I. OVERVIEW. Patients being dialyzed with venous catheters do not do as well as those using an arteriovenous access. Catheter patients develop infections more often, they have higher levels of inflammatory markers such as C-reactive protein, and they die more frequently. It is unclear if these associative risks reflect a different patient population receiving catheters, some risk factor that occurs when AV accesses fail and a catheter must be placed, or if they are due completely to some property of catheter use per se. Probably all three are important. Survival rates for catheters are about 60% at 6 months and 40% at 1 year if revisions are included. Inadequate blood flow through venous catheters remains a significant problem. Nominal flow >400 mL/min (actual flow 350 mL/min) can rarely be obtained, and usually flow is limited to a range closer to 300 mL/min. This limits use of venous catheters in larger patients and results in a lower than average urea reduction ratio (URR) or fractional urea clearance (Kt/V).
In the chronic setting, venous catheters are utilized as a long-term vascular access for patients in whom an AV access cannot be readily created. Such patients include small children, some diabetic patients with severe vascular disease, patients who are morbidly obese, and patients who have undergone multiple AV access insertions and in whom additional sites for AV access insertion are not available. Additional indications include patients with cardiomyopathy unable to sustain adequate blood pressures or access flows. Whereas catheters were initially favored for more frequent dialysis, there has been good recent experience with nocturnal dialysis and short daily dialysis using AV fistulas or grafts. There has been renewed discussion about the potential acceptability of venous catheter access for chronic dialysis in some elderly patients, especially those with comorbidities and limited expected life span (Drew and Lok, 2014). Infection rates with venous catheters in elderly patients (>75 years) are relatively low, one-third that in younger patients (Murea, 2014). Adherence to hand washing and catheter-care protocols such as those suggested by the U.S. Centers for Disease control has resulted in marked overall reduction of dialysis catheter infection rates (Patel, 2013).
II. CATHETER TYPES AND DESIGN
A. Cuffed versus uncuffed. Use of an uncuffed catheter for periods of time beyond several weeks results in a relatively high rate of infection and is not recommended. Dacron or felt cuffs bonded to the catheter reduce the incidence of line-related infection and of catheter migration and must be used whenever a longer-term use of the catheter is anticipated, or when it is anticipated that a patient will be discharged from the hospital with a catheter remaining in place.
B. Design issues. Dual-lumen venous catheters are available in a “double-D” configuration or where the two lumens are in some related, side-by-side configuration. Coaxial catheters are now less frequently used. A side-by-side port design permits the intravenous portion of the catheter to be split into two parts close to the termination point. This results in a softer, more pliable catheter end, a greater separation of inlet and outlet ports, and perhaps a lower recirculation rate. The cuffed Tesio catheter system (used primarily for chronic dialysis) consists of two completely separate catheters, each made of soft silicone material, one for inflow and one for outflow.
C. Antiseptic impregnation. Some dialysis catheters or their cuffs are impregnated with antiseptic or silver-based coatings in an attempt to inhibit bacterial growth. At present there are no large studies that demonstrate improved outcomes with such catheters.
III. ACUTE DIALYSIS
A. Indications. Venous catheters are commonly used for acute angioaccess in the following patients: (a) those with acute renal failure; (b) those requiring hemodialysis or hemoperfusion for overdose or intoxication; (c) those with late-stage chronic kidney disease needing urgent hemodialysis but without available mature access; (d) those on maintenance hemodialysis who have lost effective use of their permanent access and require temporary access until permanent access function can be reestablished; (e) patients requiring plasmapheresis; (f) peritoneal dialysis patients whose abdomens are being rested prior to new peritoneal catheter placement (usually for severe peritonitis that required peritoneal dialysis catheter removal); and (g) transplant recipients needing temporary hemodialysis during severe rejection episodes. The renascent interest in urgent start peritoneal dialysis, discussed in Chapter 24, and earlier referral of patients with CKD for access placement should lower the need for urgent placement of central venous catheters for hemodialysis.
