Introduction
“From inability to let well alone, from too much zeal for the new and contempt for what is old, from putting knowledge before wisdom, science before art and cleverness before common sense, from treating patients as cases and from making the cure of the disease more grievous than the endurance of the same, good Lord deliver us.” ( ) compiled by Deborah Cassidi in the BMJ in 1998.
Dr Hutchison’s words are prescient as healthcare continues to evolve, and it is worth remembering as we discuss telediagnosis. Telediagnosis is a portmanteau of ‘tele’, derived from the Greek word telos meaning distance, and the English word ‘diagnosis’ meaning just that. (Diagnosis is of course derived from the Greek words dia meaning “apart” and gignōskein meaning “to know” or “recognize”). Understanding the origin of the terms is indeed appropriate as it provides a framework for what telecommunication must offer, so that a physician can first recognize good health, from disease, and then crucially, discern one pathology from another.
Home-based diagnostics can at present provide targeted services for the care of some urology patients. Advances in technology have been such though, that the only impediment to the practice of entirely remote diagnosis, is a combination of innovators executing what is now entirely possible, and regulators trying to keep pace with the explosion that has occurred, and bracing for the supernova that is surely going to ensue in the area of tele-diagnosis. We find ourselves in a “Wild West” of the World Wide Web, where even legitimate providers of such tools with the best of intentions, have little guidance on what is necessary to adequately protect end users.
Ideal characteristics of medical conditions amenable to telediagnosis
There are many existing tools which will be profiled in this chapter, but the field is ever expanding. In general, the characteristics of diagnostics with the lowest barriers to entry, in other words, those that are most amenable for rapid if not immediate implementation using current telehealth networks are:
Those that:
- •
Are amenable to minimally invasive measurement (use sound, vision, or other digital sensors, for external rather than internal and invasive measurement or characterization).
- •
Require minimal hardware or minimal additional hardware (for example, just use the patient’s existing smart phone, or their existing webcam, speakers and microphone, smartwatch, or sleep tracker).
- •
Do not restrict the patient’s life—are already used every day or already exist in the patient’s environment—(don’t need to install a piece of hardware to their house that occupies physical space).
- •
Can utilize existing infrastructure that can be secured over existing Wi-Fi, Bluetooth, wired networks, or other telecommunication channels. (Something that isn’t going to be interfered with by another nearby device that could affect the reliability of its performance in measuring biomedical characteristics) ( ).
At the time of writing this book chapter, the majority of telehealth services, particularly for urological conditions, do not consider the patient as a whole, and in reality, appear to be little more than e-prescribing services, proclaiming to be democratizing healthcare. To the companies who have the legal minimum medical oversight, and employ nurse practitioners or physician extenders to complete screening questionnaires before providing a prescription for medication with little follow-up or patient monitoring, I would assert that the “healthcare” that they profess to be “democratizing” should go the way of the French monarchy circa 1789.
Making a diagnosis is fundamental to the practice of clinical medicine; this is true regardless of whether this practice occurs in-person or via telemedicine. While the practice is complex, some of the core tools of a clinician are rapport building and being able to do so in a short period of time, with a wide range of different individuals, establish the clinical features by history and examination, and interpreting elicited information according to the scientific method ( ). Arriving at a diagnosis is not as simple as completing an IPSS or IIEF survey, answering a checklist of questions and calculating a score. The aims of medicine should not just be to diagnose accurately, but holistically. One must formulate an appropriate sequence of investigations, begin appropriate treatment, assess its effectiveness, give an informed prognosis, and make follow-up arrangements. Physicians are not entirely responsible for being seduced by the alures of productified health, as market forces are as real in medicine as they are in commerce. But the medical profession should not become commerce. And while patients many be “consumers” of healthcare, they should not be considered as customers. Many consumers of healthcare have become accustomed to on-demand access, to have their whims satisfied, and I would argue this is a misstep for an effective healthcare system.
The foundations of e-health experience is being conceptualized, designed, and constructed right now. I’d advocate against e-health and telehealth following the path of e-commerce, which seems to be the prevalent model currently in use, where much of the e-health experience is akin to the checkout aisle in grocery stores:
- •
Much advertising is targeted, and predominant platforms for this are search and social media sites. While this is a powerful tool and capable of much good, these very tools have recently been subverted and used for malicious means to manipulate and even indoctrinate; something which is entirely unacceptable and particularly so when vulnerable groups are being targeted.
