Fig. 12.1
Results of two-dimensional electrophoresis and immunoblot demonstrating that circulating albumin in children migrates in the basic range of pH (lane 1–4) (Debiec et al. [18])
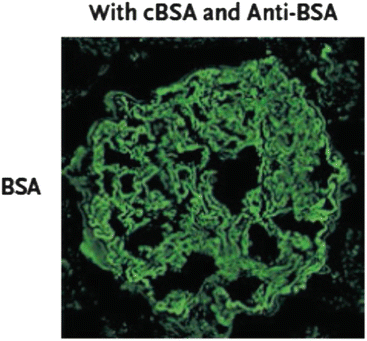
Fig. 12.2
Expression of bovine serum albumin (BSA) in renal biopsy specimen. Panel A shows biopsy specimen of patient with circulating cationic BSA (Debiec et al. [18])
Epitope mapping revealed that the target was BSA peptide 147–161; controls showed a broader spectrum of reactivity. By looking at the localization, subepithelial immune deposits of BSA were only detected in children with circulating c-BSA and high titers of anti-BSA antibodies. Lastly, after eluting IgG from one BSA deposit-positive biopsy specimen, only the combination of serum BSA and IgG4 and IgG1 caused the development of deposits, but there was no reactivity with albumin. Typical characteristic predominance of IgG4 and IgG1 for MN was present.
In summary, the study indicates that in young children c-BSA is a potential cause of MN. A TH-2 response typical for MN is one requisite and leads to IgG4 and IgG1 activation resulting in IgG4 deposits. The facts that there were no sequence homologies of BSA peptides 147–161 and podocyte antigens indicate that biochemical cross-reaction of anti-BSA with podocytes is most unlikely.
12.5 Open Questions of Debiec Study
Despite the important findings of the study by Debiec et al., several questions remain open. For instance, the authors do not present any data on how BSA enters the circulation and how c-BSA is subsequently formed prior to the development of the pathogenetic cascade. The role of food processing and the impact of gastric pH are discussed, but conclusive data are not available. It is most likely, but not proven, that these patients ingested BSA. Also, it is not clear why not all patients with high titers subsequently develop c-BSA, which ultimately results in renal disease with BSA deposits.
It is of interest that adults with high titers to BSA do not have any c-BSA in their circulation and thus do not develop BSA deposits. This could be due to different gastrointestinal handling of BSA in adults vs. children but other explanations possible, such as different immunological processing. Interestingly, the neonatal period is characterized by the absence of memory cells, which may be one reason why Th2-type reactions develop [19]. This could explain why an antigen, such as c-BSA, results in a Th2 immune response that ultimately leads to deposition of IgG4. On the other hand, in adults with MN non-c-BSA is able to elicit an IgG4 response; however deposits containing BSA do not develop.
12.6 Implications: Clinical and Treatment
Debiec et al. suggest that dietary interventions may be a therapeutic approach in patients with MN related to c-BSA, since in humans BSA is mainly ingested with food, e.g., milk.
Allergic reactions to BSA are rare but have been described after ingestion; respiratory symptoms may occur after inhalation. Allergy to BSA has been found in 13–20 % of children with cow’s milk allergy, who also have beef allergy [20]. In the series by Martelli and colleagues, 92.9 % of children with beef allergy were allergic to cow’s milk, and only children nonallergic to cow’s milk were not sensitized to BSA. These phenomena relate to IgE antibodies, and no data concerning simultaneous IgG antibody reactions to cow’s milk or BSA are available [20].
In the study by Debiec et al., no single dietary compound could be identified in the diets of four affected infants. Importantly, these data were collected retrospectively after other interventions had been initiated, mainly immunosuppression.
Anti-BSA antibodies have been involved in nonrenal disorders, such as rheumatoid arthritis [21] and multiple sclerosis [22], where they interact with collagen or myelin basic protein, respectively.
Thus, it was intriguing to study the role of BSA as an environmental antigen in the pathogenesis of MN. Theoretically children and especially infants may be prone to allergy caused by environmental (food) allergens, such as BSA, since the gastrointestinal barrier is not yet matured. This allows BSA to enter the circulation. In addition, gastric pH is higher in infants and may have an impact on protein degradation [23]. It should not be forgotten that BSA can also be inhaled and then cause hypersensitivity [24].
Effects of diet, e.g., gluten-free, dairy-free diet, on proteinuric diseases such as the childhood nephrotic syndrome have been discussed [25]. In theory, also a diet rich or poor in BSA may have an effect on glomerular disorders, although so far no systematic data are available. Intestinal interactions may be complex, and the immune system may be involved as well as gut microbiome, epigenetics, and individual genetic factors. Therefore, at present it seems too early to speculate about any beneficial dietary interventions to treat or prevent glomerular disease, including MN.
12.7 New Models of Advancing the Basic Understanding and Treatment of c-BSA-Associated MN
In general, the door to discover novel treatment modalities of MN is the generation of robust and solid murine models and comparison of these results to other in vivo models, e.g., the Heymann nephritis rat model [26]. These models may allow a better understanding of the injury itself (complement activation, podocyte injury) that ultimately lead to disruption of the glomerular basement membrane (GBM). In addition, genetic mechanisms can be more easily studied in murine models.
12.8 Genetics and Potential Biomarkers of c-BSA MN from a Murine Model to Human Disease
Wu et al. used cDNA microarray analysis to identify gene expression changes in MN caused by c-BSA [27]. Highly expressed genes were also evaluated as markers both in mice and human kidney samples. They identified 175 genes that were differentially expressed in the MN kidneys compared to healthy kidneys. Four genes [metallothionein-1 (Mt1), cathepsin D (CtsD), lymphocyte 6 antigen complex (Ly6), and laminin receptor-1 (Lamr1)] were selected and quantified. While Mt1 was mainly localized in tubules, Lamr1 and CtsD were expressed in glomeruli, and interestingly high expressions of Lamr1 and CtsD also occurred in human kidney biopsies with MN. Thus, the BSA murine model resembled the clinical and pathological features of human MN very closely even at an ultrastructural level.
The genetic background of the BSA-associated models was studied by Chen et al. [28]. Only ICR and BALB/c mice developed MN, and this was unrelated to any production of circulating immune complexes; concentration of anti c-BSA antibodies on the other hand had a strong impact. TH2 may predominate in these mouse models and could be related to IgG1 production.
12.9 Kinetics of Adaptive Immunity to c-BSA-Induced MN
Another study addressing the immunologic mechanism was reported by Wu et al. [29] who induced MN with c-BSA using a T1/T2 double transgenic mice model. These animals express human Thy1 protein under the control of interferon-gamma (IFN-gamma) and mouse Thy1.1 protein under the control of interleukin (IL)-4. The authors demonstrated a progressive Th2 response in the spleen which was followed by a compensatory Th1 response and also an augmented IL-4-production of Th2 cells and IFN-gamma-producing Th1 cells. Furthermore the authors demonstrated an increase of Th2 markers in the circulation and kidney, in addition to the upregulation of Th2 cytokines. There was a correlation of Th2, but not Th1 markers, with proteinuria and cholesterol. Taken together, the c-BSA mouse model correlates well with the human setting and may help to identify further immunological mechanisms. These findings will be ultimately translated for the better understanding and treatment of this disease.
12.10 Experimental Treatment Options
The potential usefulness of c-BSA mouse models in respect to the development of new treatment options is highlighted by the experiments of Wu et al. looking at heme oxygenase-1 (HO-1) induction in BSA-induced MN [30]. HO-1 is a rate-limiting enzyme that oxidizes heme into carbon monoxide, ferritin, and biliverdin and is induced in response to various stimuli. It exhibits cytoprotective effects but also anti-inflammatory, antioxidant, antiapoptotic, and possible immunomodulatory function. Thus, induction of HO-1 may be an attractive option in the treatment of MN. After induction of MN with c-BSA a potent HO-1 inducer, cobalt protoporphyrin in different concentrations was able to reduce the clinical consequences of MN such as proteinuria and glomerular lesions. Immune complex formation and deposition also were significantly reduced, due to decreased production and deposition of proinflammatory cytokines, antioxidative mechanisms, and antiapoptotic mechanisms. Taken together this study demonstrates that the mouse model is able to develop new treatment options, e.g., HO-1. This is also supported by a study, where HO-1 upregulation was able to suppress intrarenal cytokine production potentially slowing down the progression of chronic kidney disease [31].
Immunosuppressive treatment is often necessary and effective in MN. Kobayashi et al. studied the effects of FK506 in MN induced by c-BSA and demonstrated that pre-immunization with FK506 suppressed the development of MN. If FK506 was given after MN, developed histological lesions were comparable to untreated animals; however proteinuria was significantly improved, even if administration was delayed for 4 weeks. Thus, calcineurin inhibition is effective in experimental MN, especially if administered early [32].
12.11 Treatment of c-BSA-Associated MN: Beyond Diet
As discussed above, dietary interventions for the treatment or prevention of c-BSA-associated MN have not been studied so far. In addition, there is at present no experimental model showing that ingestion of c-BSA can cause any MN. Lastly, there have been no further clinical reports suggesting the beneficial impact of dietary intervention in c-BSA MN since the publication of Debiec et al., suggesting that human c-BSA-associated MN is very rare.
Interestingly in the study by Debiec et al., immunosuppression played an important role in the treatment of four children associated with c-BSA, supposedly because the potential antigen was detected much later. As indicated in the supplemental files, all children received steroids, and one child was treated with additional mycophenolate and cyclosporine. Response was rapid in most patients, and three of four patients are in complete remission after 3.5–8 years of follow-up, underlining that prompt institution of immunosuppression has a beneficial effect. While most patients entered remission within months after immunosuppression, one patient had a protracted course, despite addition of mycophenolate and one had a relapse after of cyclosporine. There is no information, whether in this patient still had high titers of anti-c-BSA antibodies and whether further modification of diet was considered as treatment option. It would have been interesting to re-biopsy this patient, since in the initial biopsy c-BSA deposits could not be tested. Interestingly two patients had relapses of their nephrotic syndrome that responded to immunosuppression again, which might have been due to continued BSA sensitization with production of c-BSA.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