B. Insertion location. Available sites include the right and left internal jugular vein, the femoral veins, and the subclavian veins. One order of preference for these various sites is shown in Table 7.1. The optimal insertion site is the right internal jugular vein because the venous pathway to the right atrium is relatively short and straight. The subclavian site should generally be avoided because it is associated with a higher incidence of insertion-related complications (pneumothorax, hemothorax, subclavian artery perforation, brachial plexus injury) and a high incidence (up to 40%) of central venous stenosis. Use of the left internal jugular vein for acute dialysis is not optimal, as this represents a relatively long and twisty pathway to the right atrium; if subsequent chronic dialysis is needed, ideally one would like to leave the upper extremity central venous vessels alone to limit the rate of future stenosis. The femoral vein approach has several potential advantages. Placement tends to be simpler, especially for inexperienced operators. There is no risk of pneumothorax, hemothorax, or brachial plexus injury, although femoral arterial puncture and retroperitoneal bleed can occur. Originally, it was thought that the femoral approach had a higher risk of infection, but recent experience from the Cathedia Study Group found comparable rates of infection and time to catheter tip colonization (14 days) with femoral and internal jugular catheters (Dugué, 2012). The femoral approach is useful for performing the initial hemodialysis treatment in patients who present with acute pulmonary edema because the patient’s head and chest can remain elevated during insertion. The infection risk with femoral catheters is increased in obese patients (BMI > 28 kg/m2), although the extent of this risk probably depends on the distribution of body fat. When femoral catheters are used, the length must be sufficient (usually at least 20 cm) so that the tip is in the inferior vena cava to permit better flow and to minimize recirculation. Another finding from the Cathedia Study was that delivered URR and Kt/V were similar with femoral and jugular catheters (Dugué, 2012). The European Best Practices Group does not agree with the order of preference of insertion sites listed in Table 7.1, and gives second preference to the left internal jugular vein, and recommends that femoral catheters be discouraged (Vanholder, 2010).
Selected Factors Favoring Different Temporary (Nontunneled) Hemodialysis Catheter Insertion Sites | |
1. Right internal jugular
Critically ill and bed-bound with body mass index >28
Postoperative aortic aneurysm repair
Ambulatory patient/mobility required for rehabilitation
2. Femoral
Critically ill and bed-bound with body mass index <24
Tracheostomy present or planned in near-term
Need for long-term hemodialysis access present, highly likely or planned
Emergency dialysis required plus inexperienced operator and/or no access to ultrasound
3. Left internal jugular
Contraindications to right internal jugular and femoral sites
4. Subclavian
Contraindications to internal jugular
Right side to be used preferentially
Source: Reprinted by permission from Macmillan Publishers Ltd: Clark EG, Barsuk JH. Temporary hemodialysis catheters: recent advances. Kidney Int. 2014. doi:10.1038/ki.2014.162.
C. Uncuffed versus cuffed catheter use. The risk of infection of uncuffed catheters increases markedly after the first week. For this reason, the KDOQI 2006 vascular access guidelines recommend use of a cuffed catheter if the anticipated need for dialysis is longer than 1 week. They also recommend that femoral catheters in bed-bound patients not be left in place longer than 5 days. These recommendations, especially with regard to femoral catheters, may be a bit too stringent given the results of the Cathedia Study (Dugué, 2012), where median time to catheter tip colonization was 14 days. Once the likelihood of the need for prolonged dialysis is established, an uncuffed internal jugular catheter can be replaced with a cuffed catheter. In cases where a prolonged need for dialysis is likely at the outset, a cuffed catheter can be inserted initially, in the right internal jugular vein position if possible. Recently some success has been claimed using cuffed tunneled femoral catheters (Hingwala, 2014). This has the advantage of locating the exit site away from overhanging skin folds, and easy removal, as long as removal is done within several weeks of insertion. Placing a cuffed femoral catheter allows time for more definitive access site placement, whether it be for peritoneal dialysis or hemodialysis.
D. Anatomic variation and use of real-time ultrasound guidance. The central veins of the neck exhibit anatomic variability (Fig. 7.1), and occasionally one of them may be absent. Atypical or ectatic carotid arteries are also a problem. With the use of ultrasound guidance, the rate of successful internal jugular puncture on first attempt increases markedly and the rate of carotid artery punctures and hematoma is greatly reduced (Rabindranath, 2011). In the femoral approach, the femoral vein often is behind the artery, and this overlap worsens as one proceeds down from the inguinal ligament (Beaudoin, 2011). Here, too, the use of ultrasound helps reduce complications (Clark and Barsuk, 2014).
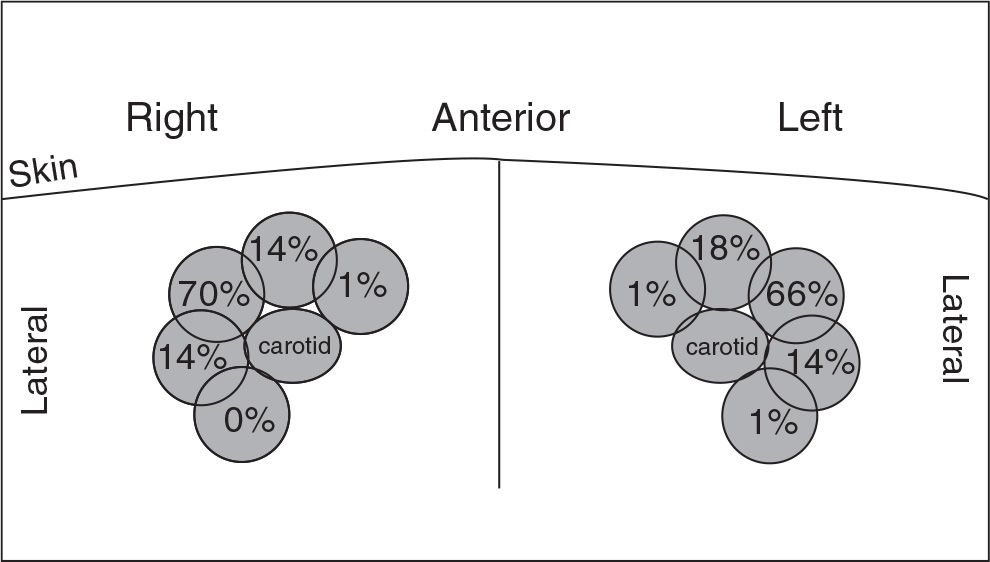
FIGURE 7.1 Anatomic variability of internal jugular vein as viewed using ultrasound localization. (Modified from Caridi JG, et al. Sonographic guidance when using the right internal jugular vein for central vein access. Am J Roentgenol. 1998;171:1259–1263.)
