Endoscopic drainage is the first-line therapy in the management of pancreatic pseudocysts. Before endoscopic drainage, clinicians should exclude the presence of pancreatic cystic neoplasms and avoid drainage of immature peripancreatic fluid collections or pseudoaneurysms. The indication for endoscopic drainage is not dependent on absolute cyst size alone, but on the presence of attributable signs or symptoms. Endoscopic management should be performed as part of a multidisciplinary approach in close cooperation with surgeons and interventional radiologists. Drainage may be performed either via a transpapillary approach or a transmural approach; additionally, endoscopic necrosectomy may be performed for patients with walled-off necrosis.
Key points
- •
Endoscopic management of pseudocysts and walled-off necrosis should be performed as part of a multidisciplinary approach.
- •
Exclusion of pancreatic cystic neoplasms and pseudoaneurysms is critical before endoscopic drainage procedures.
- •
Indications for endoscopic drainage of pseudocysts include signs or symptoms attributable to the lesion, as opposed to large cyst size alone.
- •
The use of covered metal and lumen-apposing metal stents may improve outcomes compared with plastic stents for transmural drainage.
- •
New dedicated drainage devices and stents have been developed that may facilitate transmural drainage and necrosectomy.
Introduction
Pseudocysts complicate approximately 10% to 26% of acute pancreatitis and 20% to 40% of chronic pancreatitis cases. The 2012 Revised Atlanta Classification of Acute Pancreatitis defined pancreatic pseudocysts as well-circumscribed, completely encapsulated fluid collections more than 4 weeks old; surrounded by a nonepithelial wall of fibrous or granulation tissue; homogeneous and without a nonliquid component; and arising as a consequence of acute pancreatitis, chronic pancreatitis, or pancreatic trauma with pancreatic ductal disruption. Pseudocysts are a distinct entity from walled-off necrosis, which is a mature, well-circumscribed, completely encapsulated collection of pancreatic or peripancreatic necrosis that occurs more than 4 weeks after the onset of necrotizing pancreatitis.
Endoscopic drainage has emerged as the first-line therapy in the management of pancreatic pseudocysts as well as walled-off necrosis, with significant advantages compared with surgical and percutaneous drainage. Endoscopic pseudocyst drainage was first described by Sahel and colleagues in the late 1980s, using diathermic transmural access into the pseudocyst followed by placement of a nasocystic tube for irrigation and drainage. The endoscopic management of pancreatic fluid collections has since evolved significantly with the introduction of endosonographic guidance, new catheter delivery systems, and indwelling stents, including novel lumen-apposing covered self-expanding metal stents (LAMS). This article discusses the endoscopic management of pancreatic pseudocysts and walled-off necrosis, including indications for drainage, endoscopic techniques, efficacy, and comparison with percutaneous and surgical drainage.
Introduction
Pseudocysts complicate approximately 10% to 26% of acute pancreatitis and 20% to 40% of chronic pancreatitis cases. The 2012 Revised Atlanta Classification of Acute Pancreatitis defined pancreatic pseudocysts as well-circumscribed, completely encapsulated fluid collections more than 4 weeks old; surrounded by a nonepithelial wall of fibrous or granulation tissue; homogeneous and without a nonliquid component; and arising as a consequence of acute pancreatitis, chronic pancreatitis, or pancreatic trauma with pancreatic ductal disruption. Pseudocysts are a distinct entity from walled-off necrosis, which is a mature, well-circumscribed, completely encapsulated collection of pancreatic or peripancreatic necrosis that occurs more than 4 weeks after the onset of necrotizing pancreatitis.