- •
The current e-commerce experience also makes use of prompts to encourage impulse decisions. It has been documented that people make more careful and accurate decisions in the morning time, and make more hasty and inaccurate decisions later in the day ( ). The findings of this study are particularly illuminating when cross-referenced with the peak times for e-commerce transactions—between 8 and 9 p.m. at night ( ).
The concern is the “productifying” of healthcare into consumer products. Repurposing the World Health Organization’s International Classification of Diseases into a catalog of consumer services to be sold is not useful. While it can be dressed up and presented as noble and worthy, and sold under the guise of improving access, this is clearly a wolf dressed in sheep’s clothing.
While some see telehealth as the climate change of medicine (and while in the practice of medicine, such a step change may be welcomed with open arms by many), the value of the patient–physician relationship, with clinical history and clinical examination as core elements, should not be eroded. Clinical history is essential in all cases to generate a logical differential diagnosis and to guide rational investigation and treatment. While in many developed nations, there has already been a steady move away from elements of the clinical exam, as some physical signs pathognomonic of conditions are becoming rarer, and some aspects of clinical examination are being marginalized as novel biomarkers emerge. Clinical examination too is critical to the diagnostic process—recognizing the ill patient and what ills the patient: eliciting signs that cannot be identified with tests and identifying unsuspected problems.
With this in mind, we can approach each of the usual components of the diagnostic process outside of clinical history, to identify what “teletools” are already available to the urologist, and what is possible for teleurology in the early 21st century. The first classification which can be made is whether the diagnostic criteria are subjective or objective ( Table 7.1 ).
| Diagnostic tool is: | Examples |
|---|---|
| Subjective | Questionnaires |
| Diaries | |
| Objective | Clinical specimen → blood, urine, stones, swabs, sweat, saliva |
| Electrophysiological parameter → flow rates, urodynamics | |
| Imaging → ultrasound → advanced imaging | |
| Procedures → cystoscopy |
Symptom diaries, questionnaires, and recording subjective assessments
Symptelligence Medical Informatics ( symptelligence.com ) have created the “weShare URO” app, a platform for patients with lower urinary tract symptoms (LUTSs). It provides a mobile application where patients can complete validated LUTSs questionnaires and bladder diaries. The platform was examined by some of the cofounders in a 2021 publication following a pilot study of 500 urology referrals, where 45% had among other issues, new LUTSs. The study outlined that the majority of patients were men (M:F = 182:19), and the mean age of patients was 59 years. The mean time from urology referral to initiation of remote or in-office diagnostic and treatment plan, was 7.4 and 7.7 days respectively. This study provides many valuable insights for the field of teleurology: that design and workflow should account for the higher average age of patients who present with LUTSs.
Another application was developed by a research group focusing on pediatric patients. In their prospective study from 2014, they examined a mobile voiding diary for children with voiding dysfunction and compared it with a paper voiding diary. In this study also from the north-east United States, a greater proportion of diaries were completed and had less data gaps in the paper diary than the mobile diary ( ) (see Fig. 7.1 ).
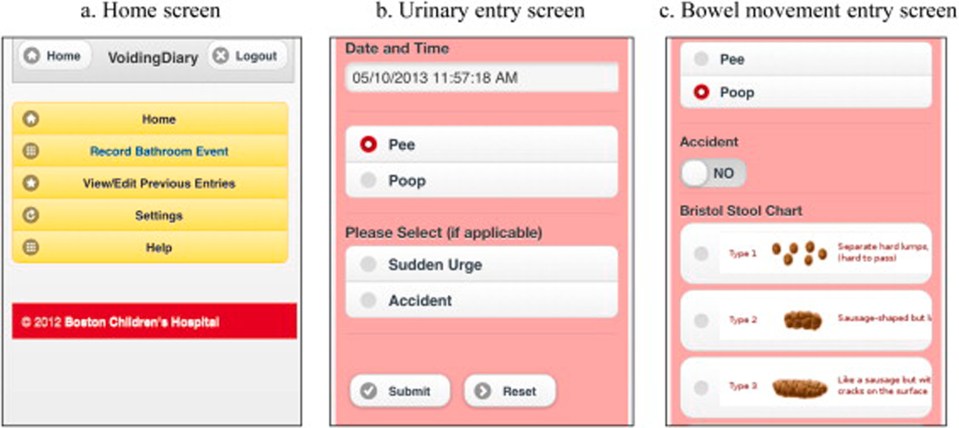
This study is referenced because it is important for a number of reasons: its findings do not suggest superiority of the digital record used over paper-based records. This study was performed at a Children’s Hospital in Boston, and families were asked to fill out the voiding diary and excluded those who did not speak English, and those who did not have internet access. The design was also a pre-, post- design, where families had already completed the paper diary, and had already been seen at clinic, and then were asked to repeat the process but this time using a mobile web app of the same questions. This study does not truly assess mobile completion of subjective symptom questionnaires compared to paper versions, and would likely not even be a subjective measure at all, as the parents of children attending the clinic were most likely those repeating the same process, only just doing so on a mobile application the second time.