E. Simulation-based training for catheter insertion. Venous catheter insertion for dialysis is a necessary skill for nephrology fellows to acquire, but many programs may not have resources to provide the required level of training. Simulation-based training has been proposed to remedy this, and provision of such intensive training has resulted in improved catheter-related outcomes (Clark and Barsuk, 2014).
IV. INSERTION TECHNIQUE
A. Initial site preparation. The catheter should be inserted using an aseptic technique, with the operator wearing a sterile surgical gown and gloves in a maximum barrier protection environment. Prior to surgical scrub, it is helpful to examine the selected site using ultrasound to ensure that the patient has a suitable vein in the selected location. The insertion site and surrounding areas should be cleansed with surgical scrub and draped appropriately (include shoulder and chest wall if a cuffed tunneled catheter is to be inserted). The ultrasound probe should be covered with a sterile sheath.
B. Internal jugular approach. The ultrasound probe may be placed parallel to the long axis of the vessel and the cannulation needle inserted adjacent to the end or short axis of the probe. Alternatively, the probe may be placed perpendicular to the long axis of the vessel. This approach gives the vein the more typical appearance of a circle but limits the visualization of the needle. The vein typically collapses with gentle pressure of the probe, whereas the artery does not. Additionally, the vein diameter increases with Valsalva maneuver and can be easily observed with ultrasound. For internal jugular vein cannulation as an example, the ultrasound probe is placed parallel and superior to the clavicle, over the groove between the sternal and clavicular heads of the sternocleidomastoid muscle. It is important to avoid inserting the catheter through the muscle, as this is uncomfortable for the patient and leads to catheter dysfunction as the neck is rotated.
1. Initial insertion of the guidewire through a 21G needle. The site for insertion is infiltrated with local anesthesia. Using real-time ultrasound guidance, a 21G micropuncture needle with an attached syringe is inserted into the vein. The small needle limits potential complications if the carotid artery is inadvertently punctured compared to a larger 18G needle, which is usually included in commercially available dialysis catheter trays. Under direct visualization, the vein will be seen to gently push in before penetration of the anterior vein wall. The syringe is removed, and a 0.018″ guidewire is inserted through the needle. The guidewire is advanced. The position of the guidewire is confirmed using fluoroscopy.
2. Insertion of the dilator over the guidewire. The needle is then removed and a coaxial 5-French dilator is then advanced over the guidewire. The guidewire and 3-French inner translational dilator are removed, leaving the 5-French outer dilator in place. A flow switch or stopcock is attached to the dilator to prevent the possibility of an air embolism.
3. Uncuffed catheter insertion. The next step depends on whether one is placing a noncuffed temporary or cuffed tunneled catheter. For temporary catheter placement, a standard 0.035″ guidewire is advanced into the vein and then the 5-French dilator is removed, leaving the guidewire. In stepwise fashion, dilators of increasing size are passed over the guidewire to progressively dilate the soft tissue and venous tract; the dilator should move freely on the guidewire. The dilator should not be forcefully advanced, as it is possible for the dilator to get off axis and impinge on the guidewire and perforate the vein and/or the mediastinum. Consequently, one does not need to advance the entire length of the dilator as only the dilatation of the track from the skin to the vein is desired. If there is any doubt as to location of the dilator or if there is hesitancy or difficulty in dilating the tract, fluoroscopy should be used to assist in placement. The last dilator is then exchanged for the temporary catheter, which is advanced over the guidewire into position. After securing the catheter in place, a chest radiograph should be obtained for confirmation of correct positioning and to check for any complications, if a fluoroscope was not available during insertion. If the patient requires long-term dialysis support, the temporary noncuffed catheter, when located in the internal jugular vein, may be safely converted to a cuffed tunneled catheter if there is no evidence of an exit-site infection.
4. Cuffed catheter insertion
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