Endoscopic drainage has emerged as the first-line therapy in the management of pancreatic pseudocysts as well as walled-off necrosis, with significant advantages compared with surgical and percutaneous drainage. Endoscopic pseudocyst drainage was first described by Sahel and colleagues in the late 1980s, using diathermic transmural access into the pseudocyst followed by placement of a nasocystic tube for irrigation and drainage. The endoscopic management of pancreatic fluid collections has since evolved significantly with the introduction of endosonographic guidance, new catheter delivery systems, and indwelling stents, including novel lumen-apposing covered self-expanding metal stents (LAMS). This article discusses the endoscopic management of pancreatic pseudocysts and walled-off necrosis, including indications for drainage, endoscopic techniques, efficacy, and comparison with percutaneous and surgical drainage.
Indications for drainage
In general, the indications for pancreatic pseudocyst drainage include persistent pain attributable to the fluid collection, gastric or duodenal obstruction, biliary obstruction, development of pancreatic ascites or pleural effusion, enlarging size on serial imaging, and signs of pseudocyst infection or bleeding ( Box 1 ). Pancreatic pseudocysts should not be drained in the absence of suspected infection if the fluid collection is not mature (ie, less than 4–6 weeks old), or the diagnosis remains in question.
Persistent abdominal pain attributable to pancreatic pseudocyst
Gastric or duodenal obstruction
Biliary obstruction
Pancreatic ascites
Development of pleural effusions
Enlarging size on serial abdominal imaging
Pseudocyst infection
Pseudocyst bleeding
Exclusion criteria
Pancreatic cystic neoplasms
Acute peripancreatic fluid collections
Acute necrotic collections
An important step before consideration of drainage is the exclusion of other cystic lesions, such as pancreatic cystic neoplasms. Although pseudocysts account for at least 75% of all pancreatic cystic lesions, they can be difficult to distinguish from pancreatic cystic neoplasms, congenital cysts, and retention cysts, especially in those patients without a clear history of pancreatitis. Such lesions often appear morphologically similar to pseudocysts on cross-sectional imaging, and additional evaluation with endoscopic ultrasonography (EUS) and fine-needle aspiration of the cyst fluid may be necessary before endoscopic drainage.
Pancreatic pseudocysts and walled-off necrosis should also be differentiated from acute peripancreatic fluid collections and acute necrotic collections, both of which occur in the acute phase of pancreatitis or necrotizing pancreatitis. These collections have yet to develop an encapsulated, well-defined wall surrounding the fluid collection, and often resolve with expectant management.
Historically, guidelines have mandated drainage if pseudocysts are present for longer than 6 weeks. This recommendation originated from observational studies of the natural history of pancreatic pseudocysts and complications associated with conservative management. Between 1971 and 1976, Bradley and colleagues followed 54 patients with pancreatic pseudocysts by serial clinical and sonographic examination until either spontaneous resolution, development of complications, or loss to follow-up. During the observation period, 41% of patients developed complications including rupture, abscess, jaundice, and hemorrhage, and 20% developed spontaneous cyst resolution. The investigators surmised that prolonged observation of pancreatic pseudocysts past 7 weeks resulted in risks that exceeded those of elective surgery. In contrast, more recent studies have suggested that longer periods of observation are safe and effective in permitting spontaneous resolution in up to 86% of patients over an average 1-year follow-up, with a 3% to 9% rate of serious complications.
The decision to pursue pseudocyst drainage should not be based on cyst size alone. Although data regarding pseudocyst size and outcomes have been mixed, a cyst of less than 4 cm has been found to be a predictor of spontaneous resolution. In the past, drainage has been indicated for pseudocysts larger than 6 cm because of lower rates of spontaneous resolution and greater risks of complications. A study from Yeo and colleagues of 36 patients with asymptomatic pseudocysts showed that 67% of pseudocysts greater than 6 cm in diameter required surgical treatment, compared with 40% of pseudocysts less than 6 cm in diameter. In contrast, Cheruvu and colleagues showed that the median pseudocyst size of those patients requiring intervention was similar to that of patients who were successfully managed conservatively (8 cm vs 7 cm). Similarly, Nguyen and colleagues determined that a cyst size greater than or less than 6 cm had no effect on rates of spontaneous resolution, need for operative management, complications, cyst recurrence, or mortality. This heterogeneity in data regarding cyst size highlights the primacy of symptoms and regional complications attributable to the cyst when considering cyst drainage.