Blood tests
It is of course possible and acceptable for many to continue to use existing services, where they receive a prescription, or a request is sent to a laboratory testing facility to collect and process specific blood tests. There is no reason why this would not still be useful and particularly for tests, for which identity verification would be wise. Some individuals may try to circumvent a diagnostic process to have medications prescribed inappropriately—such as is reputed to happen with individuals purchasing urine samples from hypogonadic men, only to submit them as their own and to receive testosterone supplements. It is also the case that a significant amount of testosterone supplementation is made without checking a baseline testosterone level ( ).
This section will also not focus on “point-of-care” (POC) tests (such as Opko’s Sangia finger stick prostate-specific antigen [PSA] test), that require the use of specific analyzers and the associated capital outlay. Such analyzers may be in use in a particular clinic, but they are often not practical for “point-of-need” testing, as may be preferred in a truly remote diagnostic setting.
Kits sent by mail
Numerous providers such as letsgetchecked.ie , everlywell.com and www.imaware.com remove the need to attend a testing center. Patients can test themselves at home or wherever they may be. The kits generally need a small volume of blood- ∼2–5 drops of blood. For these tests that are currently provided, there are no temperature controls required for the blood sample. The general workflow is:
- 1.
order a blood test kit for delivery
- 2.
collect the sample at home with a finger prick test kit
- 3.
send the sample back (in a prepaid envelope or via a collection service)
- 4.
results are processed in a Clinical Laboratory Improvement Amendments (CLIA)–certified/central laboratory
- 5.
Results reviewed and made available online via a web portal
Lateral flow tests (LFT) for immediate results
Lateral flow immunoassays are a very successful analytical platform to perform rapid on-site detection of target substances. The platform is known by a variety of terms including LFT, immunochromatographic strip test, or rapid diagnostic test. Many people have been exposed to this technology in the form of rapid lateral flow tests for COVID-19, which have become widely available.
It can be considered as a “lab in your hand” for that specific detection of whichever target it has been designed for, and provides a rapid result at the “point of need.” And the technology has been applied to a variety of other common scenarios, which can be utilized in the remote care of patients. Their “simple by design” property means no equipment or technical expertise is required; they are very useful in a variety of settings including low-resource environments.
PSA kits
It has been almost two decades since groups proposed, developed, and tested lateral flow tests for PSA—such as the “RapidScreen” test from the University of Rome “Tor Vergata,” which generated a positive result for PSA values ≥4 ng/mL ( ). However, at the time of writing, the Food and Drug Administration (FDA) in the United States of America has not yet approved a lateral flow test/lateral flow immunoassay for the detection of PSA for general use.
While such assays with a binary cutoff will not suit every purpose, it is a useful addition to a practice where an initial screening test may be required, so that patients can be reassured while they are still remote from the urology office, and the test is performed in a rapid fashion. The RapidScreen test employs a cutoff of 4 ng/mL, but other kits were developed with alternative cutoff values; in theory, these can be placed at any value above the lower threshold of detection limit, which for some assays is ∼1 ng/mL ( ) when using silica-coated CdSeZnS quantum dots and as low as 0.5 ng/mL for total PSA using an AuNSh-labeled test strip ( ).
