Several new devices and innovative adaptations of existing modalities have emerged as primary, adjunctive, or rescue therapy in endoscopic hemostasis of gastrointestinal hemorrhage. These techniques include over-the-scope clip devices, hemostatic sprays, cryotherapy, radiofrequency ablation, endoscopic suturing, and endoscopic ultrasound–guided angiotherapy. This review highlights the technical aspects and clinical applications of these devices in the context of nonvariceal upper gastrointestinal bleeding.
Key points
- •
Emerging techniques for endoscopic hemostasis of nonvariceal upper gastrointestinal bleeding include over-the-scope clips, hemostatic sprays, mucosal ablation devices (cryotherapy and radiofrequency ablation), endoscopic suturing, and endoscopic ultrasound–guided angiotherapy.
- •
These techniques may be used as primary or rescue therapy, depending on the types of lesions targeted and the response to standard hemostatic modalities.
- •
An understanding of the technical aspects and limitations of these devices as well as proper patient and lesion selection helps optimize the utilization of these techniques in nonvariceal upper gastrointestinal bleeding.
Introduction
Injection therapy (eg, epinephrine and sclerosant), thermal coagulation with contact (eg, bipolar) and noncontact (eg, argon plasma coagulation [APC]) probes, and mechanical devices (eg, hemoclips and band ligation) are established modalities for the endoscopic management of nonvariceal upper gastrointestinal bleeding (NVUGIB) lesions. Although these techniques are often effective at achieving hemostasis, the benefit is not always sustained or their application not feasible because of the location, characteristics, and/or extent of the bleeding lesion. The introduction of novel hemostatic devices and innovative use of existing techniques are likely to enhance primary or rescue therapy for NVUGIB lesions that are not amenable, or refractory, to standard therapy. In this article, the techniques and clinical applications of these emerging modalities is highlighted from a device perspective.
Introduction
Injection therapy (eg, epinephrine and sclerosant), thermal coagulation with contact (eg, bipolar) and noncontact (eg, argon plasma coagulation [APC]) probes, and mechanical devices (eg, hemoclips and band ligation) are established modalities for the endoscopic management of nonvariceal upper gastrointestinal bleeding (NVUGIB) lesions. Although these techniques are often effective at achieving hemostasis, the benefit is not always sustained or their application not feasible because of the location, characteristics, and/or extent of the bleeding lesion. The introduction of novel hemostatic devices and innovative use of existing techniques are likely to enhance primary or rescue therapy for NVUGIB lesions that are not amenable, or refractory, to standard therapy. In this article, the techniques and clinical applications of these emerging modalities is highlighted from a device perspective.
Over-the-scope clips
Devices and Techniques
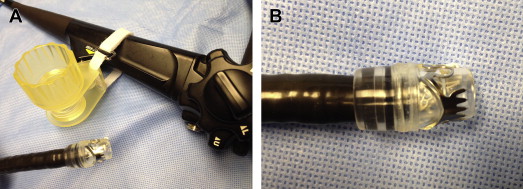
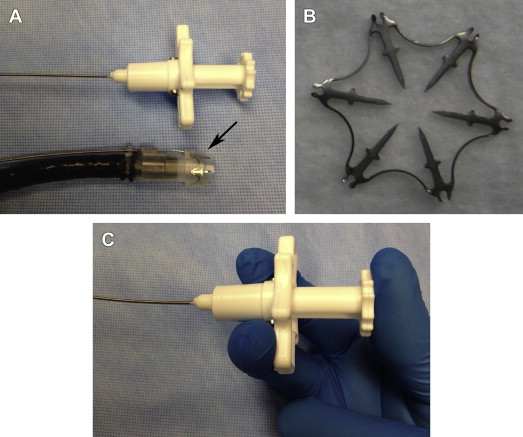
Over-the-scope (OTS) clips radically differ in design compared with standard through-the-scope (TTS) clips. In experimental studies, OTS clips provided more secure vascular closure because of the enhanced tissue capture and greater compressive force than TTS clips. Two types of OTS clips are currently available. The over-the-scope clip (OTSC, Ovesco Endoscopy AG, Tubingen, Germany) is akin to a bear claw and offers a short learning curve because its setup and deployment are similar to a band ligator. The nitinol-based OTSC is preloaded in a bent state over a clear cap ( Fig. 1 ). OTSC caps are available in 3 diameters (11, 12, and 14 mm) and 2 working depths (3 and 6 mm). The clips also come in 3 types of teeth (atraumatic [a], traumatic [t], and gastrostomy closure [gc]) ( Fig. 2 ). Both the a (atraumatic or blunt-toothed) and t (traumatic or sharp-toothed) clips have been used for the treatment of focal NVUGIB lesions. The a-type clip is adequate for most pliable bleeding lesions, especially in the thin-walled esophagus. The bite of the t-type clip likely enables more secure anchoring into indurated tissue, such as a fibrotic ulcer base. Similar to band ligation, the OTSC is affixed to the tip of the endoscope; clip deployment is achieved by triggering a string wire connected to a rotating hand wheel that is fastened to the entrance port of the working channel. Suction is applied to engage the target lesion into the cap before release of the clip, which assumes its naturally closed state ( [CR] ). For indurated lesions (eg, chronic ulcers), a dedicated tripronged anchoring device can assist in retracting the ulcer base into the cap ( Fig. 3 ).

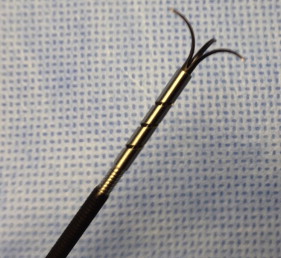
The Padlock clip (Aponos Medical Corp, Kingston, NH) is another recently approved OTS clip whose trigger wire is located alongside the shaft of the endoscope. This design, which does not use the working channel, allows more efficient suction of the target lesion into the cap, passage of other accessories, and continued suction of blood and secretions. Deployment of the clip is achieved by squeezing a handheld device ( Fig. 4 ). The Padlock clip has shown reliable closure of defects in a porcine survival study, although clinical usage remains limited. As yet, there are no published data regarding its application in the management of NVUGIB lesions.
Clinical Applications
The reported efficacy and safety of OTSC for the treatment of NVUGIB lesions are limited to a handful of single reports and small case series. The overall success rate ranges from 71% to 100% for the treatment of various bleeding lesions, including Mallory-Weiss tears, peptic ulcers ( Fig. 5 ), Dieulafoy lesions ( Fig. 6 ), anastomotic ulcers, eroding tumors, and postpolypectomy ulcers ( Fig. 7 ).
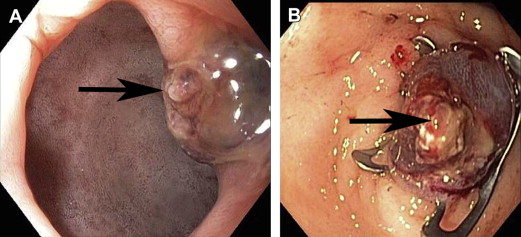
In some studies, OTSC was used primarily as salvage therapy for NVUGIB lesions in which conventional hemostatic therapies had failed. In one retrospective series, OTSC was successful in securing hemostasis in 91% of cases (n = 23), which consisted of duodenal ulcers (n = 12), gastric ulcers (n = 6), Mallory-Weiss tears (n = 2), Dieulafoy lesions (n = 2), and a surgical anastomotic bleed (n = 1). The 2 failures involved peptic ulcers, and there were no adverse events reported. In another series of 9 patients with refractory or major bleeding from nonvariceal upper gastrointestinal (GI) lesions, the technical and clinical success (absence of rebleeding) rates were 100% and 77.8%, respectively. Lesions included gastric ulcers (n = 2), duodenal ulcers (n = 5), a gastric stromal tumor (n = 1), and luminal ulceration secondary to pancreatic cancer (n = 1). The 2 failures involved duodenal ulcers. The OTSC retention time was 28 days (range 0–42).
The OTSC seems useful either as a primary modality or as a rescue therapy for NVUGIB lesions that are refractory to standard therapies. However, disadvantages of the device include the need to withdraw the endoscope to mount the OTSC, potential difficulty in traversing the upper esophageal sphincter or luminal strictures, clip misplacement that interferes with subsequent therapy, and difficulty accessing lesions in certain locations (eg, posterior-inferior wall of duodenum and gastric lesser curve). Its application may also be impaired when attempting to capture firm and/or thick lesions (eg, deeply penetrating fibrotic ulcer) into the OTSC cap despite the assistance of devices for retraction.
Although the cost of a single OTSC device is several-fold higher than that of a conventional TTS clip, the former may be more cost-effective when considering the comparative outcomes, such as the rebleeding rate or number of clips required to achieve hemostasis. Prospective randomized trials and cost-efficacy analyses comparing OTS clip devices with traditional techniques (eg, TTS clips and coaptive coagulation) are anticipated.