Surgical drainage
An overview of pancreatic pseudocyst drainage techniques is given in Box 2 . The surgical management of pancreatic pseudocysts depends on the extent of disease and local expertise, and may include cystenterostomy, partial pancreatic resections, and combined laparoscopic and endoscopic interventions. Laparoscopic surgery, such as laparoscopic anterior transgastric cystogastrostomy and lesser sac posterior cystogastrostomy, results in lower morbidity compared with conventional open surgery. Surgical series of patients undergoing laparoscopic pseudocyst drainage have shown success in 95% of cases, with 1% mortality, a 12% complication rate, and 10% conversion rate to open surgery. A study comparing the outcomes of 83 patients who underwent cystogastrostomy by either open, laparoscopic, or endoscopic approaches found comparable overall success rates of more than 90% with open and laparoscopic approaches, with no difference in complication rates.
Surgical
Open surgical drainage
Laparoscopic surgical drainage
Anterior transgastric cystogastrostomy
Lesser sac posterior cystogastrostomy
Percutaneous
Percutaneous irrigation or drainage
Endoscopic
Single or multiple transmural entry (EUS or non-EUS guided) with nasocystic irrigation
Single entry with percutaneous endoscopic gastrostomy for irrigation
Single or multiple transmural entry with plastic or metal stent placement
Transmural entry with endoscopic necrosectomy
Hybrid
Percutaneous irrigation and endoscopic transmural entry
Percutaneous endoscopic direct necrosectomy
Percutaneous drainage
Percutaneous drainage of pancreatic pseudocysts and other peripancreatic fluid collections involves placement of a needle and drainage catheter under ultrasonography or computed tomography (CT) guidance. Following successful drainage of the fluid collection, drainage catheters are kept in place until the daily flow decreases to 5 to 10 mL, with repeat CT imaging confirming resolution of the pseudocyst with the catheter tip remaining within the pseudocyst cavity. At present, percutaneous drainage is preferred for collections that are not adjacent to the gastrointestinal lumen or do not communicate with the pancreatic duct, in patients who have immature infected pseudocysts, or in patients who are poor surgical candidates. Hybrid endoscopic and percutaneous approaches for pancreatic pseudocyst irrigation as well as pancreatic necrosectomy have been described and can be performed in specialized centers under close cooperation between the endoscopist and the interventional radiologist.
Endoscopic drainage
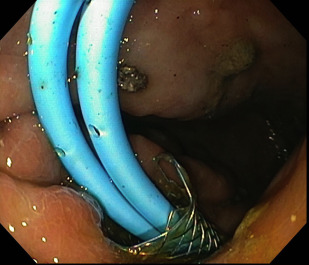
Endoscopic drainage has emerged as the first-line therapy in the management of pancreatic pseudocysts. Compared with surgical and percutaneous drainage, there are multiple advantages to endoscopic drainage, including the ability to place multiple drains, cystic cavity irrigation via nasocystic tubes, and direct endoscopic necrosectomy, all of which are performed via a minimally invasive approach. In addition, ongoing pancreatic duct disruption, leak, or obstruction may be treated via endoscopic retrograde cholangiopancreatography (ERCP) in a single session ( Fig. 1 ). A further advantage is that the development of an enterocystic fistula may reduce the risk of pseudocyst recurrence by allowing pancreatic drainage in cases of disconnected tail syndrome. In addition, endoscopic drainage has been shown to be equally if not more effective than surgical and percutaneous drainage, with lower morbidity and complication rates, particularly with respect to persistent cutaneous fistula formation. An overview of endoscopic pancreatic pseudocyst drainage techniques is given in Box 3 . Endoscopic cystogastrostomy, regardless of the technique used, is performed with fluoroscopic guidance.