These tests demonstrate many of the qualities which would make them fit for a limited purpose like initial screening to exclude one sinister cause of lower urinary tract dysfunction. The costs of such tests are estimated to be circa ∼USD$1 as of the time of writing and could form a useful part of the teleurologists toolkit. While an entirely quantitative high sensitivity test for point-of-need testing is being developed, the initial barrier to the adoption of these LFTs is completing preparatory work for submission of these devices and approval by regulatory bodies such as the FDA and European Medicines Agency (EMA).
Testosterone
Third-party private companies also offer kits including analysis of free testosterone and dehydroepiandrosterone from saliva samples—such as EverlyWell.com , in their “Men’s Health Test,” which also includes analysis of cortisol and estradiol levels. However, these are not point-of-need tests and require the kit to be sent back to a centralized lab, where EverlyWell.com report that results will be made available “within days” of returning your kit to them ( https://www.everlywell.com/products/mens-health-test ).
Other providers in this space include Cerascreen (results take 3–5 days, Testosterone Test ∼USD $59), Everywell for $49 LetsGetChecked, myLAB Box (multiple tests including T for $259, results within five days), Verisana ($49 for T test alone), and ZRT Labs. (Pricing as of June 2022).
Similarly, lateral flow immunoassays can be created to test testosterone levels. Similar limitations exist, in that these tests can either be binary—for example, is total testosterone above 300 ng/dL? Or is testosterone greater than 50 ng/dL? Or they can be semiquantitative, in that they can be used in parallel or serial to determine levels <50 ng/dL, between 50 ng/dL and 300 ng/dL, or >300 ng/dL for instance ( ).
Creatinine and eGFR estimation
POC creatinine devices allow rapid measurement of creatinine levels and calculation of estimated glomerular filtration rate (eGFR). This can show whether the kidneys are working properly. This is of particular importance in not just routine monitoring of renal function but also in circumstances where patients may be referred for imaging that also includes contrast, or to assess for an acute or chronic kidney injury in patients with LUTS.
Approved devices include the i-STAT Alinity from Abbott, which is a handheld device that can measure multiple parameters from 65 mL of blood and provide a measurement within 2 min and the Nova Biomedical Stat Sensor, another handheld device that measures creatinine from 1.2 mL of sample within 30 seconds. However, true point-of-need testing devices are not approved or available at the time of writing.
Any blood test
According to the FDA 510k database of over-the-counter (OTC) tests, there are currently no FDA-approved, point-of-need tests for other commonly requested lab values such as hemoglobin, C-reactive protein, or white blood cell count. In theory, these assays are entirely possible; however, as of yet they have not been developed. There are numerous research papers characterizing lateral flow assays to detect thrombin, troponin I, HBsAg, and human immunodeficiency virus (HIV) ( ; ). There are numerous providers who offer mail in kits providing lab results in a number of days which have already been discussed earlier in this chapter.
STI checks
Similar to the services described above, there are several mail-in services, who test for sexually transmitted infections such as chlamydia, gonorrhea, HIV, and herpes. These include Everlywell, LetsGetChecked, and QuestDirect.
Urine
Dipstick urinalysis/apps
In the 1950s, single glucose urinalysis was made possible using a paper-based dipstick through enzymatic oxidation of chromogen, followed by commercial introduction in the 1960s. Multiple manufacturers offer urinalysis kits for sale, including over the counter (OTC) in many pharmacies at prices circa $15 USD for 100 strips, albeit sometimes with less information than is typically available from the test strips commonly used in hospital settings. The main reason for this appears to be unit costs balanced with the tests that the majority of consumers are using the strip for.