Hemostatic sprays
Devices and Techniques
Hemostatic dressings or gauzes are an essential component of the military’s first-aid kits for control of external wound bleeding in the battlefield. These dressings incorporate hemostatic agents, derived from chitosan and minerals, in the form of granules and powders.
One such proprietary compound, TC-325, is an inorganic absorbent powder that forms an adherent coagulum at the targeted site; this agent has been integrated into a pressurized carbon dioxide (CO 2 ) gas–propelled, push-button device for endoscopic hemostasis ( Fig. 8 ) (Hemospray, Cook Medical Inc, Bloomington, IN).
The adoption of hemostatic sprays as part of endoscopic therapy has been facilitated by their ease of use. Unlike conventional contact (eg, bipolar probe) and noncontact (eg, APC) thermal modalities, the application of hemostatic sprays requires less precision and can be delivered to poorly accessible anatomic sites, such as the proximal lesser curve of the stomach and posterior wall of the duodenum. Given the mechanism of action, the use of hemostatic sprays is limited to lesions or surface areas that are actively bleeding ( Fig. 9 ). The Hemospray powder is delivered in short, 1- to 2-second bursts to the target site via a TTS catheter ( [CR] ). In one study, up to 150 g of the powder (21 g per cartridge) was allowed per treatment session, although the maximum tolerated dose that can be administered in a single session has not been determined. The coagulum usually sloughs off in a few days and is naturally excreted. Although endoscopic visualization is hampered during Hemospray application, the therapeutic benefit remains because precise targeting is not required. However, the delivery of subsequent therapies, should bleeding persist, is hindered by the cloudy field of view and adherent coagulum. Hemospray is commercially available in several countries but is not yet cleared by the Food and Drug Administration for use in the United States.
Plant-derived hemostatic agents, such as the Ankaferd Blood Stopper (ABS; Ankaferd Health Products Ltd, Istanbul, Turkey), have been described for endoscopic applications. ABS is an herbal extract approved in Turkey for topical treatment of dental and postsurgical external hemorrhage. It induces formation of an encapsulated proteinlike mesh that serves as a scaffold for rapid red blood cell aggregation without significantly interfering with individual coagulation factors or platelets. The ABS solution is sprayed onto the bleeding site until a yellowish-grey adherent coagulum is formed. The amount required (2–25 mL) and the time to hemostasis (seconds to minutes) vary according to bleeding severity and targeted surface area. A polysaccharide-based hemostatic spray agent (EndoClot Plus Inc, Santa Clara, CA) that could be applied endoscopically has been described, although there are no published data regarding its use for nonvariceal upper GI bleeding.
Clinical Applications
In the first prospective pilot study regarding Hemospray, initial hemostasis was achieved in 95% of patients (n = 20) with actively bleeding peptic ulcers. Recurrent bleeding occurred in 2 patients for an overall success rate of 85%. Clean base ulcers without residual coagulum were noted during second-look endoscopy at 72 hours. No adverse events were reported at the 1-month follow-up. A European multicenter registry study, presented in abstract form, included 71 patients treated with Hemospray as monotherapy (55%), first-line combination therapy (8%), or rescue therapy (34%) for a variety of NVUGIB lesions that included peptic ulcers (58%), esophageal ulcers (4%), tumors (7%), Dieulafoy lesions (4%), after mucosectomy (3%), gastric antral vascular ectasia (GAVE) (3%), and miscellaneous causes (21%). Primary hemostasis was achieved in 85% of patients; adverse events were mostly technical in nature, including occlusion of the spray catheter or instrument channel (n = 5), endoscope adhesion to the mucosa (n = 2), and device failure (n = 1). Hemospray has also been used with variable success for tumor bleeding, an often difficult condition to treat endoscopically.
Several single case reports and small case series originating primarily from Turkey have described the hemostatic potential of ABS for various NVUGIB lesions, including Mallory-Weiss tears, Dieulafoy lesions, GAVE, anastomotic ulcers, sphincterotomy sites, postpolypectomy sites, and tumor bleeding.
The available evidence supports the use of hemostatic sprays as the primary or bridge therapy, particularly in the management of oozing-type lesions. The efficacy of these agents may be limited in the setting of brisk bleeding because of the rapid wash-away effect of the hemostatic agent and in patients receiving antithrombotic therapy who present with spurting arterial bleeding. Given their mechanism of action, hemostatic agents do not seem to be of value for the management of nonactively bleeding lesions.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