Cyst entry
Transpapillary
Diathermic
Seldinger
EUS Guidance
Single-step access device (Navix system)
Cyst dilation
Balloon dilation
Bougie dilation
Stent placement
Double-pigtail plastic stent
Fully covered self-expanding metal stent (SEMS)
Combination double-pigtail plastic stent within fully covered SEMS
Biflanged covered SEMS (Nagi stent)
Lumen-apposing covered SEMS (Niti-S Spaxus and Axios stents)
Transpapillary Drainage
Endoscopic drainage can be performed using either a transpapillary or transmural technique. Endoscopic transpapillary drainage with placement of a pancreatic duct stent is typically reserved for pseudocysts smaller than 6 cm and with communication to the main pancreatic duct. Alternatively, transpapillary stenting is indicated when transmural drainage is not feasible because of contraindications such as coagulopathy, or when the pseudocyst is too distant (>1 cm) from the gastrointestinal lumen to allow safe transmural drainage. Transpapillary stenting may be combined with additional interventions such as major or minor papillotomy, dilation of pancreatic duct strictures, and placement of large-bore pancreatic duct stents across a ductal disruption or into the pseudocyst cavity, if necessary. A combined transmural and transpapillary approach is not typically required for successful resolution of most pancreatic pseudocysts.
Transmural Drainage Without Endoscopic Ultrasound Guidance
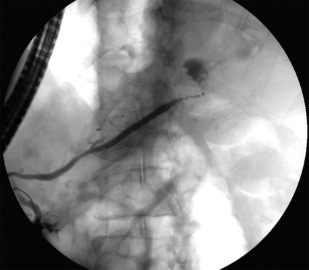
Non–EUS-guided transmural drainage requires close proximity of the pseudocyst to the gastrointestinal lumen, as well as endoscopic localization in the form of a visible luminal bulge ( Fig. 2 ). There are currently 2 well-described methods for non–EUS-guided cyst entry, known as diathermic puncture and the Seldinger technique. Both methods rely on endoscopic needle localization of the point of maximal gastric bulge to confirm the most appropriate location before cystogastrostomy tract dilation and stent placement.
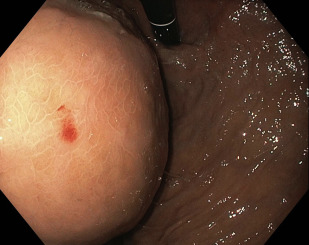
Cyst entry using diathermic puncture involves the use of a needle knife or Cystotome to gain access into and maintain close apposition of the pseudocyst to the gut lumen. The needle knife is directed perpendicularly to the axis of maximal endoscopic bulge. A pure cutting current is then used to gain access into the pseudocyst, with electrocautery discontinued immediately on entry into the cyst cavity to avoid thermal injury to surrounding structures. Once a site is found with suitable fluid return, a small quantity of contrast is injected under fluoroscopic guidance to confirm position within the pseudocyst. Stroking of the needle knife should be avoided, because a cut of even a few millimeters can result in entry of the needle knife into an adjacent gastric vessel. In addition, the needle knife should not extrude consistently in the coaxial plane of cyst entry, because it can result in iatrogenic injury. If blood return is seen once the cyst is punctured, the clinician should immediately consider evaluating for the presence of a pseudoaneurysm or gastric varices.
The Seldinger technique involves creating an initial puncture with an 18-gauge or 19-gauge needle, followed by the introduction of a 0.89-mm (0.035-inch) guidewire through the needle. In a study of 94 patients, the Seldinger technique was shown to have a comparable efficacy to diathermic puncture (95% vs 92%), although with a significantly lower bleeding complication rate (4.6% vs 15.7%).
Once access into the cyst is obtained with a guidewire, it is looped within the cavity to create 2 to 3 coils, and the needle or catheter is exchanged for a dilating balloon (typically 4-mm, 6-mm, 8-mm, or 10 mm balloons). Inflation of the balloon is then performed under fluoroscopic guidance, with the goal of obliteration of the waist of the balloon, to ensure adequate cystogastrostomy tract dilation. Passage of dilation catheters, such as a 6-French to 10-French bougie, can also be used for tract creation without electrocautery, although cautery access is often still required in cases in which there is minimal endoscopic bulge. Following tract creation and dilation, stents may then be placed for pseudocyst drainage.
Endoscopic Ultrasonography–guided Transmural Drainage
EUS is increasingly used to guide transmural drainage. EUS can be used to exclude pancreatic cystic neoplasms and pseudoaneurysms, and provides real-time image guidance to identify relative contraindications to endoscopic drainage such as gastric varices, cyst-lumen distance greater than 1 cm, and normal intervening pancreatic parenchyma. A single-step EUS-guided approach is the most commonly used method for pancreatic pseudocyst drainage. The EUS-guided method involves endosonographically guided puncture of the pseudocyst with a 19-gauge fine-needle aspiration needle. Subsequently, the stylet is withdrawn, cyst aspiration and contrast injection is performed, and a 0.89-mm (0.035-inch) guidewire is placed through the needle for tract dilatation and stent placement ( Fig. 3 ).
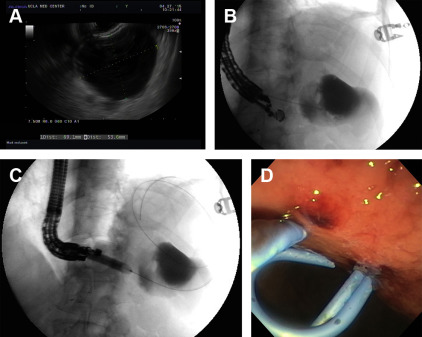
More recently, a single-step exchange-free access device (Navix; Xlumena, Mountain View, CA) was developed for transluminal pseudocyst drainage. This device comprises an endoscopic trocar with a blade that creates a 3.5-mm puncture opening, an anchor balloon that maintains access within the target, a dilation balloon that expands the tract to 10 mm, and 2 guidewire ports for subsequent stent placement. Following EUS-guided transmural entry into the pseudocyst with the trocar, the balloon catheter is then advanced over the trocar, followed by inflation of the anchor balloon. A 0.89-mm (0.035-inch) guidewire is then inserted into the cyst cavity, and the tract is dilated to 10 mm with the dilation balloon. After dilation, a second 0.89-mm (0.035-inch) guidewire is inserted. The access device is then removed from the endoscope, and a 7-French followed by 10-French double-pigtail stent can be inserted in sequence across the guidewire and into the cystogastrostomy tract for drainage. The size of the working channel of current endoscopes limits the first stent to 7-French diameter. Other methods for 1-step simultaneous double-wire pancreatic pseudocyst drainage have also been described.
Stent Placement
A variety of different stents are available for pancreatic pseudocyst drainage, including plastic pigtail stents, covered self-expanding metal stents (SEMS), and new LAMS. Based on the currently available literature, the standard of practice in endoscopic drainage of pancreatic pseudocysts remains the placement of multiple double-pigtail plastic stents, with or without a fully covered SEMS.
Transmural pancreatic pseudocyst drainage with plastic stents was first described in 1998, and covered SEMS were introduced in 2010 for transmural necrosectomy. Plastic stents have disadvantages, including a small lumen diameter, which may result in stent occlusion and need for reintervention, hence typically multiple plastic stents are placed in tandem.
SEMS have a much larger luminal diameter than plastic stents and therefore may facilitate greater drainage. However stent migration and tissue injury from stent erosion into either the wall of the pseudocyst or gastrointestinal tract are significant potential adverse events. To prevent migration, double-pigtail plastic stents, ranging in size from 7 French to 10 French, are often placed within the lumen of the covered SEMS to act as an anchor and prevent migration ( Fig. 4 ).