There are now also a number of FDA-approved urinalysis kits that pair with a mobile app for interpretation and record keeping, such as Healthy.io who call their urinalysis kit application “Dip.io.” It was reported to be the first approval of such a test that uses optical equipment designed by a third party (aka your smartphone manufacturer). It examines 10 parameters and can be used for a multitude of purposes from detecting urinary tract infections, chronic kidney disease, and prenatal checks. The parameters tested are: leukocytes (negative or 15/70/125/500 leukocytes/μL), nitrates (negative or positive), glucose (negative or 100/250/500/1000 mg/dL), ketones (negative or 5/15/40/80 mg/dL), protein (negative or 15/30/100/300 mg/dL), blood (negative or 10/25/80/200 erythrocytes/μL), pH (5.0, 5.5, 6.0, 6.5, 7.0, 7.5, 8.0, 8.5, or 9.0), urobilinogen (negative or 1/2/4/8 mg/dL), bilirubin (negative or 1/2/4 mg/dL), and specific gravity (1.000, 1.005, 1.010, 1.015, 1.020, 1.025, or 1.030). The company seems to have made a strategic change, and in early July 2022 received FDA approval for Minuteful Kidney, which is their focused urine dipstick to assess urine albumin to creatinine ratio, and they are awaiting FDA approval of a combined mobile health app and dipstick test focused on diagnosing and monitoring urinary tract infections, and another kit specifically focused on prenatal care. The company also has reported ambitions to utilize smartphones more fully and incorporate spectroscopy including analysis of infrared and ultraviolet light, which could extend utility to infection control.
There are other providers that couple a urine dipstick with a smartphone app, who are not focused on any one particular medical condition, but may be useful for kidney stone patients owing to the composition of their dipstick test. For example, Vivoo ( https://vivoo.io/ ) market their at-home urinalysis strip and mobile app to provide personalized nutrition and lifestyle advice. It should be noted that this is not an FDA or EMA-approved test. It does provide results for the presence of calcium, magnesium, pH, specific gravity (which it labels as hydration) as well as vitamin C, salinity, “oxidative stress,” ketones, and protein. The product is sold as a subscription service. Again, it is important to note that this is not an approved test, and its mention here is not an endorsement, but rather as evidence of feasibility, and that measures of interest to urologists, and relevant to the care of urological patients, can be detected and quantified to some degree, with noninvasive tests that are suitable for mobile health applications, and for use by patients in their own home.
The use of urine as a noninvasive test medium to monitor health and detect disease is primed for further development, and is an area where urologists are well placed to lead, and indeed incorporate its use into daily practice. Urine is a relevant analyte not just for urological conditions, benign or malignant, but for many others, as urine contains a wide range of biomarkers already in everyday use. A group in Korea has recently described their efforts to further develop a “lab in a cup,” incorporating the conventional paper-based urine dipstick, already placed in a disposable cup, which is coupled with a smartphone application that automates the analysis procedure. Still in the development stage, the investigators successfully applied their design and application to the detection of glucose, pH, protein, and red blood cells ( ).
Flow rates/basic urodynamics
The current standard practice of uroflowmetry in urology is equipment, clinic staff, and clinic space-intensive. It requires specialized hardware, consumes time of healthcare workers in preparing the equipment, the space and cleaning each of these afterward, as well as requiring a dedicated space for the procedure in the clinic. A number of researchers have proposed solutions, and MenHealth have received FDA clearance for their MyUroFlow application to address all of the above issues. The application uses the array microphones on the patient’s smartphone, android or iOS. To perform a uroflow, the user opens the application on their phone while next to the toilet, presses start and allows it to calibrate for background ambient noise, then urinates as they normally would. The application provides a peak flow rate, voiding volume, and voiding time, and automatically compiles a voiding diary. The application provides a cloud-based dashboard and allows patients to share their results with their urologist. MenHealth ( https://myuroflow.com ) currently price the application at $15 per month for patients who want to track their flow rates themselves, and provide health practitioners the ability to provide accounts to their patients with provider plans, with per patient prices of $2.50–$5 per month, depending on the number of patients the provider aims to provide the service to.
Soundable Health have developed and released the proudP app for Apple iPhones, but it is not currently approved by regulators in the European Union (EU) or United States of America ( https://www.soundable.health ). The application employs a similar approach and provides max flow rates and voided volumes to patients and providers via an online dashboard. There are also other applications that integrate with hardware devices which must be provided to or purchased by patients, such as iCarePath, which is discussed in more detail in the next chapter.
