the usefulness of plain radiographs has diminished. With alternative imaging readily available, the usefulness of abdominal radiographs in the nontraumatic patient is controversial. In a recent study of patients whose abdominal radiograph was interpreted as normal or nonspecific, the majority (81%) had positive findings on CT, US, or upper gastrointestinal imaging.7 The investigators found that plain radiographs led to a change in management in only 4% of patients. In another study, when compared with unenhanced helical CT, 3-view abdominal radiographs yielded only an overall sensitivity of 30%, specificity of 88%, and accuracy of 56% (95% confidence interval 0.46–0.66), with a negative predictive value of 51%.8 Given the poor performance, added cost, and increased radiation dose, plain radiographs are indicated only in specific, limited settings.
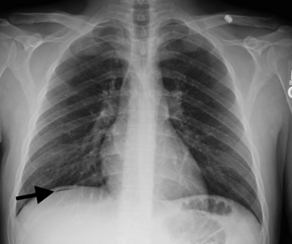
Some have suggested that abdominal radiography be used solely to identify bowel obstruction, perforation, foreign body ingestion, or localization of catheter placement.9 However, in obstruction, abdominal radiography alone does not provide the exact location or extent of the obstruction (partial, complete, or intermittent) and often requires further imaging such as CT.10 Thus, a patient may be subjected to multiple tests with ionizing radiation. Pneumoperitoneum can be seen on an abdominal series radiograph; however, is often better visualized on an upright chest radiograph, which subjects the patient to shorter exposure times, less radiographic penetration, and more tangential alignment of the x-ray beam to the diaphragm (Fig. 1).11 Abdominal radiography for the identification and localization of a foreign body or catheter may be the strongest indication for this imaging modality.
Abdominal Computed Tomography
In the United States the use of CT has increased dramatically in the past few decades, with more than 70 million CT scans performed in 2007.12,13 Although CT accounts for only 15% of radiological imaging, it accounts for approximately half of the collective medical radiation performed in the United States.14,15 The median effective radiation dose for abdomen and pelvis CT ranges from 15 to 31 millisieverts (mSv), in comparison with the chest radiograph, which is 0.1 mSv.16 In a study by Sodickson and colleagues,15 cumulative CT radiation exposure added incrementally to baseline cancer risk. Estimated lifetime cancer mortality risks attributable to the radiation exposure from a single abdominal CT in a 1-year-old is 0.18%.17 Brenner and Hall18 predicted that 1.5% to 2% of all United States population cancers may be caused by CT radiation exposure. In particular, patients with chronic or recurrent medical conditions have the greatest exposure risk.
There are two groups in whom the cost, delay, and risk of abdominal CT may outweigh the benefits.19 Those patients with (1) a high index of suspicion for the need for immediate surgical intervention (eg, unstable, obvious peritonitis) and (2) those in whom there is high confidence of a nonsurgical diagnosis based on history, physical examination, and/or other laboratory tests, yielding a low index of suspicion of serious abdominal pathology.20 For those patients that fall between these two groups, abdominal CT can be invaluable.
An abdominal CT can be performed with or without intravenous (IV) iodinated contrast medium, and with or without oral/rectal contrast. In a prospective study of 100 ED patients with abdominal pain, patients were initially scanned without oral contrast and then again 90 minutes after oral contrast with identical scanning parameters. The investigators concluded that oral contrast takes at least 90 minutes to adequately opacify the bowel, increases length of stay in the ED by almost double that amount of time, but adds little, if anything, to the accuracy of diagnosis in patients with nontraumatic abdominal pain.21 Similarly, in a study of noncontrast enhanced versus oral-contrast enhanced CTs, there was a 241-minute increase in time until disposition with the use of oral contrast.22
Summary
Ultrasonography
Abdominal ultrasound images are traditionally obtained by technicians, interpreted later by radiologists, and then results are reported to clinicians. With the advent of smaller, more portable, higher-resolution machines, US is currently being used directly by clinicians. For more than a decade emergency physicians (EPs) have been using bedside US, and the ACEP has formally endorsed and supports bedside US by EPs for multiple applications, research, and education.23 Emergency medicine residents are now required to receive training in point-of-care goal-directed US, and there are currently more than 50 ultrasound fellowships in the United States.
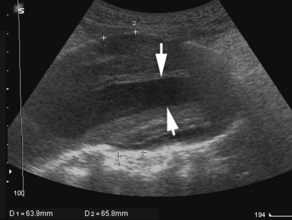
US has proved to be particularly useful in the early evaluation of the unstable patient. Appropriately trained nonradiologist clinician-performed bedside US can reliably detect intraperitoneal free fluid, the presence of an abdominal aortic aneurysm (Fig. 2), and ruptured ectopic pregnancy, and can assess the morphology and collapsibility of the inferior vena cava for volume status.24–27 In a study by Jones and colleagues,28 the investigators examined a physician-performed, goal-directed ultrasound protocol for the ED management of patients presenting with nontraumatic, symptomatic, undifferentiated hypotension. It was demonstrated that immediate goal-directed US in the evaluation of patients reduces the number of viable diagnostic possibilities and allows the physician to come to a more accurate final diagnosis.
Integration of laboratory tests and imaging studies based on location of pain and disease process
Evaluation of Generalized Abdominal Pain
Generalized abdominal pain can be secondary to infectious, mechanical, vascular, inflammatory, malignant, or traumatic processes ranging from benign to life-threatening in etiology. Some causes of generalized abdominal pain include bowel obstruction, abdominal aortic aneurysm (AAA), mesenteric ischemia, peritonitis, narcotic withdrawal, sickle cell crisis, irritable bowel syndrome, and heavy metal poisoning.29 A workup for generalized abdominal pain will depend on a multitude of clinical factors, including history and physical examination, age, comorbidities, and vital signs, to avoid missing a serious condition. Several studies have shown that CT has an advantage over other imaging modalities in patients with nontraumatic generalized abdominal pain, and can alter disposition decisions in about a quarter of patients.19,30,31
A study of 100 young women (age 15–45 years) who presented to the ED with a chief complaint of lower abdominal pain attempted to examine the effects of CBC result on clinical decision making.32 All patients in this study with appendicitis, as well as the one patient with an ectopic pregnancy, were appropriately diagnosed without the CBC result. In addition, of the 73 patients not admitted or diagnosed with an infectious disease, 17 were found to have an elevated white blood cell (WBC) count. While often ordered, the CBC in this study was found to have limited additional value in the workup of this patient population.
In the critically ill patient, lactate levels, although not specific to abdominal pain, can be a useful adjunct in the evaluation. In a prospective cohort study involving 1278 consecutive patients admitted to the hospital from an urban academic ED with a clinically significant infection (regardless of source), lactate levels were found to be useful as a risk stratification tool for inpatient mortality.2 Primary end point was 28-day in-hospital mortality with a secondary outcome of “early death” within 3 days of hospitalization. Patients were divided into 3 groups by lactate levels defined as low (<2.5), medium (2.5–3.99), and high (≥4). For the 3 groups, 28-day in-hospital mortality was 4.9%, 9.0%, and 28.4%, respectively, and early mortality was 1.5%, 4.5%, and 22.4%, respectively. Lactate levels greater than 4 were 55% specific and 91% sensitive for early death (positive likelihood ratio [LR+] 1.4, negative likelihood ratio [LR−] 0.22, authors’ calculation), and 36% specific and 92% sensitive for 28-day death (LR+ 2.02, LR− 0.16, authors’ calculation). The investigators surmise that lactate levels may be helpful in identifying patients who should be targeted for aggressive early therapy.
Mesenteric ischemia is one of the most feared diagnoses for an ED physician with its unfortunate triad of frequent lack of history, unreliable physical examination, and lethality. The identification of laboratory markers to improve the diagnosis has so far proven elusive; however, a few potential markers have been identified. In a systematic review of the literature, 20 articles were identified that investigated serologic markers for intestinal ischemia in a combined 978 patients.33 The prevalence of intestinal ischemia was 28% and included patients from a variety of settings. Of the multiple serologic markers investigated, 3 offered potential for improved diagnostic accuracy, including D-lactate, glutathione S-transferase (GST), and intestinal fatty acid binding protein (i-FABP). D-Lactate is produced by bacterial organisms and is thought to be a marker for bacterial translocation, as may follow ischemic (or other) mucosal injury. The investigators calculated a pooled positive LR of 2.64 in patients presenting with acute abdomen and a negative LR of 0.23. Physiologically, GST is a less specific marker, as it is released during oxidative stress from both the liver and the intestines; however, the analysis showed a positive LR of 3.28 and a negative LR of 0.23 in patients with acute abdomen. Intestinal fatty acid binding proteins are found in the cytoplasm of enterocytes at the tips of villi, the area most vulnerable to ischemia, thus making i-FABP an interesting candidate for a serologic marker of ischemia. In the analysis, the positive LR was 4.5 and the negative LR was 0.52. All of these markers require further research before becoming routine in the evaluation of ED patients presenting with acute abdominal pain.
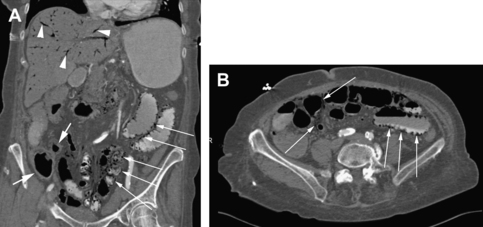
As clinical examination and laboratory tests tend to have limited ability to predict the presence of mesenteric ischemia, imaging tests are often needed. Angiography of the mesenteric arteries traditionally has been the gold-standard diagnostic test for mesenteric ischemia. This test is limited by its invasiveness, dye load, and inability to visualize surrounding structures other than the vessel lumen. With the advent of multidetector-row CT and MRI, angiography is no longer a first-choice procedure. CT has many advantages over angiography in that it is minimally invasive, can detect ischemic changes in the affected areas (bowel wall thickening, fat stranding, pneumatosis), and image reformatting can allow for angiographic reconstructions that often eliminate the need for traditional angiography in the case of high clinical suspicion and negative CT (Fig. 3).34 The limitations of CT include high radiation dose and risk of nephrotoxicity secondary to contrast dye. In a review article by Biolato and colleagues,35 they report that CT with IV contrast has a sensitivity and specificity for acute mesenteric ischemia of 64% and 92%, respectively. Multidetector-row CT with 3-dimensional reformats increases sensitivity and specificity to 96% and 94%, respectively. MR angiography can obtain high-resolution angiograms in 85% to 90% of superior mesenteric arteries, 75% to 90% of celiac arteries, and 25% of inferior mesenteric arteries (lower because of its anatomic location). The superior mesenteric and celiac arteries are clearly visualized using duplex US in more than 90% and 80% of cases, respectively. As with MR, US cannot reliably visualize the inferior mesenteric artery because of its anatomic location.35
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree