Mucosal disorders
Esophagitis
Occult acid reflux disease
Gastritis
Duodenitis
Peptic ulcer disease
H. pylori infection
Gastric cancer
Neuromuscular dysfunction
Gastroparesis
Visceral hypersensitivity
Impaired fundic relaxation
Abnormalities in the brain-gut axis
Others
Psychological disorders
Medications
Abdominal wall pain
Hepatobiliary disorders
Pancreatic disorders
Vascular disorders (median arcuate ligament syndrome; SMA syndrome)
Ischemic heart disease
Functional dyspepsia accounts for the majority of patients with dyspeptic symptoms; approximately 70 % of dyspeptic patients have a normal initial diagnostic evaluation. The Rome III criteria (see Table 6.2) currently define FD as the presence of symptoms thought to originate in the gastroduodenal region in the absence of any organic, systemic, or metabolic disease likely to explain the symptoms. The Rome committee recently introduced two new subcategories for FD into their diagnostic criteria: postprandial distress syndrome (PDS) and epigastric pain syndrome (EPS). Importantly, while epigastric pain or discomfort is the hallmark symptom in patients with FD, many patients with FD will not complain of pain but will rather state that they have burning, pressure, or fullness in their epigastric area or that they cannot finish a normal-sized meal (early satiety). Postprandial nausea, belching, and abdominal bloating are also often present in FD. Patients with abdominal pain or discomfort relieved with defecation are currently excluded from the Rome III FD definition as these symptoms are most consistent with the diagnosis of irritable bowel syndrome (IBS; see Chap. 16). It is important to note that there is a large overlap between FD and IBS, and approximately 30–40 % of patients with FD have overlapping IBS, while approximately 40 % of patients with IBS have overlapping FD.
Table 6.2
Rome III criteria for functional dyspepsia (Modified from Tack et al 2006)
For the 2 categories noted below, criteria must be fulfilled for the last 3 months with symptom onset at least 6 months prior to diagnosis. Patients must have one or more of the following symptoms: |
Postprandial fullness |
Early satiety |
Epigastric burning |
In addition, patients cannot have any evidence of structural disease that is likely to explain symptoms (i.e., upper endoscopy is normal) |
A. Postprandial distress syndrome |
Diagnostic criteria must include both of the following: |
Bothersome postprandial fullness, occurring after ordinary sized meals, at least several times per week |
Early satiation that prevents finishing a regular meal, at least several times per week |
Other supporting criteria: |
Upper abdominal bloating or postprandial nausea or excessive belching can be present |
Epigastric pain syndrome may coexist |
B. Epigastric pain syndrome |
Diagnostic criteria must include all of the following: |
Pain or burning localized to the epigastrium, of at least moderate severity at least once per week |
The pain is intermittent |
Not generalized or localized to other abdominal or chest regions |
Not relieved by defecation or passage of flatus |
Not fulfilling criteria for biliary pain |
Supportive criteria: |
The pain may be of a burning quality, but without a retrosternal component |
The pain is commonly induced or relieved by ingestion of a meal, but may occur while fasting |
Postprandial distress syndrome may coexist |
Epidemiology
Dyspepsia is one of the most commonly encountered problems in medical practice. Population studies from the United States and Europe demonstrate that 20–25 % of adults suffer from dyspepsia at any given time. Once investigated, most patients with dyspeptic symptoms are diagnosed with FD. In these populations, the prevalence of FD is approximately 12–15 %, while its incidence is 2–5 %. The natural history of FD is not well understood, likely because it is a disorder of multiple, potentially coincident pathophysiologies. Patient cohort analyses report a wide range of time to resolution, from 30 to 50 % resolution at 1–2 years post-diagnosis to 80 % with persistent symptoms at 18–24 months.
Pathophysiology
The underlying pathophysiology of FD includes heightened or otherwise abnormal visceral sensation (see Fig. 6.1) and abnormalities in gastric emptying and fundic accommodation. On gastric scintigraphy, 30–40 % of FD patients have a mild delay in gastric emptying, while 5–10 % demonstrate rapid gastric emptying. Impaired fundic accommodation, identified by a gastric barostat or single-photon emission computed tomography (SPECT) study (only performed at specialized centers), is present in 30–40 % of patients with FD. Some patients with FD are likely hypersensitive to normal amounts of gastric acid, which may explain the small but positive response of dyspeptic patients to treatment with histamine type 2 receptor antagonists (H2RA’s) or proton pump inhibitors (PPIs). Others are hypersensitive to duodenal lipids or to the presence of duodenal eosinophilia.
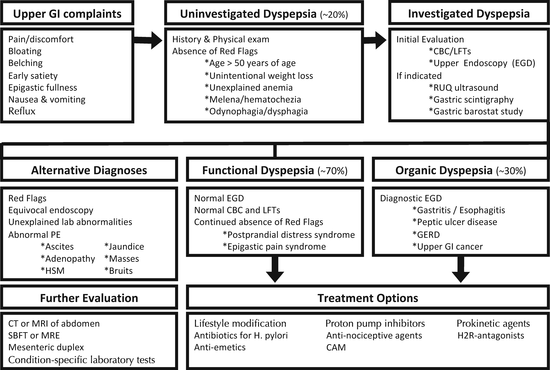

Fig. 6.1
The pathophysiology of FD
Infection with Helicobacter pylori (H. pylori) has long been implicated in the pathogenesis of dyspepsia. The prevalence of H. pylori is, however, generally the same between subjects with and without dyspepsia, and several large, randomized, placebo-controlled trials have shown little or no long-term benefit in the resolution of FD symptoms after eradication of H. pylori. Finally, FD may share the postinfectious etiology that is well described in patients with IBS. Although neither anxiety nor depression directly causes FD, their coexistence may exacerbate the experience and increase the reporting of FD symptoms.
Diagnosis and Evaluation
Because the differential diagnosis of dyspepsia is broad (see Table 6.1 and Fig. 6.2), a careful history and thorough physical examination are important first steps to distinguish dyspepsia from other disorders and to help determine whether dyspeptic symptoms represent an underlying organic process or are functional in nature. A careful review of the patient’s medication list should be performed as nonsteroidal anti-inflammatory agents (NSAIDs), aspirin products, antibiotics, alternative medications, and iron supplements can cause dyspeptic symptoms. The presence of classic symptoms of pyrosis and regurgitation makes gastroesophageal reflux disease the most likely diagnosis. Postprandial pain or discomfort raises the possibility of peptic ulcer disease and, when coupled with unintentional weight loss, raises the possibility of gastroparesis, gastric outlet obstruction, or even gastric cancer. Realizing that dyspeptic symptoms are nonspecific, clinicians should be particularly attentive to “red flags” in the history or on physical examination that may suggest the presence of underlying systemic or malignant disease (see Table 6.3). For example, findings of hepatosplenomegaly, ascites, adenopathy and masses, jaundice, an abdominal bruit, or a succussion splash should make the clinician consider alternative diagnoses and initiate an appropriate investigation.
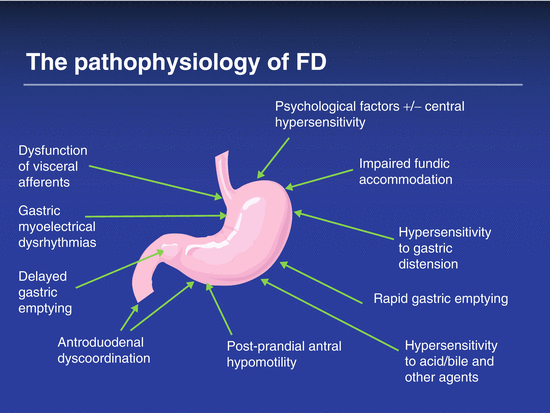





Fig. 6.2
Diagnostic algorithm
Table 6.3
Warning symptoms and signs in the evaluation of dyspepsia
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