Diagnostic imaging
Ultrasound and PVR
A number of well-known healthcare device manufacturers have miniature probes, which can be linked to a smartphone or tablet device to provide a portable ultrasound solution. In 2011, Mobisante was the first smartphone-powered ultrasound device to receive FDA approval for its MobiUS SP1 Ultrasound system. Clarius have a range of wireless miniature scanners for use by healthcare professionals (HCPs), which would be of utility to urologists who perform ultrasound as part of their practice. GE Healthcare recently added the Vscan Air to their product catalog. The Vscan Air also connects wirelessly to your smartphone or tablet and received FDA clearance in November 2020. All of these devices require an in-person visit and operation by a trained professional. They do provide the proof of concept that the hardware required to perform ultrasound can be miniaturized and connected to almost ubiquitous communication devices like smartphones and tablets, and pave a path toward low-cost devices, which could be used by patients to perform basic assessments, such as measurement of postvoid residual volume, or even testicular ultrasound and basic kidney imaging.
Advanced imaging
Most advanced imaging is available at standalone imaging centers, which in parts of the United States of America and EU have become not infrequent additions to suburban shopping malls and health centers. They provide an option for medical practitioners to refer patients to often less crowded and more conveniently located diagnostic services, and can be easily integrated into a telehealth practice workflow. Of particular interest to some is the possibility to have your preferred imaging protocol used by the imaging center, so that if you aim to have the images read by a radiologist separate from that imaging center, or compared with prior imaging, that identical acquisition protocols can be used and compared.
Conclusions
Telemedicine offers much promise, but as with most things, it is how it is executed that will ultimately matter. This platform and infrastructure afford us a relatively rare opportunity to redesign the systems that so often frustrate us, and so often frustrate patients. I do not know of an urologist or any physician who has not been frustrated by the steps, and hoops and ladders, and often seemingly unnecessary measures required to be taken, in order to complete, what at the surface, appear to be relatively simple tasks. For many, it is a simple process to help a patient from the waiting room to have their weight, height, and vitals measured, get a specimen container, and provide them the time and facilities to provide a urine specimen, have a urinalysis completed, flow rate estimation, and postvoid residual scan performed. But more often, this process can involve an administrative staff member to check the patient in, verify their identification, enter their insurance information if they didn’t leave it at home, and complete compliance paperwork to process their health information. Then asking the patient to wait for a HCP to help perform measurements, vitals, and clinical specimens, getting administrative assistance in printing and labeling specimens making sure not to confuse them with those of another patient, ensuring sufficient space and privacy for the patient to go to the bathroom, and that they actually have a sufficiently full bladder to perform a useful test at that very moment when the precious space and time constrained HCP is available. And all of that is before they ever actually get as far as seeing the doctor they came to see. I think a simple survey of physicians in practice asking: ‘have you ever had a patient respond to a name that wasn’t theirs?’, would provide sufficient evidence to demonstrate how the current system could be refined. If we were to ask clinicians, have you ever seen a patient who had been booked for the wrong investigation or even procedure, the answers would provide sufficient proof that the current systems in use are deficient and unfit for purpose.
That is not to say there is a carte blanche for physicians to institute whatever they want. While there are government regulations currently in place, many of them were not designed for a telehealthcare system. New regulations will be required, some existing regulations will be made redundant, and others will need to be revised. It is imperative though that all stakeholders meaningfully engage with this process. Physicians have traditionally allowed this process to occur in their absence; they must now ensure they are front and center to design the system that best serves all patients. This is an opportunity to design a healthcare system, knowing the strengths, limitations, opportunities, and vulnerabilities of multiple systems, which operated in a physical space over centuries. We can now build one in a metaphysical space, with all the knowledge gained from the real world, and are the best placed group to help do this.
The Health Insurance Portability and Accountability Act (HIPAA) is the perfect example of a law requiring thorough revision in an age of telemedicine. HIPAA is widely known as the piece of legislation in the United States of America that protects patient’s privacy rights, as it applies to how healthcare providers, health insurers, and clearinghouses handle some patient data. What is critical to clarify though is that HIPAA only applies to those specific entities that also receive federal funds—for example, by processing payments for patients using Medicare and Medicaid. It does not apply to entirely private practices. HIPAA also does not apply to thousands of apps, digital devices, fitness devices, or wellness apps, which record our behavior, track our activity, or measure our vital signs and biometric data. While HIPAA does not allow health plans or healthcare providers to sell personal, identifiable health data, it does not regulate what other entities do with these data. Mobile devices, wearable health trackers, and connected home devices are now as commonplace in the lives of Americans as a toaster, and the exploitation of such data has been well documented.
Bibliography
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree






